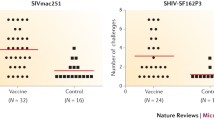
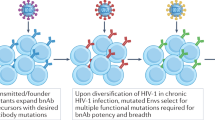
Abstract
Considerable progress has been made over the past several years in the development of an HIV vaccine. As a result, a growing number of vaccine modalities are being investigated in pre-clinical and phase I/II clinical trials. However, a number of major scientific challenges still remain. It is widely believed that the ideal vaccine should elicit both neutralizing antibodies and cytotoxic T lymphocytes (CTL) against diverse isolates of HIV, but the precise correlates of immunity have not been defined. Recombinant live vector-based vaccines and plasmid DNA vaccines have been shown to induce CTL, either alone or in combination, and these CTL-based vaccines have shown partial protective efficacy in nonhuman primates challenge studies. An immunogen that elicits broadly reactive neutralizing antibodies, however, has yet to be developed.
Article PDF
Similar content being viewed by others
References
Fowke KR, Nagelkerke NJ, Kimani J, et al. Resistance to HIV-1 infection among persistently seronegative prostitutes in Nairobi, Kenya. Lancet 1996;348:1347–1351.
Rowland-Jones S, Sutton J, Ariyoshi K, et al. HIV-specific cytotoxic T-cells in HIV-exposed but uninfected Gambian women. Nat Med 1995;1:59–64.
Wagner R, Leschonsky B, Harrer E, et al. Molecular and functional analysis of a conserved CTL epitope in HIV-1 p24 recognized from a long-term nonprogressor: Constraints on immune escape associated with target-ing a sequence essential for viral replication. J Immunol 1999;162:3727–3734.
Prince AM, Reesink H, Pascual D, et al. Prevention of HIV infection by passive immunization with HIV im-munoglobuline. AIDS Res Hum Retroviruses 1991;7: 971–973.
Putkonen P, Thorstensson R, Ghavamzadeh L, et al. Prevention of HIV-2 and SIVsm infection by passive immunization in cynomolgus monkeys. Nature 1991; 352:436–438.
Emini EA, Schleif WA, Nunberg JH, et al. Prevention of HIV-1 infection in chimpanzees by gp120 V3 domain-speci c monoclonal antibody. Nature 1992; 355:728–730.
Mascola JR, Lewis MG, Stiegler G, et al. Protection of macaques against pathogenic simian/human immunodeficiency virus 89. 6PD by passive transfer of neutralizing antibodies. J Virol 1999;73:4009–4018.
Barouch DH, Santra S, Schmitz JE, et al. Control of viremia and prevention of clinical AIDS in rhesus monkeys by cytokine-augmented DNA vaccination. Science 2000;290:486–492.
Amara RR, Villinger F, Altman JD, et al. Control of a mucosal challenge and prevention of AIDS by a mul-tiprotein DNA/MVA vaccine. Science 2001;292: 69–74.
Shiver JW, Fu T, Chen L, et al. Replication-incompe-tent adenoviral vaccine vector elicits effective anti-immunodeficiency-virus immunity. Nature 2002;415: 331–335.
Barouch DH, Kunstman J, Kuroda MJ, et al. Eventual AIDS vaccine failure in a rhesus monkey by viral escape from cytotoxic T lymphocytes. Nature 2002; 415:335–339.
Parren PW, Burton DR, Sattentau QJ. HIV-1 antibody –debris or virion? Nat Med 1997;3:366–367.
Moore JP, Cao Y, Qing L, et al. Primary isolates of human immunode ciency virus type 1 are relatively resistant to neutralization by monoclonal antibodies to gp120, and their neutralization is not predicted by studies with monomeric gp120. J Virol 1995;69:101–109.
Parren PW, Moore JP, Burton DR, et al. The neutralizing antibody response to HIV-1:Viral evasion and escape from humoral immunity. AIDS 1999;13:S137–162.
Chackerian B, Rudensey LM, Overbaugh J. Specific N-linked and O-linked glycosylation modi cations in the envelope V1 domain of simian immunodeficiency virus variants that evolve in the host alter recognitionby neutralizing antibodies. J Virol 1997;71:7719–7727.
Wei X, Decker JM, Wang S, et al. Antibody neutralization and escape by HIV-1. Nature 2003;422: 307–312.
Borrow P, Lewicki H, Hahn BH, et al. Virus-speci c CD8+cytolytic T-lymphocyte activity associated with control of viremia in primary human immunodeficiency virus type 1 infection. J Virol 1994;68:6103–6110.
Schmitz JE, Kuroda MJ, Santra S, et al. Control of viremia in simian immunodeficiency virus infection by CD8+lymphocytes. Science 1999;283:857–860.
Cao Y, Qin L, Zhang L, et al. Virologic and immunologic characterization of long-term survivors of human immunodeficiency virus type 1 infection. New Engl J Med 1995;332:201–208.
Ballegooijen van M, Bogaards JA, Weverling GJ, et al. AIDS vaccines that allow HIV-1 to infect and escape immunologic control:A mathematic analysis of mass vaccination. J Acq Immun Def Synd 2003;34:214–220.
Goulder PJ, Brander C, Tang Y, et al. Evolution and transmission of stable CTL escape mutations in HIV infection. Nature 2001;412:334–338.
Koenig S, Conley AJ, Brewah YA, et al. Transfer of HIV-1-speci c cytotoxic T lymphocytes to an AIDS patient leads to selection for mutant HIV variants and sub-sequent disease progression. Nat Med 1995;1:330–336.
