Abstract
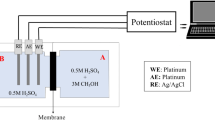
Membranes Nafion 117 are modified with poly(3,4-ethylenedioxythiophene) (PEDT) by chemical polymerization of EDT with H2O2 or FeCl3 as the oxidants in a two-compartment cell. Depending on the oxidant and polymerization conditions, PEDT is deposited either as a thin film on the membrane surface or inside the Nafion membrane depending on whether FeCl3 or H2O2 is used as the oxidant. The decrease in the ionic conductivity and methanol permeability is studied as a function of the polymerization time. A linear dependence is found with H2O2 and a t −1/2 dependence, with FeCl3. The contributions of PEDT and Nafion to the overall conductivity of the composite membranes are separated by impedance measurements. The modified membranes (FeCl3) are also tested in direct methanol fuel cells (DMFC). The methanol permeation through the membranes is measured by operating the fuel cell in an electrolysis mode. The influence of the modified membranes on the DMFC current-voltage characteristics is studied with 2 M CH3OH and O2 at 1.2 barabs and 80°C. Membrane electrode assemblies (MEAs) are prepared by hot pressing the modified membrane between two commercial gas diffusion electrodes with 1 mg cm−2 of Pt loading. A decrease of the methanol permeation of 25% is observed at MEA with the modified membrane (1 h polymerization time), compared with that of MEA with a Nafion membrane. However, the overall DMFC performance decreases in the same relation: a maximal power density of 36 W cm−2 is measured at MEA with a PEDT-modified membrane compared with 45 W cm−2 for MEA with a Nafion membrane.
Similar content being viewed by others
References
Heinzel, A. and Barragan, V.M., J. Power Sources, 1999, vol. 84, p. 70.
Ma, Z.Q., Cheng, P., and Zhao, T.S., J. Membr. Sci., 2003, vol. 215, p. 327.
Choi, W.C., Kim, J.D., and Woo, S.I., J. Power Sources, 2001, vol. 96, p. 411.
Deluga, C. and Pivovar, B.S., Proc. 3rd Int. Symp. “New Materials for Electrochemical Systems,” Montreal, 1999, p. 132.
Feichtinger, J., Kerres, J., Schulze, A., Walker, M., and Schumacher, U., J. New Mater. Electrochem. Syst., 2002, vol. 5, p. 162.
Liu, J., Wang, H.T., Cheng, S.A., and Chan, K.-Y., J. Membr. Sci., 2005, vol. 246, p. 95.
Shao, Z.G., Wang, X., and Hsing, I.M., J. Membr. Sci., 2002, vol. 210, p. 147.
Banaszak, R.A., Arbaugh, S.A., Steffee, E.D., and Pyati, R., J. Electrochem. Soc., 2004, vol. 151, p. 1020.
Dimitrova, P., Friedrich, K.A., Stimming, U., and Vogt, B., Solid State Ionics, 2002, vol. 150, p. 115.
Tricoli, V., Carretta, N., and Bartolozzi, M., J. Electrochem. Soc., 2000, vol. 147, p. 1286.
Savadogo, O., J. Power Sources, 2004, vol. 127, p. 135.
Jüttner, K., Mangold, K.-M., Lange, M., and Bouzek, K., Russ. J. Electrochem., 2004, vol. 40, p. 317.
Moravcová, S., Cilová, Z., and Bouzek, K., J. Appl. Electrochem., 2005, vol. 35, p. 991.
Moravcová, S. and Bouzek, K., J. Electrochem. Soc., 2005, vol. 152, p. A2080.
Langsdorf, B.L., Sultan, J., and Pickup, P.G., J. Phys. Chem. B, 2003, vol. 107, p. 8412.
Easton, E.B., Langsdorf, B.L., Hughes, J.A., Sultan, J., Qi, Z.G., Kaufman, A., and Pickup, P.G., J. Electrochem. Soc., 2003, vol. 150, p. 735.
Zhu, J., Sattler, R.R., Garsuch, A., Yepez, O., and Pickup, P.G., Electrochim. Acta (in press).
Li, L., Drillet, J.-F., Dittmeyer, R., and Jüttner, K., J. Solid State Electrochem. (in press).
Ehrenbeck, C. and Jüttner, K., Electrochim. Acta, 1996, vol. 41, p. 1851.
Schmitz, R.H.J. and Jüttner, K., Electrochim. Acta, 1999, vol. 44, p. 1627.
Tricoli, V., J. Electrochem. Soc., 1998, vol. 145, p. 3798.
Tan, S., Tieu, J.H., and Belanger, D., J. Phys. Chem. B, 2005, vol. 109, p. 14085.
Vorotyntsev, M.A., Daikhin, L.I., and Levi, M.D., J. Electroanal. Chem., 1994, vol. 364, p. 37.
Deslouis, C., Musiani, M.M., Tribollet, B., and Vorotyntsev, M.A., J. Electrochem. Soc., 1995, vol. 142, p. 1902.
Mathias, M.F. and Haas, O., J. Phys. Chem., 1992, vol. 96, p. 3174.
Albery, W.J., Elliott, C.M., and Mount, R., J. Electroanal. Chem., 1990, vol. 288, p. 15.
Ren, X. and Pickup, P.G., J. Electrochem. Soc., 1992, vol. 139, p. 2097.
Ehrenbeck, C., Jüttner, K., Ludwig, S., and Paasch, G., Electrochim. Acta, 1998, vol. 43, p. 2781.
Jüttner, K., Schmitz, R.H.J., and Hudson, A., Electrochim. Acta, 1999, vol. 44, p. 4177.
de Levie, R., Advances in Electrochemistry and Electrochemical Engineering, Delahay, P., Ed., New York, Intersicence, 1967, vol. 6. p. 329.
Ren, X., Springer, T.E., Zawodzinski, T.A., and Gottesfeld, S., J. Electrochem. Soc., 2000, vol. 147, p. 466.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Published in Russian in Elektrokhimiya, 2006, Vol. 42, No. 11, pp. 1330–1339.
Based on the report delivered at the 8th International Frumkin Symposium “Kinetics of the Electrode Processes,” October 18–22, 2005, Moscow.
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Li, L., Drillet, J.F., Mácová, Z. et al. Poly(3,4-ethylenedioxythiophene)-modified nafion membrane for direct methanol fuel cells. Russ J Electrochem 42, 1193–1201 (2006). https://doi.org/10.1134/S102319350611005X
Received:
Issue Date:
DOI: https://doi.org/10.1134/S102319350611005X