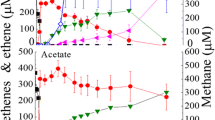
Abstract
A rod shaped, gram positive, non sporulating Acetobacterium strain was isolated that dechlorinated 1,2-dichloroethane (1,2-DCA) to ethene at a dechlorination rate of up to 2 nmol Cl- min-1 mg-1 of protein in the exponential growth phase with formate (40 mM) as the substrate. Although with other growth substrates such as pyruvate, lactate, H2/CO2, and ethanol higher biomass productions were obtained,the dechlorination rate with these substrates was more than 10-fold lower compared with formate growing cells. Neither cell extracts nor autoclaved cells of the isolatedAcetobacterium strain mediated the dechlorination of 1,2-DCA at significant rates. The addition of 1,2-DCA to the media did not result in increased cell production. No significant differences in corrinoid concentrations could be measured in cells growing on several growth-substrates. However, these measurements indicated that differences in corrinoid structure might cause the different dechlorination activity. The Acetobacterium sp. strain gradually lost its dechlorination ability during about 10 transfers in pure culture, probably due to undefined nutritional requirements. 16S rDNA analysis of the isolate revealed a 99.7% similarity with Acetobacterium wieringae. However, the type strains of A. wieringae and A. woodii did not dechlorinate 1,2-DCA.
Similar content being viewed by others
References
Assafanid N, Hayes KF & Vogel TM (1994) Reductive dechlorination of carbon tetrachloride by cobalamin(II) in the presence of dithiothreitol — mechanistic study, effect of redox potential and pH. Environ. Sci. Technol. 28: 246–252
Belay N & Daniels L (1987) Production of ethane, ethylene, and acetylene from halogenated hydrocarbons by methanogenic bacteria. Appl. Environ. Microbiol. 53: 1604–1610
Bouwer EJ & McCarty PL (1983) Transformation of 1-and 2-carbon halogenated aliphatic organic compounds under methanogenic conditions. Appl. Environ. Microbiol. 45: 1286–1294
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utitlizing the principle of protein-dye binding. Anal. Biochem. 72: 248–254
Bradley PM (2000) Microbial degradation of chloroethenes in groundwater systems. Hydrogeol. J. 8: 104–111
Braun M & Gottschalk G (1982) Acetobacterium wieringae sp. nov., a new species producing acetic acid from molecular hydrogen and carbon dioxide. Zentralblatt Fur Bakteriologie Mikrobiologie Und Hygiene 3: 368–376
Christiansen N, Ahring BK, Wohlfarth G & Diekert G (1998) Purification and characterization of the 3-chloro-4-hydroxy-phenylacetate reductive dehalogenase of Desulfitobacterium hafniense. FEBS Lett. 436: 159–162
De Wildeman S, Nollet H, Van Langenhove H, Diekert G & Verstraete W (2002) Reductive biodegradation of 1,2-DCA by methanogenic granular sludge: perspectives for in situ remediation. Wat. Sci. Technol. 45: 43–48
De Wildeman S & Verstraete W (2003) The quest for microbial reductive dechlorination of C2 to C4 chloroalkanes is warranted. Appl. Microbiol. Biotechnol. (in press)
Diekert G (1990) CO2 reduction to acetate in anaerobic bacteria. FEMS Microbiol. Rev. 87: 391–395
Egli C, Scholtz R, Cook AM & Leisinger T (1987) Anaerobic dechlorination of tetrachloromethane and 1,2-dichloroethane to degradable products by pure cultures of Desulfobacterium sp. and Methanobacterium sp. FEMS Microbiol. Lett. 43: 257–261
Glod G, Angst W, Holliger C & Schwarzenbach RP (1997) Corrinoid-mediated reduction of tetrachloroethene, trichloroethene, and trichlorofluoroethene in homogeneous aqueous solution: Reaction kinetics and reaction mechanisms. Environ. Sci. Technol. 31: 253–260
