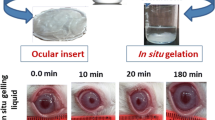
Abstract
The bioavailability of therapeutic agents from eye drops is usually limited due to corneal barrier functions and effective eye protective mechanisms. Therefore, the current study aims to enhance ocular bioavailability of brimonidine, a potent antiglaucoma drug, through the preparation of ocular inserts. Solvent casting technique was employed to prepare the inserts using polyvinylpyrrolidone K-90 (PVP K-90) as film-forming polymer blended with different viscosity grades of bioadhesive polymers namely hydroxypropyl methycellulose, carbopol, sodium alginate, and chitosan. The prepared ocular inserts were evaluated for various physicochemical parameters, swelling behavior, and in vitro release patterns. Sodium alginate-based ocular inserts revealed the most sustainment in drug release (99% at 6 h), so it was selected for further modifications via coating it, on one side or dual sides, using hydrophobic film composed of either ethylcellulose or Eudragit RSPO. The obtained in vitro release results for the modified ocular inserts revealed that ethylcellulose is superior to Eudragit RSPO in terms of brimonidine release sustainment effect. Ocular inserts composed of 7% PVP K-90, 1.5% low molecular weight sodium alginate with or without ethylcellulose coat were able to sustain the in vitro release of brimonidine. Their therapeutic efficacy regarding intraocular pressure (IOP) lowering effect when inserted in albino rabbits eyes showed superior sustainment effect compared with that of brimonidine solution. Furthermore, due to both the mucoadhesive property and the drug sustainment effect, the one-side-coated ocular insert showed more IOP lowering effect compared with that of its non-coated or dual-side-coated counterpart.









Similar content being viewed by others
References
Mundada AS, Shrikhande BK. Design and evaluation of soluble ocular drug insert for controlled release of ciprofloxacin hydrochloride. Drug Dev Ind Pharm. 2006;32(4):443–8.
Ali Y, Lehmussaari K. Industrial perspective in ocular drug delivery. Adv Drug Deliv Rev. 2006;58(11):1258–68.
Wilson CG, Zhu YP, Frier M, Rao LS, Gilchrist P, Perkins AC. Ocular contact time of a carbomer gel (GelTears) in humans. Br J Ophthalmol. 1998;82(10):1131–4.
Wei G, Xu H, Ding PT, Li SM, Zheng JM. Thermosetting gels with modulated gelation temperature for ophthalmic use: the rheological and gamma scintigraphic studies. J Control Release. 2002;83(1):65–74.
Lee VH, Robinson JR. Topical ocular drug delivery: recent developments and future challenges. J Ocul Pharmacol. 1986;2(1):67–108.
Arici MK, Arici DS, Topalkara A, Guler C. Adverse effects of topical antiglaucoma drugs on the ocular surface. Clin Exp Ophthalmol. 2000;28(2):113–7.
Gurtler F, Kaltsatos V, Boisrame B, Gumy R. Long-acting soluble bioadhesive ophthalmic drug insert (BODI) containing gentamicin for veterinary use: optimization and clinical investigation. J Control Release. 1995;33:231–6.
Hornof M, Weyenberg W, Ludwig A, Bernkop-Schnurch A. Mucoadhesive ocular insert based on thiolated poly(acrylic acid): development and in vivo evaluation in humans. J Control Release. 2003;89(3):419–28.
Malhotra M, Majumdar DK. Aqueous, oil, and ointment formulations of ketorolac: efficacy against prostaglandin E2-induced ocular inflammation and safety: a technical note. AAPS PharmSciTech. 2006;7(4):96.
Sintzel M, Bernatchez S, Tabatabay C, Gurny R. Biomaterials in ophthalmic drug delivery. J Pharm Biopharm. 1996;42:258–374.
Qi H, Chen W, Huang C, Li L, Chen C, Li W, et al. Development of a poloxamer analogs/carbopol-based in situ gelling and mucoadhesive ophthalmic delivery system for puerarin. Int J Pharm. 2007;337(1–2):178–87.
Saettone MF, Salminen L. Ocular inserts for topical delivery. Adv Drug Deliv Rev. 1995;16:95–106.
Koelwel C, Rothschenk S, Fuchs-Koelwel B, Gabler B, Lohmann C, Gopferich A. Alginate inserts loaded with epidermal growth factor for the treatment of keratoconjunctivitis sicca. Pharm Dev Technol. 2008;13(3):221–31.
Deshpande PB, Dandagi P, Udupa N, Gopal SV, Jain SS, Vasanth SG. Controlled release polymeric ocular delivery of acyclovir. Pharm Dev Technol. 2010;15(4):369–78.
Resnikoff S, Pascolini D, Etya’ale D, Kocur I, Pararajasegaram R, Pokharel GP, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82(11):844–51.
Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006;90(3):262–7.
De TK, Rodman DJ, Holm BA, Prasad PN, Bergey EJ. Brimonidine formulation in polyacrylic acid nanoparticles for ophthalmic delivery. J Microencapsul. 2003;20(3):361–74.
Burke J, Manlapaz C, Kharlamb A, Runde E, Padillo E, Spada C, et al. Therapeutic use of α2-adrenoceptor agonists in glaucoma. In: Lanier S, Limbird L, editors. α2-Adrenergic receptors: structure, function and therapeutic implications. Reading: Harwood Academic Publishers; 1996. p. 179–87.
Munk SA, Harcourt D, Arasasingham P, Gluchowski C, Wong H, Burke J, et al. Analogs of UK 14,304: Structural features responsible for α2 adrenoceptor activity. Bioorganic Med Chem Lett. 1995;5(15):1745–50.
Alphagan product leaflet. Allergan New Zealand Ltd: Allergan, Inc 2009.
