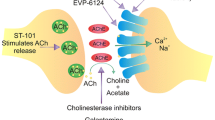
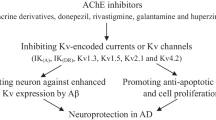
Summary
The only prescribed drugs for treatment of Alzheimer’s disease (AD) are acetylcholinesterase inhibitors (e.g., donepezil, rivastigmine, galantamine, and tacrine) and memantine, an NMDA antagonist. These drugs ameliorate mainly the symptoms of AD, such as cognitive impairments, rather than halting or preventing the causal neuropathology. There is currently no cure for AD and there is no way to stop its progression, yet there are numerous therapeutic approaches directed against various pathological hallmarks of AD that are extensively being pursued. In this context, the three major hallmark characteristics of AD (i.e., the CNS cholinergic hypofunction, formation of β-amyloid plaques, and tangles containing hyperphosphorylated tau proteins) are apparently linked. Such linkages may have therapeutic implications, and this review is an attempt to analyze these versus the advantages and drawbacks of some cholinergic compounds, such as acetylcholinesterase inhibitors, M1 muscarinic agonists, M2 antagonists, and nicotinic agonists. Among the reviewed treatments, M1 selective agonists emerge, in particular, as potential disease modifiers.
Article PDF
Similar content being viewed by others
References
Blennow K, deLeon MJ, Zetterberg H. Alzheimer’s disease. Lancet. 2006;368: 387–403.
Cuello CA. Overview of the Alzheimer’s disease pathology and potential therapeutic targets. In: Pharmacological mechanisms in Alzheimer’s therapeutic. Cuello CA, ed. New York: Springer, 2008, 1–27.
Wolfe MS, Therapeutic strategies for Alzheimer’s disease. Nat Rev Drug Discov 2002;l: 859–866.
Hooper NM, Turner AJ. The search for alpha-secretase and its potential as a therapeutic approach to Alzheimer s disease. Current Med Chem 2002;9: 1107–1119.
Racchi M, Govoni S. The pharmacology of amyloid precursor protein processing. Exp Gerontol 2003;38: 145–157.
Gandy SE, Petanceska S. Regulation of Alzheimer’s β-amyloid precursor trafficking and metabolism. Biochim Biophys Acta 2000;1502: 44–52.
Palmer AM. Pharmacotherapy for Alzheimer’s disease: progress and prospects. Trends Pharmacol Sci 2002;23: 426–433.
Hardy JA, Higgins GA. Alzheimer’s disease: the amyloid cascade hypothesis. Science 1992;256: 184–185.
Tanzi RE, Bertram L. Twenty years of the Alzheimer’s disease amyloid hypothesis: a genetic perspective. Cell 2005; 120: 545–555.
Robinson SR, Bishop GM. Abeta as a bioflocculant: implications for the amyloid hypothesis of Alzheimer’s disease. Neurobiol Aging 2002;23: 1051–1072.
Rottkamp CA, Atwood CS, Joseph JA. The state versus amyloid-beta: the trial of the most wanted criminal in Alzheimer disease. Peptides 2002;23: 1333–1341.
Salloway S, Mintzer J, Weiner MF, Cummings JL. Disease-modifying therapies in Alzheimer’s disease. Alzheimers Dement 2008;4: 65–79.
Birks J. 2006. Cholinesterase inhibitors for Alzheimer’s disease. Cochrane Database Syst Rev CD005593.
Ringman JM, Cummings JL. Current and emerging pharmacological treatment options for dementia. Behav Neurol 2006;17: 5–15.
Bartus RT. On neurodegenerative diseases, models, and treatment strategies: lessons learned and lessons forgotten a generation following the cholinergic hypothesis. Exp Neurol 2000; 163: 495–529.
Geula C, Nagykery N, Nicholas A, Wu CK. Cholinergic neuronal and axonal abnormalities are present early in aging and in Alzheimer disease. J Neuropathol Exp Neurol 2008;67: 309–318.
Ladner CJ, Lee JM. Pharmacological drug treatment of Alzheimer disease: the cholinergic system revisited. J Neuropathol Exptl Neurol 1998;57: 719–731.
Perry EK, Court J, Goodchild R, et al. Clinical neurochemistry: developments in dementia research based on brain bank material. J Neural Transm 1998;105: 915–933.
Iqbal K, Alonso A del C, Gondal JA, et al. Mechanism of neurofibrillary degeneration and pharmacologic therapeutic approach. J Neural Transm 2000;59: 213–222.
Seabrook GR, Ray WJ, Shearman M, Hutton M. Beyond amyloid: the next generation of Alzheimer’s disease therapeutics. Mol Interv 2007;7: 261–270.
