Abstract
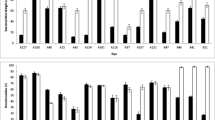
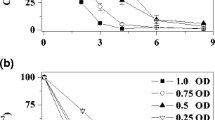
A recombinant Escherichia coli strain (E. coli NO3) containing genomic DNA fragments from azo-reducing wild-type Pseudomonas luteola strain decolorized a reactive azo dye (C.I. Reactive Red 22) at approx. 17 mg dye h−1 g cell. The ability to decolorize the azo dye probably did not originate from the plasmid DNA. Acclimation in azo-dye-containing media gave a nearly 10% increase in the decolorization rate of E. coli NO3. Growth with 1.25 g glucose l−1 completely stopped the decolorization activity. When the decolorization metabolites from E. coli NO3 were analyzed by HPLC and MS, the results suggested that decolorization of the azo dye may be due to cleavage of the azo bond.
Similar content being viewed by others
References
Banat IM, Nigam P, Singh D, Marchant R (1996) Microbial decolorization of textile-dye-containting effluents: A review. Biores.Technol. 58: 217–227.
Carliell CM, Barclay SJ, Naidoo N, Buckley CA, Mulholland DA, Senior E (1995) Microbial decolourization of a reactive azo dye under anaerobic conditions. Water SA 21: 61–69.
Chung K-T, Stevens SE Jr (1993) Degradation of azo dyes by environmental microorganisms and helminths. Environ.Toxicol.Chem. 12: 2121–2132.
Coughlin MF, Kinkle BK, Tepper A, Bishop PL (1997) Characterization of aerobic azo dye-degrading bacteria and their activity in biofilms. Wat.Sci.Technol. 36: 215–220.
Flores ER, Luijten M, Donlon BA, Lettinga G, Field JA (1997) Complete biodegradation of the azo dye azodisalicylate under anaerobic conditions. Environ.Sci.Technol. 31: 2098–2103.
Ghosh DK, Ghosh S, Sadhukhan P, Mandal A, Chaudhuri J (1993) Purification of two azoreductases from Escherichia coli K12. Ind.J.Exp.Biol. 31: 951–954.
Glenn JK, Gold MH (1983) Decolorization of several polymeric dyes by the lignin-degrading Basidiomycete Phanerochaete chrysosporium. Appl.Environ.Microbiol. 45: 1741–1747.
Hashimoto-Gotoh T, Tsujimura A, Kuriyama K-Y, Matsuda S (1993) Construction and characterization of new host-vector systems for the enforcement-cloning method. Gene 137: 211–216.
Haug W, Schmidt A, Nortemann B, Hempel DC, Stolz A, Knackmuss H-J (1991) Mineralization of the sulfonated azo dye mordant yellow 3 by 6-aminonaphthalene-2-sulfonate-degrading bacterial consortium. Appl.Environ.Microbiol. 57: 3144–3149.
Heiss GS, Gowan B, Dabbs ER (1992) Cloning of DNA from a Rhodococcus strain conferring the ability to decolorize sulfonated azo dyes. FEMS Microbiol.Lett. 99: 221–226.
Hu TL (1994) Decolourization of reactive azo dyes by transformation with Pseudomonas luteola. Biores.Technol. 49: 47–51.
Michaels GB, Lewis DL (1986) Microbial transformation rates of azo and triphenylmethane dyes. Environ.Toxicol.Chem. 5: 161–166.
Nigam P, Marchant R (1995) Selection of a substratum for composing biofilm system of a textile-effluent decolorizing bacteria. Biotechol.Lett. 17: 993–996.
Rafii F, Coleman T (1999) Cloning and expression in Escherichia coli of an azoreductase gene from Clostridium perfringens and comparison with azoreductase genes from other bacteria. J.Basic Microbiol. 39: 29–35.
Seshadri S, Bishop PL, Agha AM (1994) Anaerobic/aerobic treatment of selected azo dyes in wastewater. Waste Manage. 14: 127–137.
Yang F, Yu J (1996) Development of a bioreactor system using an immobilized white rot fungus for decolourization, Part II: Continuous decolourization tests. Bioprocess Eng. 16: 9–11.
Zhang F-M, Knapp JS, Tapley KN (1999) Development of bioreactor systems for decolorization of orange II using white rot fungus. Enzyme Microbiol.Technol. 24: 48–53.
Zimmermann T, Kulla HG, Leisinger T (1982) Properties of purified orange II azoreductase, the enzyme initiating azo dye degradation by Pseudomonas KF46. Eur.J.Biochem. 129: 197–203.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chang, JS., Kuo, TS., Chao, YP. et al. Azo dye decolorization with a mutant Escherichia coli strain. Biotechnology Letters 22, 807–812 (2000). https://doi.org/10.1023/A:1005624707777
Issue Date:
DOI: https://doi.org/10.1023/A:1005624707777