Abstract
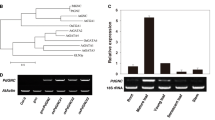
Transgenic rice (Oryza sativa) overexpressing Arabidopsis phytochrome A (phyA) was cultivated up to the T3 generation in paddy to elucidate the role of phyA in determining the plant architecture and the productivity of sunlight-grown rice plants. PhyA is light-labile and controls plant growth in response to the far-red light-dependent high-irradiance response as well as the very low fluence response. The Arabidopsis phyA gene linked to the rice rbcS promoter was transformed into embryogenic rice calli, and the calli were regenerated to whole plants. Compared to wild-type seedlings, the rbcS::PHYA transgenic seedlings contained more phyA when grown in the dark, and at least 10-fold more phyA when exposed to white light. When grown in paddy, the phyA transgenic plants in general exhibited reduced plant height (dwarfing), larger grain size, higher chlorophyll content, smaller tiller number, and low grain fertility compared to wild-type plants. The heading stage was not significantly changed. However, it is likely that a certain level of phyA is a prerequisite for induction of such changes. It is suggested that phyA overproduction in rice could be a useful tool to improve rice grain productivity by the larger grain size that increases grain yield and the dwarfing that tolerates lodging-associated damage.
Similar content being viewed by others
References
Basu D., Dehesh K., Schneider-Poetsch H.J., Harrington S.E., McCouch S.R. and Quail P.H. 2000. Rice PHYC gene: structure, expression, map position and evolution. Plant Mol. Biol. 44: 27–42.
Botto J.F. and Smith H. 2002. Differential genetic variation in adaptive strategies to a common environmental signal in Arabidopsisaccessions: phytochrome-mediated shade avoidance.Plant Cell Environ. 25: 53–63.
Boylan M.T. and Quail P.H. 1989. Oat phytochrome is biologically active in transgenic tomatoes. Plant Cell. 1: 765–773.
Boylan M.T. and Quail P.H. 1991. Phytochrome A overexpression inhibits hypocotyl elongation in transgenic Arabidopsis. Proc. Natl. Acad. Sci. USA 88: 10806–10810.
Bradford M.M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254.
Clough R.C., Casal J.J., Jordan E.T., Christou P. and Vierstra R.D. 1995. Expression of functional oat phytochrome A in transgenic rice. Plant Physiol. 109: 1039–1045.
Dehesh K., Tepperman J., Christensen A.H. and Quail P.H. 1991. PhyB is evolutionarily conserved and constitutively expressed in rice seedling shoots. Mol. Gen. Genet. 225: 305–313.
Duan X., Li X., Xue Q., Abo-el-Saad M., Xu D. and Wu R. 1996. Transgenic rice plants harboring an introduced potato proteinase inhibitor II gene are insect resistant. Nature Biotechnol. 14: 494–498.
Furuya M. 1993. Phytochormes: Their molecular species, gene families and functions. Annu. Rev. Plant physiol. Plant Mol. Biol. 44: 617–645.
Furuya M. and Hillman W.S. 1964. Observations on spectrophotometrically assayable phytochrome in vivoin etiolated Pisumseedlings. Planta 63: 31–42.
Halliday K.J., Thomas B. and Whitelam G.C. 1997. Expression of heterologous phytochrome A, B or C in transgenic tobacco plants alters vegetative development and flowering time. Plant J. 12: 1079–1090.
Hanzawa H., Inomata K., Kinoshita H., Kakiuchi T., Jayasundera K.P., Sawamoto D., Ohta A., Uchida K., Wada K. and Furuya M. 2001. In vitro assembly of phytochrome B apoprotein with synthetic analogs of the phytochrome chromophore. Proc. Natl. Acad. Sci. USA 98: 3612–3617.
Hiei Y., Ohta S., Komari T. and Kumashiro T. 1994. Efficient transformation of rice (Oryza sativaL.) mediated by Agrobacteriumand sequence analysis of the boundaries of the T-DNA. Plant J. 6: 271–282.
Jang I.-C., Nahm B.H. and Kim J.-K. 1999. Subcellular targeting of green fluorescent protein to plastids in transgenic rice plants provides a high-level expression system. Mol. Breeding 5: 453–461.
Jefferson R.A., Kavanaugh T.A. and Bevan M.W. 1987. GUS fusions: glucuronidase is a sensitive and versatile gene fusion marker in higher plants. EMBO J. 6: 3901–3907.
Jordan E.T., Hatfield P.M., Hondred D., Talon M., Zeevaart J.A.D. and Vierstra R.D. 1995. Phytochrome A overexpression in transgenic tobacco: Correlation of dwarf phenotype with high concentrations of phytochrome in vascular tissue and attenuated gibberellin levels. Plant Physiol. 107: 797–805.