Mudher A, Lovestone S. Alzheimer’s disease-do tauists and baptists finally shake hands? Trends Neurosci 2002;25: 22–26.
Fisher A. Therapeutic strategies in Alzheimer’s disease: M1 muscarinic agonists. Jap J Pharmacol 2000;84: 101–112.
Fisher A. Muscarinic agonists and antagonists-some therapeutic applications. In: Giacobini E and Pepeu G, eds. The Brain Cholinergic System in Health and Disease. Boca Raton (FL): Informa Healthcare, Taylor & Francis Publishers 2005;169-180.
Auld DS, Kornecook TJ, Bastianetto S, Quirion R. Alzheimer’s disease and the basal forebrain cholinergic system: relations to beta-amyloid peptides, cognition, and treatment strategies. Prog Neurobiol 2002;68: 209–245.
Rubio A, Perez M, Avila J. Acetylcholine receptors and tau phosphorylation. Curr Mol Med 2006;6: 423–428.
Caccamo A, Oddo S, Billings LM, Martinez-Coria H, Fisher A, LaFerla FM. M1 receptors play a central role in modulating AD-like pathology in transgenic mice. Neuron 2006;49: 671–682.
Standridge JB. Pharmacotherapeutic approaches to the treatment of Alzheimer’s disease. Curr Ther 2004;26: 615–630.
Giacobini E. Do cholinesterase inhibitors have disease-modifying effects in Alzheimer’s disease? CNS Drugs 2001;15: 85–91.
Gauthier S. Cholinergic adverse effects of cholinesterase inhibitors in Alzheimer’s disease: epidemiology and management. Drugs & Aging 2001;18: 853–862.
Schneider LS. Treatment of Alzheimer’s disease with cholinesterase inhibitors. Clin Geriat Med 2001;17: 337–358.
Courtney C, Farrell D, Gray R, et al. Long-term donepezil treatment in 565 patients with Alzheimer’s disease (AD2000): randomised double-blind trial. Lancet 2004;363(9427): 2105–2115.
Lilienfeld S. Galantamine—a novel cholinergic drug with a unique dual mode of action for the treatment of patients with Alzheimer’s disease. CNS Drug Rev 2002;8: 159–176.
Raschetti R, Albanese E, Vanacore N, Maggini M. Cholinesterase inhibitors in mild cognitive impairment: a systematic review of randomised trials. PLoS Med 2007;4(11): e338.
Lahiri DK, Farlow MR, Greig NH, et al. Current drug targets for Alzheimer’s disease treatment. Drug Develop Res 2002;56: 267–281.
Kadir A, Andreasen N, Almkvist O, et al. Effect of phenserine treatment on brain functional activity and amyloid in Alzheimer’s disease. Ann Neurol 2008;[Epub ahead of print].
Trabace L, Cassano T, Loverre A, et al. CHF2819: pharmacological profile of a novel acetylcholinesterase inhibitor. CNS Drug Reviews 2002;8: 53–69.
Ishihara Y, Goto G, Miyamoto M. Central selective acetylcholinesterase inhibitor with neurotrophic activity: structure-activity relationships of TAK-147 and related compounds. Current Med Chem 2000;7: 341–354.
Weinstock M, Luques L, Bejar C, Shoham S. Ladostigil. A novel multifunctional drug for the treatment of dementia co-morbid with depression. J Neural Transm Suppl 2006;70: 443–446.
Bai DL, Tang XC, He XC, Huperzine A. A potential therapeutic agent for treatment of Alzheimer’s disease. CNS Drug Rev 2000; 7: 355–374.
García-Palomero E, Muñoz P, Usan P, et al. Potent beta-amyloid modulators. Neurodegener Dis 2008;5: 153–156.
Bolognesi ML, Banzi R, Bartolini M, et al. Novel class of quinone-bearing polyamines as multi-target-directed ligands to combat Alzheimer’s disease. J Med Chem 2007;50: 4882–4897.
Giacobini E. Selective inhibitors of butyrylcholinesterase—a valid alternative for therapy of Alzheimer’s disease? Drugs Aging 2001;18: 891–898.
Levey AI. Muscarinic acetylcholine receptor expression in memory circuits: implications for treatment of Alzheimer disease. Proc Natl Acad Sci USA 1996;93: 13541–13546.
Volpicelli LA, Levey AI. Muscarinic acetylcholine receptor subtypes in the cerebral cortex and hippocampus. Prog Brain Res 2004;145: 59–66.
Svensson AL, Alafuzof I, Nordberg A. Characterization of muscarinic receptor subtypes in Alzheimer and control brain cortices by selective muscarinic antagonists. Brain Res 1992;596: 142–148.
