Abstract
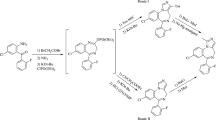
A new retrosynthetic procedure was developed for the synthesis of the macrocyclic core of a cytotoxic lactone (+)-neopeltolide utilizing cyclopropanol intermediates. The synthesis was suggested and carried out of the C7–C16 segment of (+)-neopeltolide to obtain (4S,6S)-6-[(2S)-2-hydroxypentyl]-4-methyltetrahydro-2H-pyran-2-one. The possibility was demonstrated of a formal synthesis based on the obtained product of the potential antitumor pharmaceutical (+)-neopeltolide.
Similar content being viewed by others
References
Wright, A.E., Botelho, J.C., Guzman, E., Harmody, D., Linley, P., McCarthy, P.J., Pitts, T.P., Pomponi, S.A., and Reed, J.K., J. Nat. Prod., 2007, vol. 70, p. 412.
Youngsaye, W., Lowe, J.T., Pohlki, F., Ralifo, P., and Panek, J.S., Angew. Chem., Int. Ed., 2007, vol. 46, p. 9211.
Custar, D.W., Zabawa, T.P., and Scheidt, K.A., J. Am. Chem. Soc., 2008, vol. 130, p. 804.
Ulanovskaya, O.A., Janjic, J., Suzuki, M., Sabharwal, S.S., Schumacker, P.T., Kron, S.J., and Kozmin, S.A., Nat. Chem. Biol., 2008, vol. 4, p. 418.
Woo, S.K., Kwon, M.S., and Lee, E., Angew.Chem., Int. Ed., 2008, vol. 47, p. 3242.
Vintonyak, V.V. and Maier, M.E., Org. Lett., 2008, vol. 10, p. 1239.
Fuwa, H., Naito, S., Goto, T., and Sasaki, M., Angew. Chem., Int. Ed., 2008, vol. 47, p. 4737.
Paterson, I. and Miller, N.A., Chem. Commun., 2008, p. 4708.
Kartika, R., Gruffi, Tr., and Taylor, R.E., Org. Lett., 2008, vol. 10, p. 5047.
Vintonyak, V.V., Kunze, B., Sasse, F., and Maier, M.E., Chem. Eur. J., 2008, vol. 14, p. 11132.
Gallon, J., Reymond, S., and Cossy, J., C. R. Chimie, 2008, p. 1463.
Custar, D.W., Zabawa, T.P., Hines, J., Crews, C.M., and Scheidt, K.A., J. Am. Chem. Soc., 2009, vol. 131, p. 12406.
Tu, W. and Floreancig, P.E., Angew. Chem., Int. Ed., 2009, vol. 48, p. 4567.
Kim, H., Park, Y., and Hong, J., Angew. Chem., Int. Ed., 2009, vol. 48, p. 7577.
Guinchard, X. and Roulland, E., Org. Lett., 2009, vol. 11, p. 4700.
Fuwa, H., Saito, A., Naito, S., Konoki, K., Yotsu- Yamashita, M., and Sasaki, M., Chem. Eur. J., 2009, vol. 15, p. 12807.
Yadav, J.S., Kumar, G.G., and Kumar, S.N., Tetrahedron, 2010, vol. 66, p. 480.
Cui, Y., Wangyang, T., and Floreancig, P.E., Tetrahedron, 2010, vol. 66, p. 4867.
Martinez-Solorio, D. and Jennings, M.P., J. Org. Chem., 2010, vol. 75, p. 4095.
Fuwa, H., Saito, A., and Sasaki, M., Angew. Chem., Int. Ed., 2010, vol. 49, p. 3041.
Florence, G.J. and Cadou, R.F., Tetrahedron Lett., 2010, vol. 51, p. 5761.
Hartmann, E. and Oestreich, M., Angew. Chem., Int. Ed., 2010, vol. 49, p. 6195.
Yang, Z., Zhang, B., Zhao, G., Yang, J., Xie, X., and She, X., Org. Lett., 2011, vol. 13, p. 5916.
Cui, Y., Balachandran, R., Day, B.W., and Floreancig, P.E., J. Org. Chem., 2012, vol. 77, p. 2225.
