Abstract
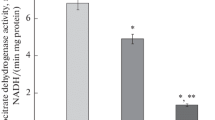
Repeated severe insulin-induced hypoglycemia in rats has led to an increase in aminotransferase, glutaminase, and glutamate dehydrogenase activities in the liver; protease activities in tissues; and in blood serum levels of free fatty acids, urea, and uric acid. These changes are indicative of gluconeogenesis activation in animals exposed to hyperinsulinization. Decreased rates of glycolysis and glycogenolysis, reduced activities of NADP-dependent dehydrogenases, and substantial changes in the activities of enzymes responsible for metabolism of nucleotides and transmitter amino acids have been observed in the brain. All these changes are mainly associated with hypoglycemia and activation of the contrainsular system and can play a significant role in pathogenesis of posthypoglycemic encephalopathy.
Similar content being viewed by others
References
Adkins, A., Basu, R., Persson, M., et al., Higher Insulin Concentrations are Required to Suppress Gluconeogenesis than Glycogenolysis in Nondiabetic Humans, Diabetes, 2003, vol. 52, pp. 2213–2220.
Ames, A. III, CNS Energy Metabolism as Related to Function, Brain Res. Rev., 2000, vol. 34, pp. 42–68.
Amiel, S.A., Studies in Hypoglycaemia in Children with Insulin-Dependent Diabetes Mellitus, Horm. Res., 1996, vol. 45, pp. 285–290.
Auer, R.N., Hypoglycemic Brain Damage, Stroke, 1986, vol. 17, pp. 699–708.
Balabolkin, M.I., Diabetologiya (Diabetology), Moscow: Meditsina, 2000.
Bolli, G.B. and Fanelli, C.G., Physiology of Glucose Counterregulation to Hypoglycemia, Endocrinol. Metab. Clin., 1999, vol. 28, pp. 467–493.
Brown, A.M., Brain Glycogen Re-Awakened, J. Neurochem., 2004, vol. 89, pp. 537–552.
Charlton, M. and Nair, K.S., Protein Metabolism in Insulin-Dependent Diabetes Mellitus, J. Nutr., 1998, vol. 128, pp. 323–327.
Cryer, P.E., Davis, S.N., and Shamoon, H., Hypoglycemia in Diabetes, Diabetes Care, 2003, vol. 26, pp. 1902–1912.
Edgerton, D.S., Cardin, S., Pan, C., et al., Effects of Insulin Deficiency or Excess on Hepatic Gluconeogenic Flux during Glycogenolytic Inhibition in the Conscious Dog, Diabetes, 2002, vol. 51, pp. 3151–3162.
Erecinska, M., Nelson, D., and Silver, I.A., Metabolic and Energetic Properties of Isolated Nerve Ending Particles (Synaptosomes), Biochim. Biophys. Acta, 1996, vol. 1277, pp. 13–34.
Ferre, P., Foretz, M., Azzout-Marniche, D., et al., Sterol-Regulatory-Element-Binding Protein 1c Mediates Insulin Action on Hepatic Gene Expression, Biochem. Soc. Trans., 2001, vol. 29, pp. 547–552.
Gastaldelli, A., Toschi, E., Pettiti, M., et al., Effect of Physiological Hyperinsulinemia on Gluconeogenesis in Nondiabetic Subjects and in Type 2 Diabetic Patients, Diabetes, 2001, vol. 50, pp. 1807–1812.
Gerlach, M., Ben-Shachar, D., Riederer, P., and Youdim, M.B.H., Altered Brain Metabolism of Iron as a Cause of Neurodegenerative Diseases?, J. Neurochem., 1994, vol. 63, pp. 793–807.
Greene, A.E., Todorova, M.T., and Seyfried, T.N., Perspectives on the Metabolic Management of Epilepsy through Dietary Reduction of Glucose and Elevation of Ketone Bodies, J. Neurochem., 2003, vol. 86, pp. 529–537.
Halliwel, B., Reactive Oxygen Species and the Central Nervous System, J. Neurochem., 1992, vol. 59, pp. 1609–1623.
Honegger, P., Braissant, O., Henry, H., et al., Alteration of Amino Acid Metabolism in Neuronal Aggregate Cultures Exposed to Hypoglycaemic Conditions, J. Neurochem., 2002, vol. 81, pp. 1141–1151.
Horber, F.F. and Haymond, M.W., Human Growth Hormone Prevents the Protein Catabolic Side Effects of Prednisone in Humans, J. Clin. Invest., 1990, vol. 86, pp. 265–272.
Inouye, K., Shum, K., Chan, O., et al., Effects of Recurrent Hyperinsulinemia with and Without Hypoglycemia on Counterregulation in Diabetic Rats, Am. J. Physiol. Endocrinol. Metab., 2002, vol. 282, pp. 1369–1379.
Lipton, P. and Robacker, K., Glycolysis and Brain Function: [K+] Stimulation of Protein Synthesis and K+ Uptake Require Glycolysis, Fed. Proc., 1983, vol. 42, pp. 2875–2880.
Lipton, P., Glycolysis Is Necessary for Normal Synaptic Transmission in Guinea-Pig Hippocampal Slices, Soc. Neurosci. Symp. Abstr., 1991, vol. 17, p. 1155.
