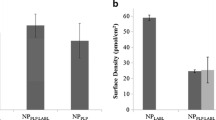
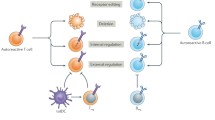
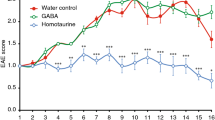
Abstract
Autoimmune diseases such as multiple sclerosis (MS) are characterized by the breakdown of immune tolerance to autoantigens. Targeting surface receptors on immune cells offers a unique strategy for reprogramming immune responses in autoimmune diseases. The B7 signaling pathway was targeted using adaptations of soluble antigen array (SAgA) technology achieved by covalently linking B7-binding peptides and disease causing autoantigen (proteolipid peptide (PLP)) to hyaluronic acid (HA). We hypothesized that co-delivery of a B7-binding peptide and autoantigen would suppress experimental autoimmune encephalomyelitis (EAE), a murine model of MS. Three independent B7-targeted SAgAs were created containing peptides to either inhibit or potentially stimulate the B7 signaling pathway. Surprisingly, all SAgAs were found to suppress EAE disease symptoms. Altered cytokine expression was observed in primary splenocytes isolated from SAgA-treated mice, indicating that SAgAs with different B7-binding peptides may suppress EAE through different immunological mechanisms. This antigen-specific immunotherapy using SAgAs can successfully suppress EAE through co-delivery of autoantigen and peptides targeting with the B7 signaling pathway.




Similar content being viewed by others
Abbreviations
- HA:
-
Hyaluronic acid
- SAgA:
-
Soluble antigen array
- PLP:
-
Proteolipid peptide
- B7AP:
-
B7 antisense peptide
- CD80-CAP:
-
CD80 competitive antagonist peptide
- SAgAPLP:LABL :
-
Soluble antigen array co-grafted with PLP and LABL peptides
- SAgAPLP:B7AP :
-
Soluble antigen array co-grafted with PLP and B7AP peptides
- SAgAPLP:CD80-CAP :
-
Soluble antigen array co-grafted with PLP and CD80-CAP peptides
- SAgAPLP:sF2 :
-
Soluble antigen array co-grafted with PLP and sF2 peptides
REFERENCES
Fletcher JM, Lalor SJ, Sweeney CM, Tubridy N, Mills KH. T cells in multiple sclerosis and experimental autoimmune encephalomyelitis. Clin Exp Immunol. 2010;162(1):1–11.
Ransohoff RM, Brown MA. Innate immunity in the central nervous system. J Clin Invest. 2012;122(4):1164–71.
Amor S, Puentes F, Baker D, Van Der Valk P. Inflammation in neurodegenerative diseases. Immunology. 2010;129(2):154–69.
Feldmann M, Steinman L. Design of effective immunotherapy for human autoimmunity. Nature. 2005;435(7042):612–9.
Bates D. Treatment effects of immunomodulatory therapies at different stages of multiple sclerosis in short-term trials. Neurology. 2011;76:S14–25.
Miller SD, Turley DM, Podojil JR. Antigen-specific tolerance strategies for the prevention and treatment of autoimmune disease. Nat Rev Immunol. 2007;7(9):665–77.
Langer-Gould A, Steinman L. Progressive multifocal leukoencephalopathy and multiple sclerosis: lessons from natalizumab. Curr Neurol Neurosci Rep. 2006;6(3):253–8.
Kindt TJ, Goldsby RA, Osborne BA, Kuby J. Kuby immunology. 6th ed. New York: W.H. Freeman and Company; 2007.
Gonsette RE. Self-tolerance in multiple sclerosis. Acta Neurol Belg. 2012;112(2):133–40.
Barnett MH, Sutton I. The pathology of multiple sclerosis: a paradigm shift. Curr Opin Neurol. 2006;19(3):242–7.
Bromley SK, Iaboni A, Davis SJ, Whitty A, Green JM, Shaw AS, et al. The immunological synapse and CD28-CD80 interactions. Nat Immunol. 2001;2(12):1159–66.
Sharpe AH, Freeman GJ. The B7-CD28 superfamily. Nat Rev Immunol. 2002;2(2):116–26.
Chittasupho C, Siahaan TJ, Vines CM, Berkland C. Autoimmune therapies targeting costimulation and emerging trends in multivalent therapeutics. Ther Deliv. 2011;2(7):873–89.
Badawi AH, Siahaan TJ. Immune modulating peptides for the treatment and suppression of multiple sclerosis. Clin Immunol. 2012;144(2):127–38.
Chittasupho C, Sestak J, Shannon L, Siahaan TJ, Vines CM, Berkland C. Hyaluronic acid graft polymers displaying peptide antigen modulate dendritic cell response in vitro. Mol Pharm. 2013;11(1):367–73. doi: 10.1021/mp4003909.