Gangavaram, S., Reddy, S., and Kallaganti, V.S.R., Org. Biomol. Chem., 2012, vol. 10, p. 3689.
Raghavan, S. and Samanta, P.K., Org. Lett., 2012, vol. 14, p. 2346.
Athe, S., Chandrasekhar, B., Roy, S., Pradhan, T.K., and Ghosh, S.J., Org. Chem., 2012, vol. 77, p. 9840.
Fuwa, H., Kawakami, M., Noto, K., Muto, T., Suga, Y., Konoki, K., Yotsu-Yamashita, M., and Sasaki, M., Chem. Eur. J., 2013, vol. 19, p. 8100.
Ghosh, A.K., Shurrush, K.A., and Dawson, Z.L., Org. Biomol. Chem., 2013, vol. 11, p. 7768.
Fuwa, H., Noguchi, T., Kawakami, M., and Sasaki, M., Bioorg. Med. Chem. Lett., 2014, vol. 24, p. 2415.
Sethofer, S.G., Staben, S.T., Hung, O.Y., and Toste, F.D., Org. Lett., 2008, vol. 10, p. 4315.
Keaton, K.A. and Phillips, A.J., Org. Lett., 2008, vol. 10, p. 1083.
Mineeva, I.V., Russ. J. Org. Chem., 2008, vol. 44, p. 1261.
Gollner, A. and Mülzer, J., Org. Lett., 2008, vol. 10, p. 4701.
Haym, I. and Brimble, M.A., Synlett., 2009, p. 2315.
Kim, K. and Cha, J.K., Angew. Chem., Int. Ed., 2009, vol. 48, p. 5334.
Hurski, A.L., Sokolov, N.A., and Kulinkovich, O.G., Tetrahedron, 2009, vol. 65, p. 3518.
Odinokov, V.N., Shakurova, E.R., Halilov, L.M., and Dzhemilev, U.M., Russ. J. Org. Chem., 2009, vol. 45, p. 1464.
Kovalenko, V.N., Masalov, N.V., and Kulinkovich, O.G., Russ. J. Org. Chem., 2009, vol. 45, p. 1318.
Gaucher, X., Jida, M., and Ollivier, J., Synlett., 2009, p. 3320.
Mineeva, I.V. and Kulinkovich, O.G., Russ. J. Org. Chem., 2009, vol. 45, p. 1623.
Gollner, A., Altmann, K.-H., Gertsch, J., and Mülzer, J., Chem. Eur. J., 2009, vol. 15, p. 5979.
Mineeva, I.V., Candidate Sci. (Chem.) Dissertation, Minsk, 2010.
Kovalenko, V.N., Sokolov, N.A., and Kulinkovich, O.G., Russ. J. Org. Chem., 2010, vol. 46, p. 1702.
Hurski, A.L. and Kulinkovich, O.G., Tetrahedron Lett., 2010, vol. 51, p. 3497.
Mineyeva, I.V. and Kulinkovich, O.G., Tetrahedron Lett., 2010, vol. 51, p. 1836.
Kananovich, D.G., Zubrytski, D.M., and Kulinkovich, O.G., Synlett., 2010, p. 1043.
Hurski, A.L. and Kulinkovich, O.G., Russ. J. Org. Chem., 2011, vol. 47, p. 1653.
Ye, Z., Gao, T., and Zhao, G., Tetrahedron, 2011, vol. 67, p. 5979.
Shklyaruck, D. and Matiushenkov, E., Tetrahedron: Asymm., 2011, vol. 22, p. 1448.
Astashko, D.A. and Tyvorskii, V.I., Tetrahedron Lett., 2011, vol. 52, p. 4792.
Haym, I. and Brimble, M.A., Org. Biomol. Chem., 2012, vol. 10, p. 7649.
Mineeva, I.V., Russ. J. Org. Chem., 2012, vol. 48, p. 977.
McCabe, J. and Phillips, A.J., Tetrahedron, 2013, vol. 69, p. 5710.
Mineeva, I.V., Russ. J. Org. Chem., 2013, vol. 49, p. 253.
Mineeva, I.V., Russ. J. Org. Chem., 2013, vol. 49, p. 712.
Mineeva, I.V., Russ. J. Org. Chem., 2013, vol. 49, p. 838.