Holliger C, Schraa G, Stams AJ & Zehnder AJ (1990) Reductive dechlorination of 1,2-dichloroethane and chloroethane by cell suspensions of methanogenic bacteria. Biodegradation 1: 253–261
Holliger C, Schraa G, Stupperich E, Stams AJM & Zehnder AJB (1992) Evidence for the involvement of corrinoids and factor F430 in the reductive dechlorination of 1,2-dichloroethane by Methanosarcina barkeri. J. Bacteriol. 174: 4427–4434
Holliger C, Wohlfarth G & Diekert G (1998) Reductive dechlorination in the energy metabolism of anaerobic bacteria. FEMS Microbiol. Rev. 22: 383–398
Magli A, Wendt M & Leisinger T (1996) Isolation and characterization of Dehalobacterium formicoaceticum gen. nov. sp. nov., a strictly anaerobic bacterium utilizing dichloromethane as source of carbon and energy. Arch Microbiol 166: 101–108
Maymo-Gatell X, Chien YT, Gossett JM & Zinder SH (1997) Isolation of a bacterium that reductively dechlorinates tetrachloroethene to ethene. Science 276: 1568–1571
Maymo-Gatell X, Anguish T & Zinder SH (1999) Reductive dechlorination of chlorinated ethenes and 1,2-dichloroethane by “Dehalococcoides ethenogenes” 195. Appl. Environ. Microbiol. 65: 3108–3113
Messmer M, Wohlfarth G & Diekert G (1993) Methylchloride metabolism of the strictly anaerobic, methylchloride utilizing homoacetogen strain Mc. Arch. Microbiol. 160: 383–387
Miller E, Wohlfarth G & Diekert G (1997) Comparative studies on tetrachloroethene reductive dechlorination mediated by Desulfitobacterium sp. strain PCE-S. Arch. Microbiol. 168: 513–519
Miller E, Wohlfarth G & Diekert G (1998) Purification and characterization of the tetrachloroethene reductive dehalogenase of strain PCE-S. Arch. Microbiol. 169: 497–502
Neumann A, Wohlfarth G & Diekert G (1996) Purification and characterization of tetrachloroethene reductive dehalogenase from Dehalospirillum multivorans. J. Biol. Chem. 271: 16515–16519
Neumann A, Siebert A, Trescher T, Reinhardt S, Wohlfarth G & Diekert G (2002) Tetrachloroethene reductive dehalogenase of Dehalospirillum multivorans: substrate specificity of the native enzyme and its corrinoid cofactor. Arch. Microbiol. 177: 420–426
Scholz-Muramatsu H, Neumann A, Messmer M, Moore E & Diekert G (1995) Isolation and characterization of Dehalospirillum multivorans gen. nov., sp. nov., a tetrachloroethene utilizing, strictly anaerobic bacterium. Arch. Microbiol. 163: 48–56
Stupperich E (1993) Recent advances in elucidation of biological corrinoid functions. FEMS Microbiol. Rev. 12: 349–366
Terzenbach DP & Blaut M (1994) Transformation of tetrachloroethylene to trichloroethylene by homoacetogenic bacteria. FEMS Microbiol. Lett. 123: 213–218
Traunecker J, Preuss A & Diekert G (1991) Isolation and characterization of a methylchloride utilizing, strictly anaerobic bacterium. Arch. Microbiol. 156: 416–421
Van Eekert MHA, Stams AJM, Field JA & Schraa G (1999) Gratuitous dechlorination of chloroethanes by methanogenic granular sludge. Appl. Microbiol. Biotechnol. 51: 46–52
Vogel TM, Criddle CS & McCarty PL (1987) Transformations of halogenated aliphatic compounds. Environ. Sci. Technol. 21: 722–736
Wild AP, Winkelbauer W & Leisinger T (1995) Anaerobic dechlorination of trichloroethene, tetrachloroethene and 1,2-dichloroethane by an acetogenic mixed culture in a fixed-bed reactor. Biodegradation 6: 309–318
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
De Wildeman, S., Neumann, A., Diekert, G. et al. Growth-Substrate Dependent Dechlorination of 1,2-Dichloroethane by a Homoacetogenic Bacterium. Biodegradation 14, 241–247 (2003). https://doi.org/10.1023/A:1024781722670
Issue Date:
DOI: https://doi.org/10.1023/A:1024781722670