Abstract
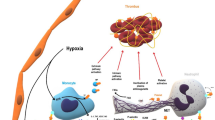
Since the reports by Weismann and Tobin in 1958 and Roberts et al. in 1964 called attention to paradoxical thrombosis in patients treated with heparin, the thrombotic aspect of the heparin-induced thrombocytopenia syndrome (HIT) has been emphasized. Yet to this day, the mechanism of thrombosis associated with HIT (HITT) is unclear. It is important to understand the etiology of HITT because of its devastating clinical consequences. We believe one rational approach to understand the mechanism underlying HITTS is to invoke Virchow's triad: stasis, vascular injury and a hypercoagulable state. A hypercoagulable state exists in all HIT patients due to platelet activation by heparin antibody binding. Thrombin generation from platelet microparticles and exposed platelet phospholipid, coupled with stasis (elderly bedridden or otherwise sedentary ill patients who comprise the majority of the HIT population), provide two risk factors that can lead to venous thrombosis. A hypercoagulable state coupled with endothelial cell dysfunction due to injury from heparin antibody, activated platelets, leukocytes, platelet microparticles, complement, atherosclerosis or medical intervention can lead to arterial thrombosis. Of patients with HIT, HITT occurs in about 25%, suggesting that a second set of patient specific risk factors, in addition to the generation of pathological heparin antibodies, determine whether HITT will develop. Interaction between activated platelets and other platelets, and with endothelial cells, leukocytes, neutrophils, monocytes and cytokines are areas of research that may provide more specific characterization of the hypercoagulable state and vascular damage. Nuances involving genetic variation in platelets, endothelial cells and immune function are also likely to be a major component of the observed variability of this disease spectrum. Virchow's triad may explain the different manifestations of HITTS.
Similar content being viewed by others
References
Weismann RE, Tobin RW. Arterial embolism occurring during systemic heparin therapy. Arch Surg 1958; 76:219–227.
Roberts B, Rosato FE, Rosato EF. Heparin: a cause of arterial emboli? Surgery (St. Louis) 1964;55:803–808.
Wallis DE, Workman DL, Lewis BE, Steen L, Pifarré R, Moran JF. Failure of early heparin cessation as treatment for heparin-induced thrombocytopenia. Am J Med 1999;106:629–635.
Nand S, Wong W, Yuen B, Yetter A, Schmulbach E, Fisher SG. Heparin-induced thrombocytopenia with thrombosis: Incidence, analysis of risk factors, and clinical outcomes in 108 consecutive patients treated at a single institution. Am J Hematol 1997;56:12–16.
Warkentin TE, Kelton JG. A 14-year study of heparin-induced thrombocytopenia. Am J Med 1996;101:502–507.
Messmore HL. Heparin-induced thrombocytopenia: historical review. Clin Appl Thrombosis/Hemostasis 1995;5(Suppl 1):S2-S6.
Kwaan HC, Sakurai S. Endothelial cell hyperplasia contributes to thrombosis in heparin-induced thrombocytopenia. Semin Thromb Hemost 1999;25(Suppl 1):23–27.
Antecol DH, Walley VM, Chan K-L. Massive acute thrombosis of the descending thoracic aorta in heparin-associated thrombocytopeniia and thrombosis. J Am Soc Echocardiogr 1994;7:550–552.
Chong BH. Heparin-induced thrombocytopenia. [Annotation] Br J Haematol 1995;89:431–439.
Virchow. Matzdorff AC, Bell WR, translators. Thrombosis and Emboli (1846–1856). Canton, MA: Science History Publications/USA, 1998.
Amiral J, Bridge F, Dreyfus M, et al. PF4 complexed to heparin is the target for antibodies generated in heparin-induced thrombocytopenia. Thromb Haemost 1992;68:95–96.
Chong BH, Ismail F, Chesterman CN, et al. Heparin-induced thrombocytopenia: mechanism of interaction of the heparin-dependent antibody with platelets. Br J Haematol 1989;73:235–240.