Sabatos-Peyton CA, Verhagen J, Wraith DC. Antigen-specific immunotherapy of autoimmune and allergic diseases. Curr Opin Immunol. 2010;22(5):609–15.
Yao S, Zhu Y, Chen L. Advances in targeting cell surface signalling molecules for immune modulation. Nat Rev Drug Discov. 2013;12(2):130–46.
Chen J, He Q, Zhang R, Chu Y, Wang Y, Liu Q, et al. Allogenic donor splenocytes pretreated with antisense peptide against B7 prolong cardiac allograft survival. Clin Exp Immunol. 2004;138(2):245–50.
Srinivasan M, Lu D, Eri R, Brand DD, Haque A, Blum JS. CD80 binding polyproline helical peptide inhibits T cell activation. J Biol Chem. 2005;280(11):10149–55.
Dudhgaonkar SP, Janardhanam SB, Kodumudi KN, Srinivasan M. CD80 blockade enhance glucocorticoid-induced leucine zipper expression and suppress experimental autoimmune encephalomyelitis. J Immunol. 2009;183(11):7505–13.
Srinivasan M, Gienapp IE, Stuckman SS, Rogers CJ, Jewell SD, Kaumaya PT, et al. Suppression of experimental autoimmune encephalomyelitis using peptide mimics of CD28. J Immunol. 2002;169(4):2180–8.
Kobayashi N, Kobayashi H, Gu L, Malefyt T, Siahaan TJ. Antigen-specific suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor. J Pharmacol Exp Ther. 2007;322(2):879–86.
Murray JS, Oney S, Page JE, Kratochvil-Stava A, Hu Y, Makagiansar IT, et al. Suppression of type 1 diabetes in NOD mice by bifunctional peptide inhibitor: modulation of the immunological synapse formation. Chem Biol Drug Des. 2007;70(3):227–36.
Sestak J, Mullins M, Northrup L, Thati S, Forrest ML, Siahaan TJ, et al. Single-step grafting of aminooxy-peptides to hyaluronan: a simple approach to multifunctional therapeutics for experimental autoimmune encephalomyelitis. J Control Release. 2013;168(3):334–40.
Sestak JO, Sullivan BP, Thati S, Northrup L, Hartwell B, Antunez L, et al. Codelivery of antigen and an immune cell adhesion inhibitor is necessary for efficacy of soluble antigen arrays in experimental autoimmune encephalomyelitis. Mol Ther Methods Clin Dev. 2014. doi:10.1038/mtm.2014.8.
Manikwar P, Kiptoo P, Badawi AH, Büyüktimkin B, Siahaan TJ. Antigen-specific blocking of CD4-Specific immunological synapse formation using BPI and current therapies for autoimmune diseases. Med Res Rev. 2012;32(4):727–64.
Badawi AH, Kiptoo P, Wang W-T, Choi I-Y, Lee P, Vines CM, et al. Suppression of EAE and prevention of blood–brain barrier breakdown after vaccination with novel bifunctional peptide inhibitor. Neuropharmacology. 2012;62(4):1874–81.
Ridwan R, Kiptoo P, Kobayashi N, Weir S, Hughes M, Williams T, et al. Antigen-specific suppression of experimental autoimmune encephalomyelitis by a novel bifunctional peptide inhibitor: structure optimization and pharmacokinetics. J Pharmacol Exp Ther. 2010;332(3):1136–45.
Constantinescu CS, Farooqi N, O’Brien K, Gran B. Experimental autoimmune encephalomyelitis (EAE) as a model for multiple sclerosis (MS). Br J Pharmacol. 2011;164(4):1079–106.
Yao YF, Zhou YM, Xiang JB, Gu XD, Cai D. Inhibition of arterial allograft intimal hyperplasia using recipient dendritic cells pretreated with B7 antisense peptide. Clin Dev Immunol. 2012;2012:892687.
Fukumoto T, Torigoe N, Kawabata S, Murakami M, Uede T, Nishi T, et al. Peptide mimics of the CTLA4-binding domain stimulate T-cell proliferation. Nat Biotechnol. 1998;16(3):267–70.
Lutterotti A, Yousef S, Sputtek A, Sturner KH, Stellmann JP, Breiden P, et al. Antigen-specific tolerance by autologous myelin peptide-coupled cells: a phase 1 trial in multiple sclerosis. Sci Transl Med. 2013;5(188):188ra75.
Bar-Or A, Vollmer T, Antel J, Arnold DL, Bodner CA, Campagnolo D, et al. Induction of antigen-specific tolerance in multiple sclerosis after immunization with DNA encoding myelin basic protein in a randomized, placebo-controlled phase 1/2 trial. Arch Neurol. 2007;64(10):1407–15.
