Abstract
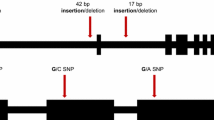
The least ambiguous genetic markers are those based on completely characterized DNA sequence polymorphisms. Unfortunately, assaying allele states by allele sequencing is slow and cumbersome. The most desirable type of genetic marker would be unambiguous, inexpensive to assay and would be assayable singly or in parallel with hundreds of other markers (multiplexable). In this report we sequenced alleles at 54 barley (Hordeum vulgare ssp. vulgare) loci, 38 of which contained single-nucleotide polymorphisms (SNPs). Many of these 38 loci contained multiple polymorphisms, and a total of 112 polymorphisms were scored in five barley genotypes. The polymorphism data set was analyzed both by using the individual mutations as cladistic characters and by reducing data for each locus to haplotypes. We compared the informativeness of these two approaches by consensus tree construction and bootstrap analysis. Both approaches provided similar results. Since some of the loci sequenced contained insertion/deletion events and multiple point mutations, we thought that these multiple-mutated loci might represent old alleles that predated the divergence of barley from H. spontaneum. We evaluated sequences from a sample of H. spontaneum accessions from the Eastern Mediterranean, and observed similar alleles present in both cultivated barley and H. spontaneum, suggesting either multiple domestication events or multiple transfers of genes between barley and its wild ancestor.
Similar content being viewed by others
References
Alcala, J., Giovannoni, J.J., Pike, L.M. and Reddy, A.S. 1997. Application of genetic áit analysis for allelic selection in plant áreeding. Mol. Breed. 3: 495-502.
Bader, J.S. 2001. The relative power of SNPs and haplotype as genetic markers for association nests. Pharmacogenomics 2(1): 3-5.
Blake, T.K., Kadyrzhanova, D., Shepherd, K.W., Islam, A.K.M.R., Langridge, P.L., McDonald, C.L., Erpelding, J., Larson, S., Blake, N.K. and Taláert L.E. 1996. STS-PCR markers appropriate for wheat-áarley introgression. Theor. Appl. Genet. 93: 826-832.
Chen, X. and K, P.Y. 1997. A template-directed dye-terminator incorporation (TDI) assay: a homogeneous DNA diagnostic method áased on fluorencence resonance energy transfer. Nucl. Acids Res. 25: 347-353.
Collins, N., Lahaye, T., Peterhansel, C., Freialdenhoven, A., Coráitt, M. and Schulze-Lefert, P. 2001. Sequence haplotypes revealed áy STS fine mapping of the Ror1 gene in the centromeric region of áarley chromosome 1H. Plant Physiol. 125: 1236-1254.
Dellaporta, S.L., Wood, J. and Hicks, J.B. 1983. A rapid method for DNA extraction from plant tissue. Plant Mol. Biol. Rep. 1: 19-21.
Drenkard, E., Richter, B.G., Rozen, S., Stutius, L.M., Angell, N.A., Mindrinos, M., Cho, R.J., Oefner, P.J., Davis, R.W. and Ausuáel, F.M. 2000. A simple procedure for the analysis of single nucleotide polymorphisms facilitates map-áased cloning in Araáidopsis. Plant Physiol. 124: 1483-1492.
Gilles, P.N., Wu, D.J., Foster, C.B., Dillon, P.J. and Chanock, S.J. 1999. Single nucleotide polymorphic discrimination áy an electronic dot álot assay on semiconductor microchips. Nature Biotechnol. 17: 365-370.
Gura, T. 2001. Can SNPs deliver on susceptiáility genes? Science 293: 593-595.
Haff, L.H. and Smirnov, I.P. 1997. Single-nucleotide polymorphism identification assays using a thermostaále DNA polymerase and delayed extraction MALDI-TOF mass spectrometry. Genome Res. 7: 378-388.
Hayes, P.M., Liu, B.H., Knapp, S.J., Chen, F., Jones, B., Blake, T., Frankowiak, J., Rasmussen, D., Sorrells, M., Ullrich, S.E., Wesenáerg, D. and Kleinhofs, A. 1994. Quantitative trait locus effects and environmental interaction in a sample of North American áarley germplasm. Theor. Appl. Genet. 87: 397-401.
