Abstract
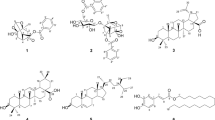
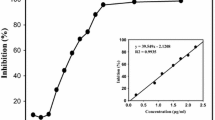
The search for the effective and safe α-glucosidase and α-amylase inhibitors from Actinomycetaceae being antidiabetic agents is actual problem. Twenty one Streptomyces spp. of soil samples collected from different places of China were screened for the ability to produce this kind of inhibitory activities. Fermentation broth of isolated strains had absorbance between 350–190 nm. The Streptomyces strains PW003, ZG636, and ZG731 were characterized by special absorption at 280, 275, and 400 nm, respectively. Ten of the collected actinomycete strains had the ability to inhibit α-glucosidase or/and α-amylase and the fermentation broth of the same strain had inhibitory activity varied greatly depending on the enzyme source. In the process to screen the leading compounds used as antidiabetic agents, human α-glucosidase and α-amylase were revealed as the best used in trail compared with the same enzymes from other sources. Active α-glucosidase inhibitor was isolated from Streptomyces strain PW638 fermentation broth and identified as acarviostatin I03 by MS and NMR spectrometry. Its IC50 value was 1.25 and 12.23 μg/ml against human intestinal N-terminal maltase-glucoamylase and human pancreatic α-amylase, respectively.
Similar content being viewed by others
References
Coniff, R. and Kron, A., Clin. Ther., 1997, vol. 19, no. 1, pp.16–26.
Eduardo, B.M., Adriane, S.G., and Ivone, C., Tetrahedron, 2006, vol. 62, no. 44, pp. 10277–10302.
Taifo, M., Nat. Prod. Rep., 2003, vol. 20, no. 1, pp. 137–166.
Waksman, S.A. and Henrici AT., J. Bacteriol., 1943, vol. 46, no.4, pp. 337–341.
Kim, S.B., Lonsdale, J., and Seong, C.N., Antonie van Leeuwenhoek, 2003, vol. 83, no. 4, pp. 107–116.
Okami, Y. and Hotta, K., in Actinomycetes in Biotechnology, Goodfellow, M., Williams, S.T., and Mordarski, M., Eds., London: Academic Press, 1988, pp. 33–67.
Myers, A.M., Morel, M.K., James, M.G., and Ball, S.G., Plant Physiol., 2000, vol. 122, pp. 989–997.
Flanagan, P.R. and Forstner, G.G., Biochem. J., 1978, vol. 173, no. 2, pp. 553–563.
Auricchio, S., Semenza, G., and Rubino, A., Biochim. Biophys. Acta, 1965, vol. 96, pp. 498–507.
Huang, Y.N., Zhao, Y.L., Gao, X.L., Zhao, Z.F., Jing, Z., Zeng, W.C., and Yang, R., J. Ethnopharmacol., 2010, vol. 58, pp. 135–144.
Hillebrand, I., Boehme, K., Frank, G., Fink, H., and Berchtold, P., Res. Exp. Med. (Berlin), 1979, vol. 175, no. 1, pp. 81–86.
Matsumoto, K., Yano, M., Miyake, S., Ueki, Y., Yamaguchi, Y., Akazawa, S., and Tominaga, Y., Diabetes Care, 1998, vol. 21, no. 2, pp. 256–260.
Gurusiddaiah, S. and Graham, S.O., Antimicrob. Agents Chemother., 1980, vol. 17, no. 6, pp. 980–987.
Werner, G., Hagenmaier, H., Drautz, H., Baumgartner, A., and Zahner, H., J. Antibiot. (Tokyo), 1984, vol. 37, pp. 110–117.
Henkel, T., Ciesiolka, T., Rohr, J., and Zeeck, A., J. Antibiot. (Tokyo), 1989, vol. 42, pp. 299–311.
Kunihiro, S. and Kaneda, M., J. Antibiot. (Tokyo), 2003, vol. 56, pp. 3330–3333.
Kuster, E. and Williams, S., Nature, 1964, vol. 202, pp.928–932.
Wipat, A., Wellington, E., and Saunders, V., Appl. Environ. Microbiol., 1991, vol. 57, no. 11, pp. 23–30.
Lechevalier, M.P. and Lechevalier, H.A., Int. J. Syst. Bacteriol., 1970, vol. 20, pp. 35–43.
Taddei, A., Rodriquez, M., Marquezvilchez, E., and Castelli, C., Microbiol. Res., 2006, vol. 161, no. 3, pp. 222–231.
Bieble, H. and Sproer, C., Syst. Appl. Microbiol., 2002, vol. 25, no. 4, pp. 491–497.
Geng, P., Qiu, F., Zhu, Y.Y., and Bai, G., Carbohydr. Res., 2008, vol. 343, no. 5, pp. 882–892.
Shirling, E.B. and Gottlieb, D., Int. J. Syst. Bacteriol., 1966, vol. 16, no. 3, pp.313–340.
Myles, D.C., Curr. Opin. Biotech., 2003, vol. 14, no. 6, pp. 627–633.
Honda, S., Akao, E., Suzuki, S., Okuda, M., and Kakehi, K., J. Anal. Biochem., 1989, vol. 180, no. 2, pp. 351–357.
Lee, D.S., J. Biosci. Bioeng., 2000, vol. 89, no. 2, pp.271–273.
Geng, P. and Bai, G., Carbohydr. Res., 2008, vol. 343, no. 3, pp. 470–476.
Mahmud, T., Nat. Prod. Rep., 2003, vol. 20, no. 1, pp. 137–166.
Davies, J.E., J. Antibiot. (Tokyo), 2007, vol. 38, no. 8, pp.529–532.
Yoichi, N., Takashi, M., Shigeki, M., Rob, W.M., Soestb, V., and Nobuhiro, F., Tetrahedron, 2000, vol.56, no.46, 8977–8987.
Choudhary, D., Jansson, I., Sarfarazi, M., and Schenkman, J.B., Pharmacogenet. Genomics, 2008, vol. 18, no. 8, pp. 665–676.
Kim, K.Y., Nama, K.A.., Kurihara, H.B., and Kim, S.M., Phytochemistry, 2008, vol. 69, no. 16, pp. 2820–2825.
Lee, S.S., Lin, H.C., and Chen, C.K., Phytochemistry, 2008, vol. 69, no. 12, pp.2347–2353.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Meng, P., Xie, C., Geng, P. et al. Inhibitory effect of components from Streptomyces species on α-glucosidase and α-amilase of different origin. Appl Biochem Microbiol 49, 160–168 (2013). https://doi.org/10.1134/S0003683813020099
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0003683813020099