Abstract
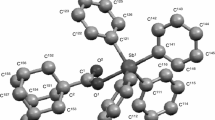
The heat capacity of triphenylantimony diacrylate Ph3Sb(O2CCH=CH2)2 was studied in an adiabatic vacuum calorimeter at 6–350 K and differential scanning calorimeter at 330–450 K. Melting was revealed at these temperatures; the melting point was estimated at 428.4 ± 0.5 K. It was accompanied by the partial decomposition of the substance. The low-temperature (20 K ≤ T ≤ 50 K) heat capacity was treated using the Debye theory of the heat capacity of solids and its multifractal model. The type of the structure topology was determined. The standard thermodynamic functions C o p (T), H o(T) − H o(0), S o(T), and G o(T) − H o(0) of the compound in the crystal state were calculated from the obtained experimental data in the range from T → 0 to 428 K. The standard entropy of the formation of the crystalline compound Ph3Sb(O2CCH=CH2)2 at T = 298.15 K was determined.
Similar content being viewed by others
References
V. V. Sharutin, O. K. Sharutina, O. V. Molokova, et al., Russ. J. Coord. Chem. 28, 544 (2002).
A. P. Pakusina, V. V. Sharutin, and O. K. Sharutina, Synthesis and Some Reactions of the Organoantimony Compounds (Dal’nevost. Gos. AUniv., Blagoveshchensk, 2005) [in Russian].
A. V. Gushchin, D. V. Shashkin, L. K. Prytkova, et al., Russ. J. Gen. Chem. 81, 493 (2011).
I. B. Rabinovich, V. P. Nistratov, V. I. Tel’noi, et al., Thermodynamics of Organometallic Compounds (Nizhegor. Gos. Univ., Nizh. Novgorod, 1996) [in Russian].
N. N. Smirnova, I. A. Letyanina, V. N. Larina, et al., J. Chem. Thermodyn. 41, 46 (2009).
N. N. Smirnova, I. A. Letyanina, A. V. Markin, et al., Russ. J. Gen. Chem. 79, 717 (2009).
I. A. Letyanina, N. N. Smirnova, A. V. Markin, et al., J. Therm. Anal. Cal. 103, 355 (2011).
A. V. Markin, I. A. Letyanina, N. N. Smirnova, et al., Russ. J. Phys. Chem. A 85, 1315 (2011).
A. V. Markin, I. A. Letyanina, V. A. Ruchenin, et al., J. Chem. Eng. Data 56, 3657 (2011).
C. E. Carraer, Jr., J. Sheats, and Ch. Pittman, Jr., Metal-Containing Polymeric Materials (Plenum, New York, 1996; Mir, Moscow, 1981).
V. A. Dodonov, A. V. Gushchin, Yu. L. Kuznetsova, et al., Vestn. NNovg. Univ., No. 1, 82 (2004).
J. R. Leebrick, USA Patent No. 3287210 (United States Patent Office, 1966).
A. V. Gushchin, L. K. Prytkova, D. V. Shashkin, et al., Vestn. NNovg. Univ., No. 3, 95 (2010).
R. M. Varushchenko, A. I. Druzhinina, and E. L. Sorkin, J. Chem. Thermodyn. 29, 623 (1997).
V. M. Malyshev, G. A. Mil’ner, E. L. Sorkin, et al., Prib. Tekh. Eksp., No. 6, 195 (1985).
G. W. H. Hohne, W. F. Hemminger, and H. F. Flammersheim, Differential Scanning Calorimetry (Springer, Berlin, Heidelberg, 2003).
V. A. Drebushchak, J. Therm. Anal. Calorim. 79, 213 (2005).
M. B. Weiser, Pure Appl. Chem. 78, 2051 (2006).
T. S. Yakubov, Dokl. Akad. Nauk SSSR 310, 145 (1990).
V. B. Lazarev, A. D. Izotov, K. S. Gavrichev, et al., Thermochim. Acta 269–270, 109 (1995).
V. V. Tarasov, Zh. Fiz. Khim. 24, 111 (1950).
V. V. Tarasov and G. A. Yunitskii, Zh. Fiz. Khim. 39, 2077 (1965).
B. V. Lebedev, Thermochim. Acta 297, 143 (1997).
J. P. McCullough and D. W. Scott, Calorimetry of Non-Reacting Systems (Butterworth, London, 1968).
Codata Key Values for Thermodynamics, Ed. by D. D. Cox, V. A. Wagman, and V. A. Medvedev (New York, 1984).
M. W. Chase, Jr., J. Phys. Chem. Ref. Data, Monograph 9, 1951 (1998).
Thermodynamic Constants of Substances, Ed. by V. P. Glushko (VINITI, Moscow, 1965–1981), Nos. 1–10 [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.A. Letyanina, A.V. Markin, N.N. Smirnova, A.V. Gushchin, D.V. Shashkin, 2012, published in Zhurnal Fizicheskoi Khimii, 2012, Vol. 86, No. 8, pp. 1329–1335.
Rights and permissions
About this article
Cite this article
Letyanina, I.A., Markin, A.V., Smirnova, N.N. et al. Thermodynamic characteristics of triphenylantimony diacrylate. Russ. J. Phys. Chem. 86, 1189–1195 (2012). https://doi.org/10.1134/S0036024412080079
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024412080079