Abstract
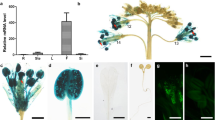
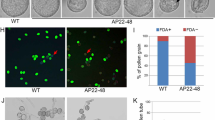
A mutant exhibiting conditional male sterility, in which fertility was restored under conditions of high humidity, was identified in T-DNA tagged lines of Arabidopsis thaliana. Scanning electron microscopy (SEM) demonstrated that the pollen surface was almost smooth and the reticulate pattern not prominent. Thus, the mutant was named faceless pollen-1 (flp1). Transmission electron microscopy (TEM) revealed that the smooth appearance was due to tryphine filling in the exine cavities and covering the pollen surface. The lipid droplets in the tryphine of mutant pollen were smaller and more numerous than those of the wild type. SEM analysis also demonstrated that pollen exine was easily damaged by acetolysis, suggesting that a component of exine, sporopollenin, was defective in the mutant. In addition, the stems and siliques had reduced amounts of wax crystals. A predicted amino acid sequence of the cDNA that corresponded to the tagged gene, flp1, showed sequence similarity to proteins involved in wax biosynthesis. The FLP1 protein is likely to play a role in the synthesis of the components of tryphine, sporopollenin of exine and the wax of stems and siliques.
Similar content being viewed by others
References
Aarts, M.G.M., Keijzer, C.J., Stiekema, W.J. and Pereira, A. 1995. Molecular characterization of the CER1 gene of Arabidopsis involved in epicuticular wax biosynthesis and pollen fertility. Plant Cell 7: 2115-2127.
Aarts, M.G.M., Hodge, R., Kalantidis, K., Florack, D., Wilson, Z.A., Mulligan, B., Stiekema, W.J., Scott, R. and Pereira, A. 1997. The Arabidopsis MALE STERILITY 2 protein shares similarity with reductases in elongation/condensation complexes. Plant J. 12: 615-623.
Ahlers, H., Thom, I., Lambert, J., Kuckuk, R. and Wiermann, R. 1999. 1H NMR analysis of sporopollenin from Typha angustifolia. Phytochemistry 50: 1095-1098.
Amagai, M., Ariizumi, T., Endo, M., Hatakeyama, K., Kuwata, C., Shibata, D., Toriyama, K. and Watanabe, M. 2003. Identification of anther-specific genes in a cruciferous model plant, Arabidopsis thaliana, by using a combination of Arabidopsis macroarray and mRNA derived from Brassica oleracea. Sex. Plant Reprod. 15: 213-220.
An, G., Ebert, P.R., Mitra, A. and Ha, S.B. 1988. Binary vectors. In: S.B. Gelvin and R.A. Schilperoort (Eds.) Plant Molecular Biology Manual, Kluwer Academic Publishers, Dordrecht, Netherlands, pp. 1-19.
Bai, X.F., Peirson, B.N., Dong, F.G., Xue, C. and Makaroff, C.A. 1999. Isolation and characterization of SYN1, a RAD21-like gene essential for meiosis in Arabidopsis. Plant Cell 11: 417-430.
Bevan, M. 1984. Binary Agrobacterium vector for plant transformation. Nucl. Acids Res. 12: 8711-8721.
Clough, S.J. and Bent, A.F. 1998. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16: 735-743.
De Block, M., Botterman, J., Vandewiele, M., Dockx, J., Thoen, C., Gosselé, V., Rao Movva, N., Thompson, C., Van Montagu, M. and Leemans, J. 1987. Engineering herbicide resistance in plants by expression of a detoxifying enzyme. EMBO J. 6: 2513-2518.
Fiebig, A., Mayfield, J.A., Miley, N.L., Chau, S., Fischer, R.L. and Preuss, D. 2000. Alterations in CER6, a gene identical to CUT1, differentially affect long-chain lipid content on the surface of pollen and stem. Plant Cell 12: 2001-2008.
