Abstract
Background: Genetic mutation is responsible for approximately 10% of breast cancers. The purpose of this study was to compare breast cancer survival and recurrence rates between BRCA1/2 mutation carriers and noncarriers.
Methods: Using the Columbia Presbyterian breast cancer database, we collected the tissue blocks of all patients younger than 65 years of age and of Jewish descent. The patients were contacted and the data updated. DNA was extracted from the tissue blocks and tested for the common mutations. The results of the genetic mutation and updated database were anonymized and merged. The survival and recurrence rates were compared between mutation carriers and noncarriers.
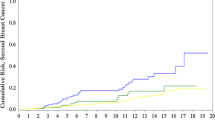
Results: A total of 739 breast cancer cases in 715 patients were identified. We were able to test 487 patients. We identified 30 BRCA1 and 21 BRCA2 mutation carriers, for an incidence of 10.36%. The median follow-up for the patients tested was 50 months. BRCA1 patients more frequently had estrogen- and progesterone-negative tumors and had a higher incidence of positive nodes. BRCA1 patients received chemotherapy more frequently. The incidence of in situ disease was similar for mutation and non–mutation carriers. BRCA1/2 mutation carriers had a higher incidence of bilateral disease. There was no difference in 5- or 10-year overall and breast cancer–specific survival between mutation and non–mutation carriers.
Conclusions: Breast cancer patients with BRCA1/2 mutations have a similar outcome as non–mutation carriers.
Similar content being viewed by others
References
Newman B, Mu H, Butler ML, et al. Frequency of breast cancer attributable to BRCA1 in a population-based series of American women. JAMA 1998;279:915–921.
Malone KE, Daling JR, Neal C, et al. Frequency of BRCA1/BRCA2 mutations in a population based sample of young breast carcinoma cases. Cancer 2000;88:1393–1402.
Fodor F, Weston A, Bleiweiss I, et al. Frequency and carrier risk associated with common BRCA1 and BRCA2 mutations in Ashkenazi Jewish breast cancer patients. Am J Hum Genet 1998;63:45–51.
Warner E, Foulkes W, Goodwin P, et al. Prevalence and penetrance of BRCA1 and BRCA2 gene mutations in unselected Ashkenazi Jewish women with breast cancer. J Natl Cancer Inst 1999;91:1241–1247.
Robson M, Rajan P, Rosen PP, et al. BRCA-associated breast cancer: absence of a characteristic immunophenotype. Cancer Res 1998;58:1839–1842.
Marcus JN, Watson P, Page DL, et al. Hereditary breast cancer: pathobiology, prognosis, and BRCA1 and BRCA2 gene linkage. Cancer 1996;77:697–709.
Porter DE, Dixon M, Smyth E, Steel CM. Breast cancer survival in BRCA1 carriers. Lancet 1993;341:184–185.
Porter DE, Cohen BB, Wallace MR, et al. Breast cancer incidence, penetrance and survival in probable carriers of BRCA1 germline mutation in families linked to BRCA1 on chromosome 17q12–21. Br J Surg 1994;81:1512–1515.
Verhoog LC, Brekelmans CT, Seynaeve C, et al. Survival and tumour characteristics of breast-cancer patients with germline mutations of BRCA1. Lancet 1998;351:316–321.
Johannsson OT, Ranstam J, Borg A, Olsson H. Survival of BRCA1 breast and ovarian cancer patients: a population-based study from southern Sweden. J Clin Oncol 1998;16:397–404.
Robson M, Gilewski T, Haas B, et al. BRCA-associated breast cancer in young women. J Clin Oncol 1998;16:1642–1649.
Ansquer Y, Gautier C, Fourquet A, Asselain B, Stoppa-Lyonnet D. Survival in early-onset BRCA1 breast-cancer patients. Institut Curie Breast Cancer Group. Lancet 1998;352:541.
Robson M, Levin D, Federici M, et al. Breast conservation therapy for invasive breast cancer in Ashkenazi women with BRCA founder mutations. J Natl Cancer Inst 1999;91:2112–2117.
Foulkes WD, Wong N, Brunet JS, et al. Germ-line BRCA1 mutation is an adverse prognostic factor in Ashkenazi Jewish women with breast cancer. Clin Cancer Res 1997;3:2465–2469.
Foulkes WD, Chappuis PO, Wong N, et al. Primary node negative breast cancer in BRCA1 mutation carriers has a poor outcome. Ann Oncol 2000;11:307–313.
Karp SE, Tonin PN, Begin LR, et al. Influence of BRCA1 mutations on nuclear grade and receptor status of breast carcinoma in Ashkenazi Jewish women. Cancer 1997;80:435–441.