Greinacher A, Pötazsch B, Amiral J, et al. HIT: isolation of the antibody and characterization of a multi-molecular PF4-heparin complex as the major antigen. Thromb Haemost 1994;71:247–251.
Kelton JG, Sheridan DP, Santos AV, et al. Heparin-induced thrombocytopenia: laboratory studies. Blood 1988;72:925–930.
Anderson GP. Insights into heparin-induced thrombocytopenia. Br J Haematol 1992;80:504–508.
Aster RH. Heparin-induced thrombocytopenia: understanding improves but questions remain. J Clin Lab Med 1996;127:418–419.
Horne MK, Alkins BR. Platelet binding of IgG from patients with heparin-induced thrombocytopenia. J Clin Lab Med 1996;127:435–442.
Arepally G, McKenzie SE, Jiang XM, et al. Fc RIIA H/R 131 polymorphism, subclass-specific IgG anti-heparin/platelet factor 4 antibodies and clinical course in patients with HIT. Blood 1997;89:370–375.
Brandt JT, Isenhart CE, Osborne JM, et al. On the role of platelet Fc RIIa a phenotype in heparin-induced thrombocytopenia. Thromb Haemost 1995; 74:1564–1572.
Burgess JK, Lindeman R, Chesterman CN, et al. Single amino acid mutation of Fc gamma receptor is associated with the development of heparin-induced thrombocytopenia. Br J Haematol 1995;91:761–766.
Denomme GA, Warkentin TE, Horsewood P, et al. Activation of platelets by IgG1 heparin-dependent antibodies: explanation for predominance of Fc RIIa ‘low responder’ (His 131) gene in patients with heparin-induced thrombocytopenia. J Lab Clin Med 1997;130:278–284.
Walenga JM, Jeske WP, Wood JJ, Ahmad S, Lewis BE, Bakhos M. Laboratory tests for heparin-induced thrombocytopenia: a multicenter study. Semin Hematol 1993;36(Suppl 1):22–28.
Ahmad S, Jeske W, Walenga JM, Fareed J. Functional anti-heparin-PF4 antibody-mediated platelet activation is associated with intraplatelet Ca2+ signaling. International Society of Hematology, Turk J Haematol 2000;17(Suppl 3):S14.
Jeske W, Walenga JM, Szatkowski E, et al. Effect of glycoprotein IIb/IIIa antagonists on the HIT serum induced activation of platelets. Thromb Res 1997; 88:271–281.
Jeske WP, Jay AM, Haas S, Walenga JM. Heparin-induced thrombocytopenic potential of GAG and non-GAG-based antithrombotic agents. Clin Appl Thrombosis/Hemostasis 1999;5(Suppl 1):S56-S62.
Walenga JM, Jeske WP, Fasanella AR, Wood JJ, Bakhos M. Laboratory tests for the diagnosis of heparin-induced thrombocytopenia. Semin Thromb Hemost 1999;25(Suppl 1):43–49.
Lee DH, Warkentin TE, Denomme GA, et al. A diagnostic test for heparin-induced thrombocytopenia. detection of platelet microparticles using flow cytometry. Br J Haematol 1996;95:724–731.
Tomer A, Masalunga C, Abshire TC. Determination of heparin-induced thrombocytopenia: a rapid flow cytometric assay for direct demonstration of antibody-mediated platelet activation. Am J Hematol 1999; 61:53–61.
Warner MN, Pavord S, Moore JC, Warkentin TE, Hayward CP, Kelton JG. Serum-induced platelet procoagulant activity: An assay for the characterization of prothrombotic disorders. J Lab Clin Med 1999; 133:129–133.
Iwamoto S, Kawasaki T, Kambayashi J, et al. The release mechanism of platelet-activating factor during shear-stress induced platelet aggregation. Biochem Biophys Res Comm 1997;239:101–105.
Piccardoni P, Evangelista V, Piccoli A, de Gaetano G, Walz A, Cerletti C. Thrombin-activated human platelets release two NAP-2 variants that stimulate polymorphonuclear leukocytes. Thromb Haemost 1996;76:780–785.