Hatakeyama, K., Takasaki, T., Suzuki, G., Nishio, T, Watanabe, M, Isogai, A. and Hinata, K. 2001. The S receptor kinase gene determines dominance relationships in stigma expression of selfincompatibility in Brassica. Plant J. 26: 69-76.
Hannoufa, A., McNevin, J.P. and Leumieux, B. 1993. Epicuticular waxes of eceriferum mutants of Arabidopsis thaliana. Phytochemistry 33: 851-855.
Hannoufa, A., Negruk, V., Eisner, G. and Leumieux, B. 1996. The CER3 gene of Arabidopsis thaliana is expressed in leaves, stems, roots, flowers and apical meristems. Plant J. 10: 459-467.
Hansen, J.D., Pyee, J., Xia, Y., Wen, T.J., Robertson, D.S., Kolattukudy, P.E., Nikolau, B.J. and Schnable, P.S. 1997. The glossy 1 locus of maize and an epidermis-specific cDNA from Klenia odora define a class of receptor-like proteins required for the normal accumulation of cuticular waxes. Plant Physiol. 113: 1091-1100.
Hauge, B.M., Hanley, S.M., Cartinhour, S., Cherry J.M. and Goodman, H.M. 1993. An integrated genetic/RFLP map of the Arabidopsis thaliana genome. Plant J. 3: 745-754.
Hofmann, K. and Stoffel, W. 1993. TMbase: a database of membrane spanning protein segments. Biol. Chem. Hoppe-Seyler 347: 166.
Hulskamp, M., Kopczak, S.D., Horejsi, T.F., Kihl, B.K. and Pruitt, R.E. 1995. Identification of genes required for pollen-stigma recognition in Arabidopsis thaliana. Plant J. 8: 703-714.
Ishiguro, S., Kawai-Oda, A., Ueda, J., Nishida, I. and Okada, K. 2001. The DEFECTIVE IN ANTHER DEHISCENCE1 gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 13: 2191-2209.
Kimura, R., Sato, K., Fujimoto, R. and Nishio, T. 2002. Recognition specificity of self-incompatibility maintained after the divergence of Brassica oleracea and Brassica rapa. Plant J. 29: 215-223.
Koornneef, M., Hanhart, C.J. and Thiel, F. 1989. A genetic and phenotypic description of eceriferum (cer) mutants in Arabidopsis thaliana. J. Hered. 80: 118-122.
Lemieux, B. 1996. Molecular genetics of epicuticular wax biosynthesis. Trends Plant Sci. 1: 312-318.
Liu, Y.G. and Whittier, R.F. 1995. Thermal asymmetric interlaced PCR: automatable amplification and sequencing of insert end fragments from P1 and YAC clones for chromosome walking. Genomics 25: 674-681.
Lolle, S. J., Hsu, W. and Pruitt, R.E. 1998. Genetic analysis of organ fusion in Arabidopsis thaliana. Genetics 149: 607-619.
Meuter-Gerhards, A., Riegart, S. and Wiermann, R. 1999. Studies on sporopollenin biosynthesis in Curcurbita maxima (DUCH)-II: the involvement of aliphatic metabolism. J. Plant Physiol. 154: 431-436.
Millar, A.A., Clemens, S., Zachgo, S., Giblin, E.M., Taylor, D.C. and Kunst, L. 1999. CUT1, an Arabidopsis gene required for cuticular wax biosynthesis and pollen fertility, encodes a verylong-chain fatty acid condensing enzyme. Plant Cell 11: 825-838.
Murashige, T. and Skoog, F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473-497.
Nakai, K. and Kanehisa, M. 1992. A knowledge base for predicting protein localization sites in eukaryotic cells. Genomics 14: 897-911.
Negruk, V., Yang, P., Subramanian, M., McNevin, J.P. and Lemieux, B. 1996. Molecular cloning and characterization of the CER2 gene of Arabidopsis thaliana. Plant J. 9: 137-145.