Armes JE, Trute L, White D, et al. Distinct molecular pathogenesis of early-onset breast cancers in BRCA1 and BRCA2 mutation carriers: a population based study. Cancer Res 1999;59:2011–2017.
Breast Cancer Linkage Consortium. Pathology of familial breast cancer: differences between breast cancers in carriers of BRCA1 and BRCA2 mutations and sporadic cases. Lancet 1997;349:1505–10.
Lakhani SR, Jacquemier J, Sloane JP, et al. Multifactorial analysis of differences between sporadic breast cancers and cancers involving BRCA1 and BRCA2 mutations. J Natl Cancer Inst 1998;90:1138–1145.
Armes JE, Venter DJ. The pathology of inherited breast cancer. Pathology 2002;34:309–314.
Lakhani SR, van de Vijver MJ, Jacquemier J, et al. The pathology of familial breast cancer: predictive value of immunohistochemical markers estrogen receptor, progesterone receptor, HER-2, and p53 in patients with mutations in BRCA1 and BRCA2. J Clin Oncol 2002;20:2310–2318.
Levine DA, Federici MG, Reuter VE, Boyd J. Cell proliferation and apoptosis in BRCA-associated hereditary ovarian cancer. Gynecol Oncol 2002;85:431–434.
Jazaeri AA, Yee CJ, Sotiriou C, et al. Gene expression profiles of BRCA1-linked, BRCA2-linked, and sporadic ovarian cancers. Natl Cancer Inst 2002;94:990–1000.
Eisinger F, Stoppa-Lyonnet D, Londy M, et al. Germ line mutation at BRCA1 affects the histoprognostic grade in hereditary breast cancer. Cancer Res 1996;56:471–474.
Pierce LJ, Strawderman M, Narod SA, et al. Effect of radiotherapy after breast-conserving treatment in women with breast cancer and germline BRCA1/2 mutations. J Clin Oncol 2000;18:3360–3369.
Haffty BG, Harrold E, Khan AJ, et al. Outcome of conservatively managed early-onset breast cancer by BRCA1/2 status. Lancet 2002;359:1471–1477.
Early Breast Cancer Trialists’ Collaborative Group. Systemic treatment of early breast cancer by hormonal, cytotoxic, or immune therapy. 133 randomized trials involving 31,000 recurrences and 24,000 deaths among 75,000 women. Lancet 1992;339:1–15.
Hoogerbrugge N, Bult P, de Widt-Levert JM, et al. High prevalence of premalignant lesions in prophylactically removed breasts from women at hereditary risk for breast cancer. J Clin Oncol 2003;21:41–45.
Ren Q, Carbonne C, Potoczek M, et al. Differential response to BRCA1 wild type and BRCA1 mutant breast cancer cells to ionizing radiation and paclitaxel (abstract). Clin Cancer Res 2001; 7(Suppl):3723s.
Fan S, Yuan R, Ma YX, et al. Mutant BRCA1 genes antagonize phenotype of wild-type BRCA1. Oncogene 2001;20:8215–8235.
Bhattacharyya A, Ear US, Koller BH, et al. The breast cancer susceptibility gene BRCA1 is required for subnuclear assembly of Rad51 and survival following treatment with the DNA cross-linking agent cisplatin. J Biol Chem 2000;275:23899–23903.
Husain A, He G, Venkatraman ES, et al. BRCA1 up-regulation is associated with repair-mediated resistance to cis-diamminedichloroplatinum(II). Cancer Res 1998;58:1120–1123.
Abbott DW, Freeman MI, Holt JT. Double-strand repair deficiency and radiation sensitivity in BRCA2 mutant cancer cells. J Natl Cancer Inst 1998;90:978–985.
Freneaux P, Stoppa-Lyonnet D, Mouret E, et al. Low expression of bcl-2 in BRCA1-associated cancers. Br J Cancer 2000;83:1318–1322.
Chappuis PO, Goffin J, Wong N, et al. A significant response to neoadjuvant chemotherapy in BRCA1/2 related breast cancer. J Med Genet 2002;39:608–610.
Goffin JR, Chappuis PO, Begin LR, et al. Impact of germline BRCA1 mutations and overexpression of p53 on prognosis and response to treatment following breast carcinoma. Cancer 2003;97:527–536.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Tamer, M., Russo, D., Troxel, A. et al. Survival and Recurrence After Breast Cancer in BRCA1/2 Mutation Carriers. Ann Surg Oncol 11, 157–164 (2004). https://doi.org/10.1245/ASO.2004.05.018
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1245/ASO.2004.05.018