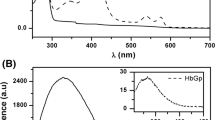
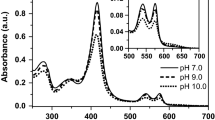
Abstract
The giant, ∼3.6-MDa hexagonal bilayer hemoglobin (Hb) of Lumbricus terrestris consist of twelve 213-kDa globin subassemblies, each comprised of three disulfide-bonded trimers and three monomer globin chains, tethered to a central scaffolding of 36–42 linkers L1–L4 (24–32 kDa). It is known to contain 50–80 Ca and 2–4 Cu and Zn; the latter are thought to be responsible for the superoxide dismutase activity of the Hb. Total reflection X-ray fluorescence spectrometry was used to determine the Ca, Cu, and Zn contents of the Hb dissociated at pH ∼2.2, the globin dodecamer subassembly, and linker subunits L2 and L4. Although the dissociated Hb retained 20 Ca2+ and all the Cu and Zn, the globin subassembly had 0.4 to ∼3 Ca2+, depending on the method of isolation, and only traces of Cu and Zn. The linkers L2 and L4, isolated by reversed-phase high-pressure liquid chromatography at pH ∼2.2, had 1 Ca per mole and very little Cu and Zn. Electrospray ionization mass spectrometry of linker L3 at pH ∼2.2 and at neutral pH demonstrated avid binding of 1 Ca2+ and additional weaker binding of 7 Ca2+ in the presence of added Ca2+. Based on these and previous results which document the heterogeneous nature of the Ca2+-binding sites in Lumbricus Hb, we propose three classes of Ca2+-binding sites with affinities increasing in the following order: (i) a large number of sites (>100) with affinities lower than EDTA associated with linker L3 and dodecamer subassembly, (ii) ∼30 sites with affinities higher than EDTA occurring within the cysteine-rich domains of linker L3 and dodecamer subassembly, and (iii) ∼25 very high affinity sites associated with the linker subunits L1, L2, and L4. It is likely that the low-affinity type (i) sites are the ones involved in the effects of 1–100 mM Group IIA cations on Lumbricus Hb structure and function, namely increased stability of its quaternary structure and increased affinity and cooperativity of its oxygen binding.
Similar content being viewed by others
REFERENCES
Antonini, A., and Chiancone, E. (1977). Annu. Rev. Biophys. Bioeng. 6, 239–271.
Blacklow, S., and Kim, P. S. (1996). Nature Struct. Biol. 3, 758–761.
Chiancone, E., Bull, T. E., Norne, J. E., Forsen, S., and Antonini, E. (1976). J. Mol. Biol. 107, 25–34.
Chiancone, E., Ferruzzi, G., Bonaventura, C., and Bonaventura, J. (1980). Biochim. Biophys. Acta 670, 84–92.
Chottard, G., El Ajouz, N., and Hervé, G (1992). Biochim. Biophys. Acta 1122, 113–117.
Chung, M. C. C., and Ellerton, H. D. (1979). Progr. Biophys. Mol. Biol. 35, 51–102.
De Haas, F., Taveau, J.-C., Boisset, N., Lambert, O., Vinogradov, S. N., and Lamy, J. N. (1996a). J. Mol. Biol. 255, 140–163.
De Haas, F., Taveau, J.-C., Boisset, N., Lambert, O., Vinogradov, S. N., and Lamy, J. N. (1996b). Biophys. J. 70, 1973–1984.
De Haas, F., Zal, F., Lallier, F. H., Toulmond, A., and Lamy, J. N. (1996c). Proteins Struct. Funct. Genet. 26, 241–256.
De Haas, F., Zal, F., You, V., Lallier, F., Toulmond, A., and Lamy, J. N. (1996d). J. Mol. Biol. 264, 111–120.
De Haas, F., Kuchumov, A., Taveau, J.-C., Boisset, N., Vinogradov, S. N., and Lamy, J. N. (1997). Biochemistry 36, 7330–7338.
Fass, D., Blacklow, S., Kim, P. S., and Berger, J. M. (1997). Nature 388, 691–693.
Fenn, J. B., Mann, M., Meng, C. K., Wong, S. F., and Whitehouse, C. M. (1980). Science 246, 64–71.
Fushitani, K., Higashiyama, K., Asao, M., and Hosokawa, K. (1996). Biochim. Biophys. Acta 1292, 273–280.
Gotoh, T., and Suzuki, T. (1990). Zool. Sci. 7, 1–16.
Green, B. G., Bordoli, R. S., Hanin, L. G., Lallier, F. H., Toulmond, A., and Vinogradov, S. N. (1999). J. Biol. Chem. 274, 28206–2812.
Harrington, J. P. (1994). Comp. Biochem. Physiol. 109A, 799–802.
Krebs, A., Kuchumov, A. R., Shaarma, P. K., Braswell, E. H., Zipper, P., Weber, R. E., Chottard, G., and Vinogradov, S. N. (1996). J. Biol. Chem. 271, 18695–18704.
Kuchumov, A. R., Taveau, J.-C., Lamy, J. N., Wall, J. S., Weber, R. E. and Vinogradov, S. N. (1999). J. Mol. Biol. 289, 1361–1374.
Lamy, J. N., Green, B. N., Toulmond, A., Walls, J. S., Weber, R. E., and Vinogradov, S. N. (1996). Chem. Rev. 96, 3113–3124.
Liochev, S. I., Kuchumov, A. R., Vinogradov, S. N., and Fridovich, I. (1996). Arch. Biochem. Biophys. 330, 281–284.
Loo, J. A., and Pesch, R. (1994). Anal. Chem. 66, 3659–3663.