Osthoff, K.S. and Wiermann, R. 1987. Phenols as integrated compounds of sporopollenin from Pinus pollen. J. Plant Physiol. 131: 5-15.
Paxson-Sowders, D.M., Owen, H.A. and Makaroff, C.A. 1997. A comparative ultrastructural analysis of exine pattern development in wild-type Arabidopsis and a mutant defective in pattern formation. Protoplasma 198: 53-65.
Paxson-Sowders, D.M., Dodrill, C.H., Owen, H.A. and Makaroff, C.A. 2001. DEX1, a novel plant protein is required for exine pattern formation during development in Arabidopsis. Plant Physiol. 127: 1739-1749.
Peirson, B.N., Bowling, S.E. and Makaroff, C.A. 1997. A defect in synapsis causes male sterility in a T-DNA-tagged Arabidopsis thaliana mutant. Plant J. 11: 659-669.
Piffanelli, P., Ross, J.H.E. and Murphy, D.J. 1998. Biogenesis and function of the lipidic structures of pollen grains. Sex. Plant Reprod. 11: 65-80.
Preuss, D., Lemieux, B., Yen, G. and Davis, R.W. 1993. A conditional sterile mutation eliminates surface components from Arabidopsis pollen and disrupts cell signaling during fertilization. Genes Dev. 7: 974-985.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual, 2nd ed. Cold Spring Harbor Laboratory Press, Plainview, NY.
Sanders, P.M., Bui, A.Q., Weterings, K., Mclntire, K.N., Hsu, Y.C., Lee, P.Y., Truong, M.T., Beals, T.P. and Goldberg, R.B. 1999. Anther developmental defects in Arabidopsis thaliana male-sterile mutants. Sex. Plant Reprod. 11: 297-322.
Schnable, P.S., Stinard, P.S., Wen, T.-J., Heinen, S., Weber, D., Schneerman, M., Zhang, L, Hansen, J.D. and Nikolau, B.J. 1994. The genetics of cuticular wax biosynthesis. Maydica 39: 279-287.
Scott, R.J. 1994. Pollen exine: the sporopollenin enigma and the physics of pattern. In: R.J. Scott and M.A. Stead (Eds.) Molecular and Cellular Aspects of Plant Reproduction, Cambridge University Press, Cambridge, UK, pp. 49-81.
Shanklin, J., Whittle, E. and Fox, B.G. 1994. Eight histidine residues are catalytically essential in a membrane-associated iron enzyme, stearoyl-CoA desaturase, and are conserved in alkane hydroxylase and xylene monooxygenase. Biochemistry 33: 12787-12794.
Taylor, P.E., Glover, J.A., Lavithis, M., Craig, S., Singh, M.B., Knox, R.B., Dennis, E.S. and Chaudhury, A.M. 1998. Genetic control of male fertility in Arabidopsis thaliana: structural analyses of postmeiotic developmental mutants. Planta 205: 492-505.
Xia, Y., Nikolau, B.J. and Schnable, P. 1996. Cloning and characterization of CER2, an Arabidopsis gene that affects cuticular wax accumulation. Plant Cell 8: 1291-1304.
Zinkl, G.M., Zwiebel, B.I., Grier, D.G. and Preuss, D. 1999. Pollen-stigma adhesion in Arabidopsis: a species-specific interaction mediated by lipophilic molecules in the pollen exine. Development 126: 5431-5440.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ariizumi, T., Hatakeyama, K., Hinata, K. et al. A novel male-sterile mutant of Arabidopsis thaliana, faceless pollen-1, produces pollen with a smooth surface and an acetolysis-sensitive exine. Plant Mol Biol 53, 107–116 (2003). https://doi.org/10.1023/B:PLAN.0000009269.97773.70
Issue Date:
DOI: https://doi.org/10.1023/B:PLAN.0000009269.97773.70