Jy W, Mao WW, Hortsman L, Tao J, Ahn YS. Platelet microparticles bind, activate and aggregate neutrophils in vitro. Blood Cells, Molecules and Disease 1995;21:217–231.
Siljander P, Carpen O, Lassila R. Platelet-derived microparticles associate with fibrin during thrombosis. Blood 1996;87:4651–4663.
Brown K, Henson PM, Maclouf J, Moyle M, Ely JA, Worthen GS. Neutrophil-platelet adhesion: relative roles of platelet P-selectin and neutrophil β2 (ced18) integrins. Am J Respir Cell Mol Biol 1998;18:100–110.
Jeske WP, Schlenker R, Bakhos M, Walenga JM. Are leukocytes important for platelet activation in heparin-induced. Annal Hematol 2000;79(Suppl 1):A46.
Osterud B. A global view on the role of monocytes and platelets in atherogenesis. Thromb Res 1997;85:1–22.
Neumann FJ, Marx N, Gawaz M, et al. Induction of cytokine expression in leukocytes by binding of thrombin-stimulated platelets. Circulation 1997;95:2387–2394.
Pellegrini G, Malandra R, Celi A, Furie BC, Furie B, Lorenzet R. 12-Hydroxyeicosatetraenoic acid upregulates P-selectin-induced tissue factor activity on monocytes. FEBS Letters 1998;441:463–466.
Yang J, Furie BC, Furie B. The biology of P-selectin glycoprotein ligand-1: its role as a selectin counter-receptor in leukocyte-endothelial and leukocyte-platelet interaction [Review]. Thromb Haemost 1999;81:1–7.
Walenga JM, Michal K, Hoppensteadt D, Wood JJ, Robinson JA, Bick RL. Vascular damage correlates between heparin-induced thrombocytopenia and the antiphospholipid syndrome. Clin Appl Thrombosis/Hemostasis 1999;(Suppl 1):S56-S62.
Warkentin TE, Hayward CPM, Boshkov LK, et al. Sera from patients with heparin-induced thrombocytopenia generate platelet-derived microparticles with procoagulant activity: an explanation for the thrombotic complications of heparin-induced thrombocytopenia. Blood 1994;84:3691–3699.
Nieuwland R, Berckmans RJ, Rotteveel-Eijkman RC, et al. Cell-derived microparticles generated in patients during cardiopulmonary bypass are highly procoagulant. Circulation 1997;96:3534–3541.
Grisaru D, Zwang E, Peyser MR, Lessing JB, Eldor A. The procoagulant activity of red blood cells from patients with severe preeclampsia. Am J Obstet Gyn 1997;177:1513–1516.
Gillis S, Furie BC, Furie B. Interactions of neutrophils and coagulation proteins [Review]. Semin Hematol 1997;34:336–342.
Lewis BE, Wallis DE, Berkowitz S, et al. Argatroban anticoagulant therapy in patients with heparin-induced thrombocytopenia: a prospective, historical controlled study. Circulation submitted 2000.
Greinacher A, Völpel H, Janssens U, et al. Recombinant hirudin (Lepirudin) provides safe and effective anticoagulation in patients with heparin-induced thrombocytopenia. A prospective study. Circulation 1999;99:73–80.
Haas S, Walenga JM, Jeske WP, et al. Heparin-induced thrombocytopenia: the role of platelet activation and therapeutic implications. Semin Thromb Hemost 1999;25(Suppl 1):67–75.
Polgar J, Eichler P, Greinacher A, et al. Adenosine diphosphate (ADP) and ADP receptor play a major role in platelet activation/aggregation induced by sera from HIT patients. Blood 1998;91:549–554.
Walenga JM, Jeske W, Bakhos M, et al. Clinical experience with combined treatment of thrombin inhibitors and GPIIb/IIIa inhibitors in patients with HIT. Semin Thromb Hemost 1999;25(Suppl 1):77–81.
Cines DB, Kaywin P, Bina M, Tomaski A, Schreiber AD. Heparin-associated thrombocytopenia. N Engl J Med 1980;303:788–793.