Loo, J. A., Edmunds, C. G., and Smith, R. D. (1993). Anal. Chem. 65, 4225–4238.
Mainwaring, M. G., Lugo, S. D., Fingal, R. A., Kapp, O. H., and Vinogradov, S. N. (1986). J. Biol. Chem. 261, 10899–10908.
Martin, P. D., Eisele, K. L., Doyle, M. A., Kuchumov, A. R., Walz, D. A., Arutyunyan, E. G., Vinogradov, S. N., and Edwards, B. F. P. (1996). J. Mol. Biol. 255, 170–175.
Ochiai, T. (1984). Arch. Biochem. Biophys. 231, 136–143.
Ochiai, T., Hoshina, S., and Usuki, I. (1993). Biochim. Biophys. Acta 1203, 310–314.
Ownby, D. W., Zhu, H., Schneider, K., Beavis, R. C., Chait, B. T., and Riggs, A. F. (1993). J. Biol. Chem. 268, 13539–13547.
Perutz, M. (1990). Mechanisms of Cooperativity and Allosteric Regulation in Proteins, Cambridge University Press, Cambridge.
Polidori, G., Maionwaring, M. G., Kosinski, T., Schwarz, C., Fingal, R. A., and Vinogradov, S. N. (1984). Arch. Biochem. Biophys. 233, 800–814.
Polidori, G., Mainwaring, M. G., and Vinogradov, S. N. (1988). Comp. Biochem. Physiol. 89A, 541–545.
Rokosz, M. J., and Vinogradov, S. N. (1982). Biochim. Biophys. Acta 707, 291–293.
Royer, W. E., Jr., Pardanani, A., Gibson, Q. E., Peterson, E. S., and Friedman, J. M. (1996). Proc. Natl. Acad. Sci. USA 93, 14526–14531.
Schatz, M., Orlova, E. V., Dube, P., Jäger, J., and Van Heel, M. (1995). J. Struct. Biol. 114, 28–40.
Senko, M. W., and McLafferty, F. W. (1994). Annu. Rev. Biophys. Biomol. Struct. 23, 763–785.
Sharma, P. K., Kuchumov, A., Chottard, G., Martin, P. D., Wall, J. S., and Vinogradov, S. N. (1996). J. Biol. Chem. 271, 8754–8762.
Smith, R. M., and Martell, A. E. (1989). Critical Stability Constants, Vol. 6, Plenum Press, New York, p. 96.
Standley, P. M., Mainwaring, M. G., Gotoh, T., and Vinogradov, S. N. (1988). Biochem. J. 249, 915–916.
Suzuki, T., and Riggs, A. F. (1993). J. Biol. Chem. 268, 1354–1361.
Suzuki, T., Takagi, T., and Ohta, S. (1990a). J. Biol. Chem. 265, 1551–1555.
Suzuki, T., Takagi, T., and Gotoh, T. (1990b). J. Biol. Chem. 265, 12168–12177.
Suzuki, T., Ohta, T., Yasa, H. J., and Takagi, T. (1994). Biochim. Biophys. Acta 1217, 291–296.
Taveau, J.-C., Boisset, N, Vinogradov, S. N., and Lamy, J. N. (1999). J. Mol. Biol. 289, 1343–1349.
Tsuneshige, A., Imai, K., Hori, H., Tyuma, I., and Gotoh, T. (1989). J. Biochem. 106, 406–417.
Vinogradov, S. N., Shlom, J. M., Hall, B. C., Kapp, O. H., and Mizukami, H. (1977). Biochim. Biophys. Acta 492, 136–155.
Vinogradov, S. N. (1985). In Respiratory Pigments in Animals (Lamy, J., Truchot, J. P., and Gilles, R., eds.), Springer-Verlag, Berlin. pp. 9–20.
Vinogradov, S. N., and Sharma, P. K. (1994). Meth. Enzmol. 231, 112–124.
Vinogradov, S. N., Kapp, O. H., and Ohtsuki, M. (1982). Electron Microscopy of Proteins, Vol. 3 (Harris, J., ed.), Academic Press, New York. p. 135.
Vinogradov, S. N., Standley, P. R., Mainwaring, M. G., Kapp, O. H., and Crewe, A. V. (1985). Biochim. Biophys. Acta 828, 43–50.
Vinogradov, S. N., Lugo, S. D., Mainwaring, M. G., Kapp, O. H., and Crewe, A. V. (1986). Proc. Natl. Acad. Sci. USA 83, 8034–8038.
Vinogradov, S. N., Sharma, P. K., Qabar, A. N., Wall, J. S., Westrick, J. A., Simmons, J. H., and Gill, S. J. (1991). J. Biol. Chem. 266, 13091–13096.
Vinogradov, S. N., Walz, D. A., Pohajdak, B., Moens, L., Kapp, O. H., Suzuki, T., and Trotman, C. N. A. (1993). Comp. Biochem. Physiol. 106B, 1–26.
Weber, R. E., and Vinogradov, S. N. (2000). Physiol. Rev. (in press).
Zhu, H., Hargrove, M., Xie, Q., Nozaki, Y., Linse, K., Smith, S. S., Olson, J. S., and Riggs, A. F. (1996a). J. Biol. Chem. 271, 29999–30006.
Zhu, H., Ownby, D. W., Riggs, C. K., Nolasco, N. J., Stoops, J. K., and Riggs, A. F. (1996b). J. Biol. Chem. 271, 30007–30021.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kuchumov, A.R., Loo, J.A. & Vinogradov, S.N. Subunit Distribution of Calcium-Binding Sites in Lumbricus Terrestris Hemoglobin. J Protein Chem 19, 139–149 (2000). https://doi.org/10.1023/A:1007086717412
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1007086717412