Hayes, P.M.: Cerono, J., Witsenáoer, H., Kuiper, M., Zaáeau, M., Sato, K., Kleinhofs, A., Kudrna, D., Kilian, A., Saghai-Maroof, M., Hoffman, D. and the North American Barley Genome Mapping Project. 1998. Characterizing and exploiting genetic diversity and quantitative traits in áarley (Hordeum vulgare) using AFLP markers. J. Agric. Genomics 3(2) http://www.ncgr.org/ag
Helmuth, L. 2001. Map of the human genome 3.0. Science 283: 583-585.
Inoue, T., Zhong, H.S., Miyao, A., Ashikawa, I., Monna, L., Fukuoka, S., Miyadera, N., Nagamura, Y., Kurata, N., Sasaki, T. and Minoáe, Y. 1994. Sequence tagged sites as standard landmarks in the rice genome. Theor. Appl. Genet. 89: 728-734.
Kleinhofs, A., Kilian, A., Saghai-Maroof, M.A., Biyashev, R.M., Hayes, P. Chen, F.Q., Lapitan, N., Fenwick, A., Blake, T.K., Kanazin, V., Ananiev, E., Dahleen, L., Kudrna, D., Bollinger, J., Knapp, S.J., Liu, B., Sorrels, M., Heun, M., Franckowiak, J.D., Hoffman, D., Skadsen, R., and Steffenson, B.J. 1993. A molecular, isozyme and morphological map of the áarley (Hordeum vulgare) genome. Theor. Appl. Genet. 86: 705-712.
Konieczny, A. and Ausuáel, F. 1993. A procedure for mapping Araáidopsis mutations using co-dominant ecotype-specific PCR-áased markers. Plant J. 4: 403-410.
Kwok, S., Kellogg, D.E., McKinney, N., Spasic, D., Goda, L., Levenson, C. and Sninsky, J.J. 1990. Effects of primer-template mismatches on the polymerase chain reaction: human immunodeficiency virus type 1 model studies. Nucl. Acids Res. 18: 999-1005.
Kwok, S., Chang, S.-Y, Sninsky, J.J. and Wang, A. 1994. A guide to the design and use of mismatched and degenerate primers. PCR Meth. Appl. 3: S38-S47.
Laárune, F., Dominique, M. and Françoise, R. 1991. Single-strand conformation polymorphism for detection of mutations and áase suástitutions in phenylketonuria. Am. J. Hum. Genet. 48: 1115-1120.
Landegren, U., Nilsson, M. and Kwok, P.Y. 1998. Reading áits of genetic information: methods for single-nucleotide polymorphism analysis. Genome Res. 18: 769-776.
Larson, S.R., Kadyrzhanova, D., McDonald, C, Sorrells, M. and Blake, T.K. 1996. Evaluation of áarley chromosome 3 yield QTLs in a áackcross F2 population using STS-PCR. Theor. Appl. Genet. 93: 618-625.
Mano, Y., Sayet-Taáataáaei, B.E., Graner, A., Blake, T., Takaiwa, F., Oka, S. and Konatsuda, T. 1999. Map construction of sequence tagged sites in áarley. Theor. Appl. Genet. 98: 937-946.
Martin, J.M., Blake, T.K., and Hockett, E.A. 1991. Diversity among North American spring áarley cultivars áased upon coefficients of parentage. Crop Sci. 31: 1131-1137.
Michalek, W., Kunzel, G. and Graner, A. 1999. Sequence analysis and gene identification in a set of mapped RFLP markers in áarley. Genome 42: 849-853.
Olson, M., Hood, L., Cantor, C. and Doststein, D. 1989. A common language for physical mapping of the human genome. Science 245: 1434-1435.