Cerletti C, Evangelista V, deGaetano G. P-selectin-β2-integrin cross-talk: a molecular mechanism for polymorphonuclear leukocyte recruitment at the site of vascular damage. Thromb Haemost 1999;82:787–793.
Barry OP, FitzGerald GA. Mechanisms of cellular activation by platelet microparticles. Thromb Haemost 1999;82:794–800.
Gawaz M, Neumann F-J, Dickfeld T, et al. Activated platelets induced monocyte chemotactic protein-1 secretion and surface expression of intercellular adhesion molecule-1 on endothelial cells. Circulation 1998;98:1164–1171.
Cines DB, Tomaski A, Tannenbaum S. Immune endothelial-cell injury in HIT. N Engl J Med 1987; 316:581–589.
Herbert JM, Savi P, Jeske WP, et al. Effect of SR121566A, a potent GPIIb/IIIa antagonist on the HIT serum/heparin induced platelet mediated activation of endothelial cells. Thromb Haemost 1998;80:326–331.
Blank M, Shoenfeld Y, Tavor S, Praprotnik S, et al. HIT anti-PF4/heparin antibodies from patients with heparin-induced thrombocytopenia provoke direct activation of microvascular endothelial cells. Blood accepted 2000.
Fareed J, Walenga JM, Hoppensteadt DA, et al. Selectins in the HIT syndrome: pathophysiologic role and therapeutic modulation. Semin Thromb Hemost 1999;25Suppl 1):37–42.
Brandt JT. Platelet aggregation in heparin-induced thrombocytopenia is dependent on complement activation. Blood 1996;88:517a.
Oppenheim JJ, Shevach EM, eds. Immunophysiology The Role of Cells and Cytokines in Immunity and Inflammation. New York: Oxford University Press, 1990.
Lee DH, Warkentin TE, Denomme GA, Lagrotteria DD, Kelton JG. Factor V Leiden and thrombotic complications in heparin-induced thrombocytopenia. Thromb Haemost 1998;79:50–53.
Solum NO. Procoagulant expression in platelets and defects leading to clinical disorders. Arterioscler Thromb Vasc Biol 1999;19:2841–2846.
Pfueller SL, David R. Different platelet specificities of heparin-dependent platelet aggregating factors in heparin-associated immune thrombocytopenia. Br J Haematol 1986;64:149–159.
Boshkov LK, Warkentin TE, Hayward CPM, Andrew M, Kelton JG. Heparin-induced thrombocytopenia and thrombosis: clinical and laboratory studies. Br J Haematol 1993;84:322–328.
Ahmad S, Walenga JM, Jeske WP, Cella G, Fareed J. Functional heterogeneity of antiheparin-platelet factor 4 antibodies: implications in the pathogenesis of the HIT syndrome. Clin Appl Thrombosis/Hemostasis 1999;5(Suppl 1):S32-S37.
Horne MK, Hutchison KJ. Simultaneous binding of heparin and platelet factor-4 to platelets: further insights into the mechanism of heparin-induced thrombocytopenia. Am J Hematol 1998;58:24–30.
Warkentin TE, Levine MN, Hirsh J, et al. Heparin-induced thrombocytopenia in patients treated with low-molecular-weight heparin or unfractionated heparin. N Engl J Med 1995;332:1330–1335.
Hach-Wunderle V, Kainer I, Krug B, et al. Heparin-associated thrombosis despite normal platelet counts. Lancet 1994;344:469.
Marcus AJ, Safier LB, Broekman MJ, et al. Thrombosis and inflammation as multicelluar processes: significance of cell-cell interactions. Thromb Haemost 1995;74:213–217.
Furie B, Furie BC. P-selectin induction of tissue factor biosynthesis and expression [Review]. Haemostasis 1996;26(Suppl 1):60–65.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Walenga, J.M., Jeske, W.P. & Messmore, H.L. Mechanisms of Venous and Arterial Thrombosis in Heparin-Induced Thrombocytopenia. J Thromb Thrombolysis 10 (Suppl 1), 13–20 (2000). https://doi.org/10.1023/A:1027372901367
Issue Date:
DOI: https://doi.org/10.1023/A:1027372901367