Abstract
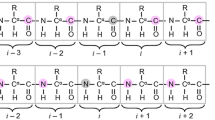
The TROSY principle has been introduced into a HNCA experiment, which is designed for measurements of the intraresidual and sequential Hα-Cα/HN-N dipole/dipole and Hα-Cα/N dipole/CSA cross-correlated relaxation rates. In addition, the new experiment provides values of the 3,4 J Hα HN coupling constants measured in an E.COSY manner. The conformational restraints for the ψ and φ angles are obtained through the use of the cross-correlated relaxation rates together with the Karplus-type dependencies of the coupling constants. Improved signal-to-noise is achieved through preservation of all coherence transfer pathways and application of the TROSY principle. The application of the [15N,13C]-DQ/ZQ-[15N,1H]-TROSY-E.COSY experiment to the 16 kDa apo-form of the E. coli Heme Chaperon protein CcmE is described. Overall good agreement is achieved between ψ and φ angles measured with the new experiment and the average values determined from an ensemble of 20 NMR conformers.
Similar content being viewed by others
REFERENCES
Abragam, A. (1961) The Principles of Nuclear Magnetism, Clarendon Press, Oxford.
Case, D.A. (1999) J. Biomol. NMR, 15, 95–102.
Chiarparin, E., Pelupessy, P., Ghose, R. and Bodenhausen, G. (1999) J. Am. Chem. Soc., 121, 6876–6883.
Ernst, M. and Ernst, R.R. (1994) J. Magn. Reson. Ser., A110, 202–213.
Geen, H. and Freeman, R. (1991) J. Magn. Reson., 93, 93–141.
Griesinger, C., Sorensen, O.W. and Ernst, R.R. (1987) J. Magn. Reson., 75, 474–492.
Güntert, P., Dotsch, V., Wider, G. and Wüthrich, K. (1992) J. Biomol. NMR, 2, 619–629.
Karplus, M. (1959) J. Chem. Phys., 30, 11–15.
Kloiber, K. and Konrat, R. (2000a) J. Biomol. NMR, 17, 265–268.
Kloiber, K. and Konrat, R. (2000b) J. Am. Chem. Soc., 122, 12033–12034.
Kloiber, K., Schuler, W. and Konrat, R. (2002) J. Biomol. NMR, 22, 349–363.
Korzhnev, D.M., Billeter, M., Arseniev, A.S. and Orekhov, V.Y. (2001) Prog. Nucl. Magn. Reson. Spectrosc., 38, 197–266.
McCoy, M.A. and Mueller, L. (1992) J. Am. Chem. Soc., 114, 2108–2112. 300
Oas, T.G., Hartzell, C.J., Dahlquist, F.W. and Drobny, G.P. (1987) J. Am. Chem. Soc., 109, 5962–5966.
Pang, Y.X., Wang, L.C., Pellecchia, M., Kurochkin, A.V. and Zuiderweg, E.R.P. (1999) J. Biomol. NMR, 14, 297–306.
Pelupessy, P., Chiarparin, E., Ghose, R. and Bodenhausen, G. (1999a) J. Biomol. NMR, 13, 375–380.
Pelupessy, P., Chiarparin, E., Ghose, R. and Bodenhausen, G. (1999b) J. Biomol. NMR, 14, 277–280.
Pervushin, K. (2000) Quart. Rev. Biophys, 33, 161–197.
Pervushin, K., Riek, R., Wider, G. and Wüthrich, K. (1997) Proc. Natl. Acad. Sci. USA, 94, 12366–12371.
Pervushin, K., Wider, G. and Wüthrich, K. (1998) J. Biomol. NMR, 12, 345–348.
Reif, B., Diener, A., Hennig, M., Maurer, M. and Griesinger, C. (2000) J. Magn. Reson., 143, 45–68.
Reif, B., Hennig, M. and Griesinger, C. (1997) Science, 276, 1230–1233.
Roberts, J.E., Harbison, G.S., Munowitz, M.G., Herzfeld, J. and Griffin, R.G. (1987) J. Am. Chem. Soc., 109, 4163–4169.
Sørensen, O.W., Eich, G.W., Levitt, M.H., Bodenhausen, G. and Ernst, R.R. (1983) Prog. Nucl. Magn. Reson. Spectrosc., 16, 163–192.
Sprangers, R., Bottomley, M.J., Linge, J.P., Schultz, J., Nilges, M. and Sattler, M. (2000) J. Biomol. NMR, 16, 47–58.
Tjandra, N., Szabo, A. and Bax, A. (1996) J. Am. Chem. Soc., 118, 6986–6991.
Vincent, S.J.F., Zwahlen, C., Bolton, P.H., Logan, T.M. and Bodenhausen, G. (1996) J. Am. Chem. Soc., 118, 3531–3532.
Vuister, G.W. and Bax, A. (1994) J. Biomol. NMR, 4, 193–200.
Wagner, G., Schmieder, P. and Thanabal, V. (1991) J. Magn. Reson., 93, 436–440.
Wang, A.C. and Bax, A. (1996) J. Am. Chem. Soc., 118, 2483–2494.
Weisemann, R., Ruterjans, H., Schwalbe, H., Schleucher, J., Bermel, W. and Griesinger, C. (1994) J. Biomol. NMR, 4, 231–240.
Wüthrich, K. (1986) NMR of Proteins and Nucleic Acids, Wiley, New York, NY.
Yang, D.W. and Kay, L.E. (1998) J. Am. Chem. Soc., 120, 9880–9887.
Yang, D.W., Gardner, K.H. and Kay, L.E. (1998) J. Biomol. NMR, 11, 213–220.
Yang, D.W., Konrat, R. and Kay, L.E. (1997) J. Am. Chem. Soc., 119, 11938–11940.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vögeli, B., Pervushin, K. TROSY experiment for refinement of backbone ψ and φ by simultaneous measurements of cross-correlated relaxation rates and 3,4 J HαHN coupling constants. J Biomol NMR 24, 291–300 (2002). https://doi.org/10.1023/A:1021677216126
Issue Date:
DOI: https://doi.org/10.1023/A:1021677216126