Gilhotra RM, Gilhotra N, Mishra DN. Piroxicam bioadhesive ocular inserts: physicochemical characterization and evaluation in prostaglandin-induced inflammation. Curr Eye Res. 2009;34(12):1065–73.
El-Gendy NA, Abdelbary GA, El-Komy MH, Saafan AE. Design and evaluation of a bioadhesive patch for topical delivery of gentamicin sulphate. Curr Drug Deliv. 2009;6(1):50–7.
Juliano C, Cossu M, Pigozzi P, Rassu G, Giunchedi P. Preparation, in vitro characterization and preliminary in vivo evaluation of buccal polymeric films containing chlorhexidine. AAPS PharmSciTech. 2008;9(4):1153–8.
U.S. Department of Health and Human Services. Guidance for industry: dissolution testing of immediate release solid oral dosage forms. Food and Drug Administration Center for Drug Evaluation and Research (CDER) 1997.
Chandak AR, Verma PRP. Development and evaluation of HPMC based matrices for transdermal patches of Tramadol. Clin Res Regul Aff. 2010;6:1370–9.
Jain D, Carvalho E, Banerjee R. Biodegradable hybrid polymeric membranes for ocular drug delivery. Acta Biomater. 2010;6(4):1370–9.
Bialer M, Yacobi A, Moros D, Levitt B, Houle JM. Criteria to assess in vivo performance and bioequivalence of generic controlled-release formulations of carbamazepine. Epilepsia. 1998;39:513–9.
Panomsuk SP, Hatanaka T, Aiba T, Katayama K, Koizumi T. A study of the hydrophilic cellulose matrix: effect of drugs on swelling properties. Chem Pharm Bull. 1996;44:1039–42.
Eouani C, Piccerelle P, Prinderre P, Bourret E, Joachim J. In-vitro comparative study of buccal mucoadhesive performance of different polymeric films. Eur J Pharm Biopharm. 2001;52(1):45–55.
Alanazi FK, Abdel Rahman AA, Mahrous GM, Alsarra IA. Formulation and physicochemical characterization of buccoadhesive films containing ketorolac. J Drug Del Sci. 2007;17(3):183–92.
Cafaggi S, Leardi R, Parodi B, Caviglioli G, Russo E, Bignardi G. Preparation and evaluation of a chitosan salt-poloxamer 407 based matrix for buccal drug delivery. J Control Release. 2005;102(1):159–69.
Joshi HN, Wilson TD. Calorimetric studies of dissolution of hydroxypropyl methylcellulose E5 (HPMC E5) in water. J Pharm Sci. 1993;82(10):1033–8.
Hamza Yel S, Aburahma MH. Design and in vitro evaluation of novel sustained-release double-layer tablets of lornoxicam: utility of cyclodextrin and xanthan gum combination. AAPS PharmSciTech. 2009;10(4):1357–67.
Singh KH, Shinde UA. Development and Evaluation of Novel Polymeric Nanoparticles of Brimonidine Tartrate. Curr Drug Deliv 2011; (in press).
Patel RP, Patel MM. Physicochemical characterization and dissolution study of solid dispersions of Lovastatin with polyethylene glycol 4000 and polyvinylpyrrolidone K30. Pharm Dev Technol. 2007;12(1):21–33.
Xiao C, Lu Y, Liu H, Zhang L. Preparation and physical properties of blend films from sodium alginate and polyacrylamide solutions. Macromol Sci Pure Appl Chem. 2000;A37(12):1663–75.
Çaykara T, Demirci S, Kantoğlu Ö. Thermal, spectroscopic, and mechanical properties of blend films of poly(N–vinyl-2-pyrrolidone) and sodium alginate. Polymer Plast Tech Eng. 2007;46(7):737–41.
Phromsopha T, Baimark Y. Methoxy poly(ethylene glycol)-b-poly(d, l-lactide) films for controlled release of ibuprofen. Trends Appl Sci Res. 2009;4:107–15.
Elmotasem H. Chitosan–alginate blend films for the transdermal delivery of meloxicam. Asian J Pharm Sci. 2008;3(1):12–29.
Badr-Eldin SM, Elkheshen SA, Ghorab MM. Inclusion complexes of tadalafil with natural and chemically modified beta-cyclodextrins. I: preparation and in-vitro evaluation. Eur J Pharm Biopharm. 2008;70(3):819–27.
Ruan LP, Yu BY, Fu GM, Zhu DN. Improving the solubility of ampelopsin by solid dispersions and inclusion complexes. J Pharm Biomed Anal. 2005;38(3):457–64.
Redenti E, Peveri T, Zanol M, Ventura P, Gnappi G, Montenero A. A study on the differentiation between amorphous piroxicam:β-cyclodextrin complex and a mixture of the two amorphous components. Int J Pharm. 1996;129:289–94.
Kaur IP, Smitha R. Penetration enhancers and ocular bioadhesives: two new avenues for ophthalmic drug delivery. Drug Dev Ind Pharm. 2002;28(4):353–69.
Ludwig A. The use of mucoadhesive polymers in ocular drug delivery. Adv Drug Deliv Rev. 2005;57(11):1595–639.
Sasaki H, Nagano T, Sakanaka K, Kawakami S, Nishida K, Nakamura J, et al. One-side-coated insert as a unique ophthalmic drug delivery system. J Control Release. 2003;92(3):241–7.
Declaration of Interest
The authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aburahma, M.H., Mahmoud, A.A. Biodegradable Ocular Inserts for Sustained Delivery of Brimonidine Tartarate: Preparation and In Vitro/In Vivo Evaluation. AAPS PharmSciTech 12, 1335–1347 (2011). https://doi.org/10.1208/s12249-011-9701-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9701-3