Kay S.A., Keith B., Shinozaki K. and Chua N.H. 1989. The sequence of the rice phytochrome gene. Nucleic. Acids Res. 17: 2865–2866.
Keller J.M., Shanklin J., Vierstra R.D. and Hershey H.P. 1989. Expression of a functional monocotyledonous phytochorme in transgenic tobacco. EMBO J. 8: 1005–1012.
Kim J.-K., Duan X., Wu R., Seok S.J., Boston R.S., Jang I.-C., Eun M.-Y. and Nahm B.H. 1999. Molecular and genetic analysis of transgenic rice plants expressing the maize ribosome-inactivating protein b-32 gene and the herbicide resistance bargene. Mol. Breeding 5: 85–94.
Kyozuka J., McElroy D., Hayakawa T., Xie Y., Wu R. and Shimamoto K. 1993. Light-regulated and cell-specific expression of tomato rbcS-gusA and rice rbcS-gusA fusion genes in transgenic rice. Plant Physiol. 102: 991–1000.
Mathews S. and Sharrock R.A. 1997. Phytochrome gene diversity. Plant Cell Environ. 20: 666–671.
Pjon C.J. and Furuya M. 1968. Phytochrome: Phytochrome Action in Oryza sativaL. II. The spectrophotometeric versus the physiological status of phytochrome in coleoptiles. Planta 81: 303–313.
Porra R.J., Thompson W.A. and Kriedemann P.E. 1989. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim. Biophys. Acta 975: 384–394.
Qin M., Kuhn R., Moran S. and Quail P.H. 1997. Overexpressed phytochrome C has similar photosensory specificity to phytochrome B but a distinctive capacity to enhance primary leaf expansion. Plant J. 12: 1163–1172.
Quail P.H., Boylan M.T., Parks B.M., Short T.W., Xu Y. and Wagner D. 1995. Phytochromes: Photosensory perception and signal transduction. Science 268: 675–680.
Robson P.R.H., McCormac A.C., Irvine A.S. and Smith H. 1996. Genetic engineering of harvest index in tobacco through overexpression of a phytochrome gene. Nature Biotechnol. 14: 995–998.
Robson P.R.H. and Smith H. 1997. Fundamental and biotechnological applications of phytochrome transgenes. Plant Cell Environ. 20: 831–839.
Sharrock R.A. and Quail P.H. 1989. Novel phytochrome sequences in Arabidopsis thaliana: Structure, evolution, and differential expression of a plant regulatory photoreceptor family. Genes Dev. 3: 1745–1757.
Shlumukov L.R., Barro F., Barcelo P., Lazzeri P. and Smith H. 2001. Establishment of far-red high irradiance responses in wheat through transgenic expression of an oat phytochrome A gene. Plant Cell Environ. 24: 703–712.
Smith H. 1982. Light quality, photoperception and plant strategy. Annu Rev Plant physiol Plant Mol. Biol. 33: 481–518.
Smith H. 2000. Phytochromes and light signal perception by plants-an emerging synthesis. Nature 407: 585–591.
Somers D.E. and Quail P.H. 1995. Phytochrome-mediated light regulation of PHYA-and PHYB-GUStransgene in Arabidopsis thalianaseedling. Plant Physiol. 107: 523–534.
Takano M., Kanegae H., Shinomura T., Miyao A., Hirochika H. and Furuya M. 2001. Isolation and characterization of rice phytochrome A mutants. Plant Cell 13: 521–534.
Thiele A., Herold M., Lenk I., Quail P.H., and Gatz C. 1999. Heterologous expression of Arabidopsis phytochrome B in transgenic potato influences photosynthetic performance and tuber development. Plant Physiol. 120: 73–82.
Vaucheret H., Beclin C., Elmayan T., Feuerbach F., Godon C., Morel J.B., Mourrain P., Palauqui J.C., Vernhettes S. 1998. Transgene-induced gene silencing in plants. Plant J. 16: 651–659.
Yanovsky M.J., Alconada-Magliano T.M., Mazzella M.A., Gatz C., Thomas B. and Casal J. 1998. Phytochrome A affects stem growth, anthocyanin synthesis, sucrose-phosphate-synthase activity and neighbour detection in sunlight-grown potato. Planta 205: 235–241.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kong, SG., Lee, DS., Kwak, SN. et al. Characterization of sunlight-grown transgenic rice plants expressing Arabidopsis phytochrome A. Molecular Breeding 14, 35–46 (2004). https://doi.org/10.1023/B:MOLB.0000037993.79486.7b
Issue Date:
DOI: https://doi.org/10.1023/B:MOLB.0000037993.79486.7b