Abstract
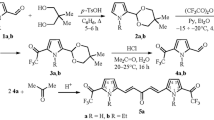
Chemo- and stereoselectivity of the reaction of 1-[2-(vinyloxy)ethyl]-1H-pyrroles with trifluoroacetic anhydride have been studied. The reaction with an equimolar amount of trifluoroacetic anhydride chemoselectively involves the free α-position of the pyrrole ring with formation of the corresponding α-trifluoroacetylpyrroles. In the reaction with 2 equiv of trifluoroacetic anhydride, acylation of both α-position of the pyrrole ring and β-position of the vinyloxy group leads to the formation of 1-(2-{[(1E)-4,4,4-trifluoro-3-oxobut-1-en-1-yl]oxy}ethyl)-2-trifluoroacetyl derivatives with high stereoselectivity.
Similar content being viewed by others
References
Purser, S., Moore, P.R., Swallow, S., and Gouverneur, V., Chem. Soc. Rev., 2008, vol. 37, p. 320.
Giornal, F., Pazenok, S., Rodefeld, L., Lui, N., Vors, J.-P., and Leroux, F.R., J. Fluorine Chem., 2013, vol. 152, p. 2.
Kirsch, P., Modern Fluoroorganic Chemistry: Synthesis, Reactivity, Applications, Weinheim: Wiley-VCH, 2013, 2nd ed., p. 245.
Wang, J., Sánchez-Roselló, M., Aceña, J.L., del Pozo, C., Sorochinsky, A.E., Fustero, S., Soloshonok, V.A., and Liu, H., Chem. Rev., 2014, vol. 114, p. 2432.
Bassetto, M., Ferla, S., and Pertusati, F., Future Med. Chem., 2015, vol. 7, p. 527.
Müller, K., Faeh, C., and Diederich, F., Science, 2007, vol. 317, p. 1881.
Zhu, W., Wang, J., Wang, S., Gu, Z., Aceña, J.L., Izawa, K., Liu, H., and Soloshonok, V.A., J. Fluorine Chem., 2014, vol. 167, p. 37.
Nenajdenko, V., Muzalevskiy, V., and Serdyuk, O., Fluorine in Heterocyclic Chemistry, Nenajdenko, V, Ed., Heidelberg: Springer, 2014, vol. 1, p. 55.
Clementi, S. and Marino, G., Tetrahedron, 1969, vol. 25, p. 4599.
Clementi, S. and Marino, G.J., Chem. Soc., Perkin Trans. 2, 1972, p. 71.
Yarmoliuk, D.V., Arkhipov, V.V., Stambirskyi, M.V., Dmytriv, Y.V., Shishkin, O.V., Tolmachev, A.A., and Mykhailiuk, P.K., Synthesis, 2014, vol. 46, p. 1254.
Nenajdenko, V.G. and Balenkova, E.S., Arkivoc, 2011, part (i), p. 246.
Trofimov, B.A., Schmidt, E.Yu., and Sobenina, L.N., Chemistry of Pyrroles, Boca Raton, FL: CRC, 2014, p. 272.
Anderson, H.J. and Loader, C.E., Pyrroles. Part One: The Synthesis and the Physical and Chemical Aspects of the Pyrrole Ring, Jones, R.A., Ed., Hoboken, NJ: Wiley, 1990, p. 398.
Hojo, M., Masuda, R., Kokuryo, Y., Shioda, H., and Matsuo, S., Chem. Lett., 1976, vol. 5, p. 499.
Hojo, M., Masuda, R., and Okada, E., Synthesis, 1990, no. 4, p. 347.
Kelly, C.B., Mercadante, M.A., and Leadbeater, N.E., Chem. Commun., 2013, vol.49, p. 11 133.
Abdel-Aal, Y. and Hammock, B., Science, 1986, vol. 233, p. 1073.
Sadykov, E.K., Lobanova, N.A., and Stankevich, V.K., Synthesis, 2015, vol. 47, p. 67.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.Kh. Sadykov, N.A. Lobanova, V.K. Stankevich, 2016, published in Zhurnal Organicheskoi Khimii, 2016, Vol. 52, No. 4, pp. 547–551.
Rights and permissions
About this article
Cite this article
Sadykov, E.K., Lobanova, N.A. & Stankevich, V.K. Reaction of 1-[2-(vinyloxy)ethyl]-1H-pyrroles with trifluoroacetic anhydride. Russ J Org Chem 52, 533–537 (2016). https://doi.org/10.1134/S1070428016040096
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428016040096