Price DA, Goulder PJ, Klenerman P, et al. Positive selection of HIV-1cytotoxic T lymphocyte escape variants during primary infection. Proc Natl Acad Sci USA 1997;94:1890–1895.
Rosenberg ES, Billingsley JM, Caliendo AM, et al. Vigorous HIV-1-specific CD4+T cell responses asso-ciated with control of viremia. Science 1997;278:1447–1450.
McMichael AJ, Rowland-Jones SL. Cellular immune responses to HIV. Nature 2001;410:980–987.
Cohen J. AIDS vaccine trial produces disappointment and confusion. Science 2003;299:1290–1291.
Baalen van CA, Pontesilli O, Huisman RC, et al. Human immunode ciency virus type 1 Rev-and Tat-speci c cytotoxic T lymphocyte frequencies inversely correlate with rapid progression to AIDS. J Gen Virol 1997;78:1913–1918.
Pontesilli O, Klein MR, Kerkhof-Garde SR, et al. Longitudinal analysis of human immunode ciency virus type 1-speci c cytotoxic T lymphocyte responses: A predominant gag-speci c response is associated with nonprogressive infection. J Infect Dis 1998;178:1008–1018.
Klein MR, Baalen van CA, Holwerda AM, et al. Kinetics of Gag-specific cytotoxic T lymphocyte responses during the clinical course of HIV-1 infection: A longitudinal analysis of rapid progressors and long-term asymptomatics. J Exp Med 1995;181:1365–1372.
Seth A, Ourmanov I, Schmitz JE, et al. Immunization with a modi ed vaccinia virus expressing simian immunodeficiency virus (SIV)Gag-Pol primes for an anamnestic Gag-speci c cytotoxic T-lymphocyte response and is associated with reduction of viremia after SIV challenge. J Virol 2000;74:2502–2509.
Abimiku AG, Robert-Guro. M, Benson J, et al. Long-term survival of SIVmac251-infected macaques previ-ously immunized with NYVAC-SIV vaccines. J Acq Immun Def Synd 1997;15:S78–S85.
Barouch DH, Santra S, Kuroda MJ, et al. Reduction of simian-human immunode ciency virus 89.6P viremia in rhesus monkeys by recombinant modified vaccinia virus Ankara vaccination. J Virol 2001;75: 5151–5158.
Kent SJ, Zhao A, Best SJ, et al. Enhanced T-cell immunogenicity and protective efficacy of a human immunode ciency virus type 1 vaccine regimen consisting of consecutive priming with DNA and boosting with recombinant fowlpox virus. J Virol 1998;72: 10180–10188.
Hammond SA, Bollinger RC, Stanhope PE, et al. Comparative clonal analysis of human immunodeciency virus type 1 (HIV-1)-specific CD4+and CD8+ cytotoxic T lymphocytes isolated from seronegative humans immunized with candidate HIV-1 vaccines. J Exp Med 1992;176:1531–1542.
Evans TG, Keefer MC, Weinhold KJ, et al. A canarypox vaccine expressing multiple HIV-1 genes given alone or with rgp120 elicits broad and durable CD8+CTL responses in seronegative volunteers. J Infect Dis 1999;180:290–298.
Belshe RB, Stevens C, Gorse GJ, et al. Safety and immunogenicity of a canarypox-vectored human immunode ciency virus type 1 vaccine with or without gp120:A phase 2 study in higher and lower-risk volunteers. J Infect Dis 2001;183:1343–1352.
Sullivan NJ, Sanchez A, Rollin PE, et al. Development of a preventive vaccine for Ebola virus infection in primates. Nature 2000;30:605–609.
Casimiro DR, Chen L, Fu TM, et al. Comparative immunogenicity in rhesus monkeys of DNA plasmid, recombinant vaccinia virus, and replication-defective adenovirus vectors expressing a human immunodeficiency virus type 1 gag gene. J Virol 2003;77:6305–6313.
Farina SF, Gao GP, Xiang ZQ, et al. Replication-defective vector based on a chimpanzee adenovirus. J Virol 2001;75:11603–11613.
Vogels R, Zuijdgeest D, Rijnsoever van R, et al. Replication-deficient human adenovirus type 35 vectors for gene transfer and vaccination:Eficient human cell interaction and bypass of preexisting adenovirus immunity. J Virol 2003;77:8263–8271.
Shiver JW, Emini EA. Recent advances in the development of HIV-1 vaccines using replication-incompe-tent adenovirus vectors. Annu Rev Med 2004;55: 355–372.
Mossman SP, Bex F, Berglund P, et al. Protection against lethal simian immunode ciency virus SIV-smmPBj14 disease by a recombinant Semliki Forest virus gp160 vaccine and by a gp120 subunit vaccine. J Virol 1996;70:1953–1960.
Davis NL, Caley IJ, Brown KW, et al. Vaccination of macaques against pathogenic simian immunode ciency virus with Venezuelan equine encephalitis virus replicon particles. J Virol 2000;74:371–378.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lemckert, A.A., Goudsmit, J. & Barouch, D.h. Challenges in the Search for an HIV Vaccine. Eur J Epidemiol 19, 513–516 (2004). https://doi.org/10.1023/B:EJEP.0000032423.87658.68
Issue Date:
DOI: https://doi.org/10.1023/B:EJEP.0000032423.87658.68