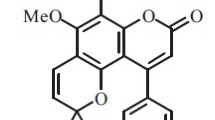
Abstract
The reaction of a linearly fused furocoumarin, oreoselone, with p-toluenesulfonyl chloride gave 2-tosyloreoselone which showed a high reactivity in palladium-catalyzed cross-coupling reactions with formation of a new carbon-carbon bond. 2-Tosyloreoselone reacted with terminal alkynes in the presence of Pd(PPh3)2Cl2 to give the corresponding 2-alkynylfuro[3,2-g]chromen-3-ones. 2-Aryl(hetaryl)alkynyloreoselones were obtained in high yield directly by palladium-catalyzed reaction of oreoselone with tosyl chloride and aryl(hetaryl)acetylenes. The reaction of 2-tosyloreoselone with aryl(hetaryl)boronic acids in the presence of palladium complexes with uni- and bidentate ligands, tetrabutylammonium bromide, and a base afforded 2-aryl(hetaryl)-substituted oreoselones. 2-Vinyloreoselone was synthesized from 2-tosyloreoselone and potassium trifluoro(vinyl)borate.
Similar content being viewed by others
References
Makhneva, E.A., Lipeeva, A.V., Shul’ts, E.E., Shakirov, M.M., and Tolstikov, G.A., Russ. J. Org. Chem., 2012, vol. 48, p. 1094.
Lipeeva, A.V., Shul’ts, E.E., Bagryanskaya, I.Yu., Shakirov, M.M., and Tolstikov, G.A., Russ. J. Org. Chem., 2011, vol. 47, p. 1083.
Lipeeva, A.V., Shul’ts, E.E., Shakirov, M.M., and Tolstikov, G.A., Russ. J. Org. Chem., 2011, vol. 47, p. 1390.
Lipeeva, A.V., Shul’ts, E.E., Shakirov, M.M., and Tolstikov, G.A., Russ. J. Org. Chem., 2011, vol. 47, p. 1404.
Bruchhausen, F. and Hoffman, H., Chem. Ber., 1941, vol. 74, p. 1584.
Osadchii, S.A., Shul’ts, E.E., Shakirov, M.M., and Tolstikov, G.A., Izv. Akad. Nauk, Ser. Khim., 2006, p. 362.
Lipeeva, A.V., Shul’ts, E.E., Shakirov, M.M., and Tolstikov, G.A., Russ. J. Org. Chem., 2010, vol. 46, p. 1858.
Luo, Y. and Wu, J., Tetrahedron, 2009, vol. 65, p. 6810.
Saito, S. and Yamamoto, Y., Chem. Rev., 2000, vol. 100, p. 2901.
Mandai, T., Matsumoto, T., and Tsuji, J., Synlett, 1993, no. 2, p. 113.
Jin, Z. and Fuchs, P.L., Tetrahedron Lett., 1996, vol. 37, p. 5253.
Li, M.-B., Tang, X.L., and Tian, S.-K., Adv. Synth. Catal., 2011, vol. 353, p. 1980.
Ackermann, L. and Mulzer, M., Org. Lett., 2008, vol. 10, p. 5043; Kang, F.-A., Sui, Z., and Murray, W.V., J. Am. Chem. Soc., 2008, vol. 130, p. 11 300; Luo, Y. and Wu, J., Tetrahedron Lett., 2009, vol. 50, p. 2103.
Molander, G.A. and Rivero, M.R., Org. Lett., 2002, vol. 4, p. 107.
Darses, S., Michaud, G., and Genêt, J.-P., Eur. J. Org. Chem., 1999, no. 8, p. 1875.
Shul’ts, E.E., Petrova, T.N., Shakirov, M.M., Chernyak, E.I., Pokrovskii, L.M., Nekhoroshev, S.A., and Tolstikov, G.A., Khim. Inter. Ustoich. Razv., 2003, vol. 11, p. 683.
Dzhemilev, U.M., Popod’ko, N.R., and Kozlova, E.V., Metallokompleksnyi kataliz v organicheskom sinteze (Metal Complex Catalysis in Organic Synthesis), Moscow: Khimiya, 1999, p. 10
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.B. Lipeeva, E.E. Shul’ts, M.M. Shakirov, G.A. Tolstikov, 2013, published in Zhurnal Organicheskoi Khimii, 2013, Vol. 49, No. 1, pp. 105–112.
For communication X, see [1].
Rights and permissions
About this article
Cite this article
Lipeeva, A.B., Shul’ts, E.E., Shakirov, M.M. et al. Plant coumarins: XI. Cross coupling reactions with 2-(tosyl)oreoselone. Russ J Org Chem 49, 99–107 (2013). https://doi.org/10.1134/S107042801301017X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107042801301017X