Summary
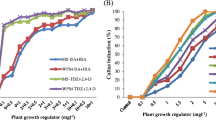
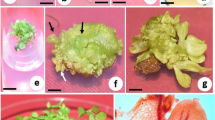
Chlorophytum borivilianum Sant. et Fernand. (‘Safed Musli’), an endangered Indian medicinal herb, is valued for its tuberous roots reputed to have aphrodisiac properties. Farmers in India cultivate this medicinal herb on a commercial scale because of its high economic value. To cater to the growing demand for planting material, a highly reproducible field-tested and cost-effective micropropagation scheme has been developed. Best shoot multiplication was achieved on agargelled Murashige and Skoog medium containing 22.2 μM 6-benzylaminopurine (BA) and 3% sucrose. Phytagel™ at 0.2% showed slightly better response than BDH agar in terms of shoot multiplication, but the use of BDH agar was preferred due to its low cost. With the optimized conditions, more than 15 000 plantlets could be produced in 20 wk. Plantlets subjected to hardening under agro-shadenet conditions during the monsoon months of high humidity showed better survival rate and growth compared to plantlets hardened in vitro and subsequently transferred to the greenhouse for acclimatization. Rate of plantlet survival was 87 and 90% under open field and agro-shadenet conditions, respectively. Plantlets grown ex vitro under agro-shadenet and field conditions produced tuberous roots which could be grown in the next season as a secondary propagule. We concluded that in vitro production of ‘Safed Musli’ was cost-effective compared to conventional propagation and holds great potential for commercial production.
Similar content being viewed by others
References
Anonymous. Poplar. Plant tissue culture from research to commercialization —a decade of support. A report by the Department of Biotechnology, Ministry of Science and Technology, Government of India, New Delhi; 2000:74–75.
Chu, I. Y. E.; Kurtz, S. L. Commercialization of plant micropropagation. In: Ammirato, P. V.; Evans, D. A.; Sharp, W. R.; Bajaj, Y. P. S., eds. Handbook of plant cell culture, Vol. 5. New York: McGraw Hill; 1990:126–164.
Debergh, P. C.; Maene, L. J. A scheme for commercial propagation of ornamental plants by tissue culture. Sci. Hort. 14:335–345; 1981.
Gamborg, O. L.; Miller, R. A.; Ojima, K. Nutrient requirements of suspension cultures of soyabean root cells. Exp. Cell. Res. 50:151–158; 1968.
Gupta, P. K.; Mascarenhas, A. F. Eucalyptus. In: Bonga, J. M.; Durzan, D. J., eds. Cell and tissue culture in forestry, Vol. 3. Dordrecht: Martinus Nijhoff; 1987:385–399.
Gupta, P. K.; Shaw, D.; Durzan, D. J. Loblolly pine. Micropropagation, somatic embryogenesis and encapsulation. In: Bonga, J. M.; Durzan, D. J., eds. Cell and tissue culture in forestry, Vol. 3. Dordrecht: Martinus Nijhoff; 1987:101–108.
Hussey, G. In vitro propagation of horticultural and agricultural crops. In: Mantell, S. H.; Smith, H., eds. Plant biotechnology. London: Cambridge University Press; 1983:111–138.
Jat, R. D.; Bordia, P. C. Propagation studies in safed musli (Chlorophytum species). In: Chaudhary, B. L.; Aery, N. C.; Katewa, S. S., eds. Proceedings of the national symposium on advances in plant sciences: current status and emerging challenges. Udaipur: Department of Botany, M. L. Sukhadia University; 1990:46.
Kirtikar, K. R.; Basu, B. D. Liliaceae: Chlorophytum. In: Kirtikar, K. R.; Basu, B. D., eds. Indian medicinal plants. Allahabad: L. M. Basu; 1975:2508–2509.
Litz, R. E. Papaya (Carica papaya). In: Bajaj, Y. P. S., ed. Biotechnology in agriculture and forestry 1. Trees I. Berlin: Springer-Verlag; 1986:220–232.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Nayar, M. P.; Sastry, A. R. K. Chlorophytum borivilianum. In: Nayar, M. P.; Sastry, A. R. K., eds. Red data book of Indian plants, Vol. 2. Calcutta: Botanical Survey of India; 1988:142.
Purohit, S. D.; Dave, A.; Kukda, G. Micropropagation of safed musli (Chlorophytum borivilianum)—a rare Indian medicinal herb. Plant Cell Tiss. Organ Cult. 39:93–96; 1994a.
Purohit, S. D.; Kukda, G.; Sharma, P.; Tak, K. In vitro propagation of adult tree Wrightia tomentosa through enhanced axillary branching. Plant Sci. 103:67–72; 1994b.
Purohit, S. D.; Singhvi, A. Micropropagation of Achras sapota through enhanced axillary branching. Sci. Hort. 76:219–229; 1998.
Schenk, R. U.; Hildebrandt, A. C. Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can. J. Bot. 50:199–204; 1972.
Shah, R. R.; Patel, D. B.; Dalal, K. C.; Gupta, R. Feasibility of scale-up and commercial application of tissue culture technique for mass multiplication of Glycyrrhiza glabra. In: Tata, S. N., ed. Proceedings of the national seminar on plant tissue culture. New Delhi: Publications and Information Directorate, Indian Council of Agricultural Research; 1988:98–103.
Singh, F. In vitro orchid seed germination and cloning of orchids—a success story. In: Prakash, J.; Peirik, R. L. M., eds. Plant biotechnology. New Delhi: Oxford & IBH; 1993:85–109.
Suri, S. S.; Jain, S. K.; Arora, D. K.; Ramawat, K. G. In vitro high frequency regenaration of plantlets and tuberous root formation in Chlorophytum borivilianum. Gartenbauwissenschaft 64(3):106–110; 1999.
Varshney, A.; Dhawan, V.; Srivastava, P. S. A protocol for in vitro mass propagation of asiatic hybrids of lily through liquid culture. In Vitro Cell. Dev. Biol. Plant 36:383–391; 2000.
White, P. R., ed. The cultivation of animal and plant cells. New York: Ronald Press; 1963.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dave, A., Bilochi, G. & Purohit, S.D. Scaling-up production and field performance of micropropagated medicinal herb ‘Safed Musli’ (Chlorophytum borivilianum). In Vitro Cell.Dev.Biol.-Plant 39, 419–424 (2003). https://doi.org/10.1079/IVP2003431
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1079/IVP2003431