Abstract
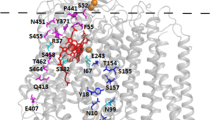
Succinate:quinone reductase catalyzes electron transfer from succinate to quinone in aerobic respiration. Carboxin is a specific inhibitor of this enzyme from several different organisms. We have isolated mutant strains of the bacterium Paracoccus denitrificans that are resistant to carboxin due to mutations in the succinate:quinone reductase. The mutations identify two amino acid residues, His228 in SdhB and Asp89 in SdhD, that most likely constitute part of a carboxin-binding site. This site is in the same region of the enzyme as the proposed active site for ubiquinone reduction. From the combined mutant data and structural information derived from Escherichia coli and Wolinella succinogenes quinol:fumarate reductase, we suggest that carboxin acts by blocking binding of ubiquinone to the active site. The block would be either by direct exclusion of ubiquinone from the active site or by occlusion of a pore that leads to the active site.
Similar content being viewed by others
REFERENCES
Ackrell, B. A. C. (2000). FEBS Lett. 466, 1–5.
Ackrell, B. A. C., Kearney, E. B., Coles, C. J., Singer, T. P., Beinert, H., Wan, Y. P., and Folkers, K. (1977). Arch. Biochem. Biophys. 182, 107–117.
Ackrell, B. A. C., Johnson, M. K., Gunsalus, R. P., and Cecchini, G. (1992). In Chemistry and Biochemistry of Flavoenzymes III: Structure and Function of Succinate Dehydrogenase and Fumarate Reductase (Müller, F., ed.), CRC Press, Boca Raton, FL, pp. 229–297.
Broomfield, P. L. E. and Hargreaves, J. A. (1992). Curr. Genet. 22, 117–121.
Coles, C. J., Singer, T. P., White, G. A., and Thorn, G. D. (1978). J. Biol. Chem. 253, 5573–5578.
de Vries, G. E., Harms, N., Hoogendijk, J., and Stouthamer, A. H. (1989). Arch. Microbiol. 152, 52–57.
Gerhus, E., Steinrücke, P., and Ludwig, B. (1990). J. Bacteriol. 172, 2392–2400.
Grigolava, I. V., Konstantinov, A. A., Ksenzenko, M. Y., Ruuge, É. K., and Tikhonov, A. N. (1982). Biokhimiia 47, 1970–1982.
Grivennikova, V. G. and Vinogradov, A. D. (1985). Biokhimiia 50, 375–383.
Hägerhäll, C. (1997). Biochim. Biophys. Acta 1320, 107–141.
Hägerhäll, C. and Hederstedt, L. (1996). FEBS Lett. 389, 25–31.
Iverson, T. M., Luna-Chavez, C., Cecchini, G., and Rees, D. C. (1999). Science 284, 1961–1966.
Keon, J. P. R., White, G. A., and Hargreaves, J. A. (1991). Curr. Genet. 19, 475–481.
Lancaster, C. R. D. and Kröger, A. (2000). Biochim. Biophys. Acta 1459, 422–431.
Lancaster, C. R. D., Kröger, A., Auer, M., and Michel, H. (1999). Nature London 402, 377–385.
Matsson, M., Ackrell, B. A. C., Cochran, B., and Hederstedt, L. (1998). Arch. Microbiol. 170, 27–37.
Ohnishi, T., Moser, C. C., Page, C. C., Dutton, P. L., and Yano, T. (2000). Structure 8, 23–32.
Ramsay, R. R., Ackrell, B. A. C., Coles, C. J., Singer, T. P., White, G. A., and Thorn, G. D. (1981). Proc. Natl. Acad. Sci. USA 78, 825–828.
Schirawski, J. and Unden, G. (1998). Eur. J. Biochem. 257, 210–215.
Shenoy, S. K., Yu, L., and Yu, C. A. (1999). J. Biol. Chem. 274, 8717–8722.
Simon, R., Priefer, U., and Puhler, A. (1983). Biotechnology 1, 784–791.
Skinner,W., Bailey, A., Renwick, A., Keon, J., Gurr, S., and Hargreaves, J. (1998). Curr. Genet. 34, 393–398.
Vibat, C. R. T., Cecchini, G., Nakamura, K., Kita, K., and Gennis, R. B. (1998). Biochemistry 37, 4148–4159.
Rights and permissions
About this article
Cite this article
Matsson, M., Hederstedt, L. The Carboxin-Binding Site on Paracoccus denitrificans Succinate:Quinone Reductase Identified by Mutations. J Bioenerg Biomembr 33, 99–105 (2001). https://doi.org/10.1023/A:1010744330092
Issue Date:
DOI: https://doi.org/10.1023/A:1010744330092