Magen, A., Koren-Schwartzer, N., Chen-Zion, M., and Beitner, R., Effect of Insulin-Induced Hypoglycemia on Cytoskeleton-Bound and Cytosolic Phosphofructokinase and the Levels of Glucose 1,6-Bisphosphate in Rat Brain, Biochem. Mol. Med., 1995, vol. 56, pp. 94–98.
Magistretti, P.J. and Pellerin, L., Cellular Mechanisms of Brain Energy Metabolism: Relevance to Functional Brain Imaging and to Neurodegenerative Disorders, Ann. N.Y. Acad. Sci., 1996, vol. 777, pp. 380–387.
Marks, V. and Teale, J.D., Hypoglycemia: Factitious and Felonious, Endocrinol. Metab. Clin., 1999, vol. 28, pp. 579–601.
Men’shikov, V.V., Laboratornye metody issledovaniya v klinike (Laboratory Research Methods in Clinical Medicine), Moscow: Meditsina, 1987.
Murti, V., Prakash, G.S., and Subramanyan, K., Activities of Acidic and Neutral Proteases in Different Brain Departments of Rats: Distribution in Glia and Neurons, Neirokhimiya, 1985, no. 1, pp. 52–55.
Nissim, I., Brosnan, M., Yudkoff, M., et al., Studies of Hepatic Glutamine Metabolism in the Perfused Rat Liver with 15N-Labeled Glutamine, J. Biol. Chem., 1999, vol. 274, pp. 28958–28965.
O’Brien, R.M., Streeper, R.S., Ayala, J.E., et al., Insulin-Regulated Gene Expression, Biochem. Soc. Trans., 2001, vol. 29, pp. 552–558.
Panin, L.E., Tret’yakova, T.A., Russkikh, G.S., and Voitsekhovskaya, E.E., Features of Regulation of Key Glycolytic Enzymes and the Pentose Phosphate Pathway in Differently Specialized Tissues, Vopr. Med. Khim., 1982, no. 2, pp. 26–30.
Pellerin, L. and Magistretti, P.J., Glutamate Uptake into Astrocytes Stimulates Aerobic Glycolysis: A Mechanism Coupling Neuronal Activity to Glucose Utilization, Proc. Natl. Acad. Sci. USA, 1994, vol. 91, pp. 10625–10629.
Prokhorova, M.I., Metody biokhimicheskikh issledovanii: Lipidnyi i energeticheskii obmen (Methods of Biochemical Research: Lipid and Energy Metabolism), Leningrad: Leningr. Gos. Univ., 1982.
Ros, J., Pecinska, N., Alessandri, B., et al., Lactate Reduces Glutamate-Induced Neurotoxicity in Rat Cortex, J. Neurosci. Res., 2001, vol. 66, pp. 790–794.
Sapolsky, R.M., Cellular Defenses against Excitotoxic Insults, J. Neurochem., 2001, vol. 76, pp. 1601–1611.
Staehr, P., Hother-Nielsen, O., Landau, B.R., et al., Effects of Free Fatty Acids per se on Glucose Production, Gluconeogenesis, and Glycogenolysis, Diabetes, 2003, vol. 52, pp. 260–267.
Telushkin, P.K., Intensity of Lipid Peroxidation, Activity of NADP-Dependent Dehydrogenases and Proteases in the Brain of Rats Repeatedly Injected with Insulin, Probl. Endokrinol., 1998, no. 3, pp. 35–38.
Telushkin, P.K. and Nozdrachev, A.D., Hypoglycemia and Brain: Metabolism and Mechanisms of Neuron Damage, Usp. Fiziol. Nauk, 1999, no. 4, pp. 14–27.
Telushkin, P.K., Nozdrachev, A.D., and Potapov, P.P., Activities of Deamination Enzymes in the Brain of Rats during Recovery from Insulin-Induced Hypoglycemia, Probl. Endokrinol., 2001, no. 5, pp. 43–45.
Timothy, G.R., Obesity. Fat Cells, Endocrinol. Metab. Clin., 1996, vol. 25, pp. 847–867.
Todorov, I., Klinicheskie laboratornye issledovaniya v pediatrii (Clinical Laboratory Investigations in Pediatrics), Sofia: Meditsina i Fizkul’tura, 1961.
Tombaugh, G.C. and Sapolsky, R.M., Evolving Concepts about Role of Acidosis in Ischemic Neuropathology, J. Neurochem., 1993, vol. 61, pp. 793–803.
Watford, M., Hepatic Glutaminase Expression: Relationship to Kidney-Type Glutaminase and to the Urea Cycle, FASEB J., 1993, vol. 7, pp. 1468–1474.
Zeevalk, G.D. and Nicklas, W.J., Lactate Prevents the Alterations in Tissue Amino Acids, Decline in ATP, and Cell Damage due to Aglycemia in Retina, J. Neurochem., 2000, vol. 75, pp. 1027–1034.
Author information
Authors and Affiliations
Additional information
Original Russian Text © P.K. Telushkin, A.D. Nozdrachev, P.P. Potapov, 2008, published in Izvestiya Akademii Nauk, Seriya Biologicheskaya, 2008, No. 3, pp. 324–332.
Rights and permissions
About this article
Cite this article
Telushkin, P.K., Nozdrachev, A.D. & Potapov, P.P. Parameters of energy and nitrogen metabolism in rats under insulin-induced hypoglycemia. Biol Bull Russ Acad Sci 35, 279–286 (2008). https://doi.org/10.1134/S1062359008030084
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359008030084