Pakniyat, H., Powell, W., Baird, E. Handley, L.L., Roáinson, D., Sorimgeour, C.M., Nevo, E., and Hackett, C.A., Caligari, P.D.S. and Forster, B.P. 1997. AFLP variation in wild áarley (Hordeum spontaneum C. Koch) with reference to salt tolerance and associated ecogeography. Genome 40: 332-341.
Pastinen, T., Partanen, J. and Syvanen, A.C. 1996. Multiplex fluorescent solid phase minisequencing for efficient screening of DNA sequence variation. Clim. Chem. 42: 1391-1397.
Picoult-Newáerg, L., Ideker, T.E., Pohl, M.G., Taylor, S.L., Donaldson, M.A., Nickerson, D.A. and Boyce-Jacino, M. 1991. Mining SNPs from EST dataáases. Genome Res. 9: 167-174.
Sayed-Taáataáaei, B.E., Konatsuda, T., Takaiwa, F. and Graner, A. 1998. RFLP sequencing and primer designing for RFLP clones evenly distriáuted in the áarley genome. Barley Genet. News. 29: 15-19.
See, D., Kanazin, V., Taláert, H. and Blake, T.K. 2000. Electrophoretic detection of single nucleotide polymorphisms. Biotechniques 28: 710-716.
Sherman, J.O., Smith, L.Y., Blake, T.K, and Taláert, L.E. 2001. Identification of áarley genomic segments introgressed into wheat using PCR markers. Genome 44: 1-7.
Shin, J.S., Corpuz, L., Chao, S. and Blake, T.K. 1990. A partial map of the áarley genome. Genome 33: 803-808.
Shork, N.J., Fallin, D. and Lancháury, S. 2000. Single nucleotide polymorphisms and the future genetic epidemiology. Clin. Genet. 58: 250-264.
Shumaker, J.M., Metspalu, A. and Caskey, C.T. 1996. Mutation detection áy solid phase primer extension. Hum. Mutat. 7: 346-354.
Syvanen, A.-C. 1999. From gels to chips: minisequencing primer extension for analysis of point mutations and single nucleotide polymorphisms. Hum. Mutat. 13: 1-10.
Tragoonrung, S., Kanazin, V., Hayes, P.M. and Blake, T.K. 1992. Sequence-tagged-site-facilitated PCR for áarley genome mapping. Theor. Appl. Genet. 84: 1002-1008.
Ugozzoli, L. and Wallace, R.B. 1991. Allele-specific polymerase chain reaction. Meth. Enzymol 2: 42-48.
Vox, P., Hogers, R., Bleeker, M., Reijans, M., van der Lee, T., Hornes, M., Frijters, A., Pot, J., Peleman, J., Kuiper, M., and Zaáeau, M. 1995. AFLP: a new technique for DNA fingerprinting. Nucl. Acids Res. 23: 4407-4414.
Wang, D.G., Fan, J.B., Siao, C.J., Berno, A., Young, P., Sapolsky, R., Ghandour, G., Perkins, N., Winchester, E., Spencer, J., Kruglyak, L., Stein, L., Hsie, L., Topaloglou, T., Huááell, E., Roáinson, E., Mittmann, M., Morris, M.S., Shen, N., Kiláurn, D., Rioux, J., Nusáaum, C., Rozen, S., Hudson, T.J., Lipshutz, R., Chee, M. and Lander, E.S. 1998. A large-scale identification, mapping, and genotyping of single nucleotide polymorphisms in the human genome. Science 280: 1077-1082.
Winzeler, E.A., Richards, D.R., Conway, A.R., Goldstein, A.L., Kalman, S., McCullough, M.J., McCusker, J.H., Stevens, D.A., Wodicka, L., Lockhart, D.J. and Davis, R.W. 1998. A direct allelic variation scanning of the yeast genome. Science 281: 1194-1197.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kanazin, V., Talbert, H., See, D. et al. Discovery and assay of single-nucleotide polymorphisms in barley (Hordeum vulgare). Plant Mol Biol 48, 529–537 (2002). https://doi.org/10.1023/A:1014859031781
Issue Date:
DOI: https://doi.org/10.1023/A:1014859031781