Kang Y, Xu L, Wang B, Chen A, Zheng G. Cutting edge: immunosuppressant as adjuvant for tolerogenic immunization. J Immunol. 2008;180(8):5172–6.
Kang Y, Zhao J, Liu Y, Chen A, Zheng G, Yu Y, et al. FK506 as an adjuvant of tolerogenic DNA vaccination for the prevention of experimental autoimmune encephalomyelitis. J Gene Med. 2009;11(11):1064–70.
Cousens LP, Su Y, McClaine E, Li X, Terry F, Smith R, et al. Application of IgG-derived natural Treg epitopes (IgG Tregitopes) to antigen-specific tolerance induction in a murine model of type 1 diabetes. J Diabetes Res. 2013;2013:621693.
Eri R, Kodumudi KN, Summerlin DJ, Srinivasan M. Suppression of colon inflammation by CD80 blockade: evaluation in two murine models of inflammatory bowel disease. Inflamm Bowel Dis. 2008;14(4):458–70.
Deppong CM, Bricker TL, Rannals BD, Van Rooijen N, Hsieh CS, Green JM. CTLA4Ig inhibits effector T cells through regulatory T cells and TGF-beta. J Immunol. 2013;191(6):3082–9.
Ward FJ, Dahal LN, Wijesekera SK, Abdul-Jawad SK, Kaewarpai T, Xu H, et al. The soluble isoform of CTLA-4 as a regulator of T-cell responses. Eur J Immunol. 2013;43(5):1274–85.
Yamamoto T, Hattori M, Yoshida T. Induction of T-cell activation or anergy determined by the combination of intensity and duration of T-cell receptor stimulation, and sequential induction in an individual cell. Immunology. 2007;121(3):383–91.
Petitto JM, Streit WJ, Huang Z, Butfiloski E, Schiffenbauer J. Interleukin-2 gene deletion produces a robust reduction in susceptibility to experimental autoimmune encephalomyelitis in C57BL/6 mice. Neurosci Lett. 2000;285(1):66–70.
Rodgers JM, Miller SD. Cytokine control of inflammation and repair in the pathology of multiple sclerosis. Yale J Biol Med. 2012;85(4):447–68.
Lambrecht BN, Kool M, Willart MA, Hammad H. Mechanism of action of clinically approved adjuvants. Curr Opin Immunol. 2009;21(1):23–9.
Peek LJ, Middaugh CR, Berkland C. Nanotechnology in vaccine delivery. Adv Drug Deliv Rev. 2008;60(8):915–28.
Acknowledgments
This work was supported by the NIH (1R56AI091996-01A1), KINBRE (P20 RR016475/P20 GM103418), American Foundation for Pharmaceutical Education (AFPE) Pre-Doctoral Fellowship in Clinical Pharmaceutical Science, and the Takeru Higuchi Graduate Fellowship (University of Kansas). In addition, the authors thank the laboratories of C. Russell Middaugh and David Volkin for use of laboratory equipment and Lorena Antunez, Cavan Kalonia, and Brian Kaiser for their contributions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editor: Aliasger Salem
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
The molecular weight of each SAgA was calculated from GPC data. (A) The retention time of each SAgA was used to determine the molecular weight as compared to a (B) pullulan polymer standard curve. Results shown are an average from triplicate injections of a single batch preparation. All samples had a RSD ≤ 0.01. (DOCX 32 kb)
Supplemental Figure 2
SAgAPLP:sF2 was analyzed for subvisible particulate formation at a concentration of 0.1 mg/ml in PBS using micoflow imaging (MFI). Representative images of insoluble aggregates from 25 to 70 μm equivalent circular diameter are shown. (DOCX 1558 kb)
Supplemental Figure 3
ICAM-1 and B7-targeted SAgAs were found to reduce disease incidence in EAE mice. EAE was induced on day zero, and mice were treated on days four, seven, and ten with a dose of SAgA equivaent to 200 nMol PLP. Treatments of HA were administered at a dose equivalent of the SAgAs, 29 nMol. EAE disease incidence was evaluated such that disease free animals maintained a clinical score <1. In all the SAgA treatment groups, over half of the animals remained disease free over the course of the study, while in both negative control groups all animals became diseased. (n = 6 mice per group) (DOCX 152 kb)
Rights and permissions
About this article
Cite this article
Northrup, L., Sestak, J.O., Sullivan, B.P. et al. Co-Delivery of Autoantigen and B7 Pathway Modulators Suppresses Experimental Autoimmune Encephalomyelitis. AAPS J 16, 1204–1213 (2014). https://doi.org/10.1208/s12248-014-9671-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-014-9671-y