Mineeva, I.V., Russ. J. Org. Chem., 2013, vol. 49, p. 979.
Mineeva, I.V., Russ. J. Org. Chem., 2013, vol. 49, p. 1647.
Mineeva, I.V., Russ. J. Org. Chem., 2014, vol. 50, p. 100.
Mineeva, I.V., Russ. J. Org. Chem., 2014, vol. 50, p. 168.
Mineeva, I.V., Russ. J. Org. Chem., 2014, vol. 50, p. 398.
Kovalenko, V.N. and Mineeva, I.V., Russ. J. Org. Chem., 2014, vol. 50, p. 934.
Mineeva, I.V., Russ. J. Org. Chem., 2014, vol. 50, p. 1558
Mineeva, I.V., Masyuk, V.S., Kovalenko, V.N., and Bondarenko, M.M., Russ. J. Org. Chem., 2014, vol. 50, p. 1621.
Shklyaruck, D., Tetrahedron: Asymm., 2014, vol. 25, p. 644.
Han, W.-B., Li, S.-G., Lu, X.-W., and Wu, Y., Eur. J. Org. Chem., 2014, vol. 30, p. 3841.
Zubrytski, D.M., Kananovich, D.G., and Kulinkovich, O.G., Tetrahedron, 2014, vol. 70, p. 2944.
Gibson, D.H. and De Puy, Ch., Chem. Rev., 1974, vol. 74, p. 605.
Kulinkovich, O.G. and de Meijere, A., Chem. Rev., 2000, vol. 100, p. 2789.
Kulinkovich, O.G., Chem. Rev., 2003, vol. 103, p. 2597.
Kulinkovich, O.G., Russ. Chem. Bull., 2004, vol. 53, p. 1065.
Wolan, A. and Six, Y., Tetrahedron, 2010, vol. 66, p. 15.
Keck, G.E., Tarbet, Kh., and Geraci, L.S., J. Am. Chem. Soc., 1993, vol. 115, p. 8467.
Keck, G.E. and Geraci, L.S., Tetrahedron Lett., 1993, vol. 34, p. 7827.
Costa, A.L., Piazza, M.G., Tagliavini, E., Trombini, C., and Umani-Ronchi, A., J. Am. Chem. Soc., 1993, vol. 115, p. 7001.
Fallner, J.W., Sams, D.W.I., and Liu, X., J. Am. Chem. Soc., 1996, vol. 118, p. 1217.
Keck, G.E. and Krishnamurthy, D., Org. Synth., 1998, vol. 75, p. 12.
Williams, D.R., Brooks, D.A., Meyer, K.G., and Clark, M.P., Tetrahedron Lett., 1998, vol. 39, p. 7251.
Keck, G.E. and Yu, T., Org. Lett., 1999, vol. 1, p. 289.
Doucet, H. and Santelli, M., Tetrahedron: Asymm., 2000, vol. 11, p. 4163.
Sanchez, C.C. and Keck, G.E., Org. Lett., 2005, vol. 7, p. 3053.
Von Gyldenfeldt, F., Marton, D., and Tagliavini, G., Organometallics, 1994, vol. 13, p. 906.
Carofiglio, T., Marton, D., and Tagliavini, G., Organometallics, 1992, vol. 11, p. 2961.
Dale, J.A., Dull, D.L., and Mosher, H.S., J. Org. Chem., 1969, vol. 34, p. 2543.
Berliner, M.A. and Belecki, K., J. Org. Chem., 2005, vol. 70, p. 9618.
Yadav, J.S. and Reddy, P.S.R., Synthesis, 2007, p. 1070.
Kurosu, M. and Lorca, M., Tetrahedron Lett., 2002, vol. 43, p. 1765.
Kurosu, M. and Lorca, M., Synlett., 2005, p. 1109.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.V. Mineeva, 2015, published in Zhurnal Organicheskoi Khimii, 2015, Vol. 51, No. 8, pp. 1083–1092.
Rights and permissions
About this article
Cite this article
Mineeva, I.V. New approach to the synthesis of macrocyclic core of cytotoxic lactone (+)-neopeltolide. Synthesis of C7–C14 segment basing on cyclopropanol intermediates. Russ J Org Chem 51, 1061–1070 (2015). https://doi.org/10.1134/S1070428015080023
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428015080023