Mulugeta E, Karlsson E, Islam A, et al. Loss of muscarinic M(4) receptors in hippocampus of Alzheimer patients. Brain Res 2003; 960: 259–262.
Anagnostaras SG, Murphy GG, Hamilton SE, et al. Selective cognitive dysfunction in acetylcholine Ml muscarinic receptor mutant mice. Nature Neurosci 2003;6: 51–58.
Beach TG. Muscarinic agonists as preventative therapy for Alzheimer’s disease. Curr Opin Invest Drugs 2002;3: 1633–1636.
Fox R, Kontinnen Y, Fisher A. Use of muscarinic agonists in the treatment of Sjogren’s syndrome. Clin Immunol 2002;101: 249–263.
Alfa Cisse M, Sunyach C, Slack BE, Fisher A, Vincent B and Checler F. M1 and M3 muscarinic receptors control physiological processing of cellular prion by modulating ADAM17 phosphorylation and activity. J Neurosci 2007;27: 4083–4092.
Wienrich M, Ceci A, Ensinger HA, et al. Talsaclidine (WAL 2014 FU): a muscarinic M1 receptor agonist for the treatment of Alzheimer’s disease. Drug Develop Res 2002;56: 321–334.
Wanibuchi F, Konishi T, Harada M, et al. Pharmacological studies on novel muscarinic agonists, l-oxa-8-azaspiro[4.5]decane derivatives YM796 and YM954. Eur J Pharmacol 1990;187: 479–486.
Tecle H, Schwarz RD, et al. CI-1017, a functionally M1-selective muscarinic agonist: design, synthesis, and preclinical pharmacology. Pharm Acta Helv 2000;74: 141–148.
Bartolomeo AC, Morris H, Buccafusco JJ, et al. The preclinical pharmacological profile of WAY-132983, a potent M1 preferring agonist. J Pharmacol Exp Ther 2000;292: 584–596.
Iwata N, Kozuka M, Hara T, et al. Activation of cerebral function by CS-932, a functionally selective M1 partial agonist: neuro-chemical characterization and pharmacological studies. Jpn J Pharmacol 2000;84: 266–280.
Hitchcock S, Allen J, Baker A, et al. Discovery of a novel class of selective M1 agonists suitable for clinical development. Abstracts of papers, 225th ACS National Meeting, New Orleans, LA, March 23–27, 2003; Stanley PK. Development of a scalable process for the synthesis of LY593093, www.albmolecular.com/ Chemlinks/reviews/vo108/no21/v08n21.pdf.
Spalding TA, Ma JN, Ott TR, et al. Structural requirements of transmembrane domain 3 for activation by the ml muscarinic receptor agonists AC-42, AC-260584, clozapine, and N-desmethylclozapine: evidence for three distinct modes of receptor activation. Mol Pharmacol 2006;70: 1974–1983.
Sur C, Mallorga PJ, Wittmann M, et al. N-desmethylclozapine, an allosteric agonist at muscarinic 1 receptor, potentiates N-methyl-D-aspartate receptor activity. Proc Natl Acad Sci U S A 2003; 100: 13674–13679.
May LT, Avlani VA, Langmead CJ, et al. Structure-function studies of allosteric agonism at M2 muscarinic acetylcholine receptors. Mol Pharmacol 2007;72: 463–476; Langmead.CJ. www.monash.edu.au/cmo/molecular/Program.pdf.
Feuerstein TJ, Lehman J, Suerman W, et al. The autoinhibitory feedback control of acetylcholine release in human neocortex tisue. Brain Res 1992;572: 64–71.
Wess J, Eglen RM, Gautam D. Muscarinic acetylcholine receptors: mutant mice provide new insights for drug development. Nat Rev Drug Discov 2007;6: 721–733.
Clader JW, Wang Y. Muscarinic receptor agonists and antagonists in the treatment of Alzheimer’s disease. Curr Pharm Des 2005;11: 3353–3361.
Boyle CD, Lachowicz JE. Orally active and selective benzylidene ketal M2 muscarinic receptor antagonists for the treatment of Alzheimer’s disease. Drug Dev Res 2002;56: 310–320.
Mash DC, Flynn DD, Potter LT. Loss of M2 muscarine receptors in cerebral cortex in Alzheimer’s disease and experimental cholinergic denervation. Science 1985;228: 1115–1117.
Sheardown MJ. Muscarinic M1 receptor agonists and M2 receptor antagonists as therapeutic targets in Alzheimer’s disease. Expert Opin Ther Patents 2002; 12: 863–870.
Woodruff-Pak DS. Preclinical experiments on cognition enhancement in Alzheimer’s disease: Drugs affecting nicotinic acetylcholine receptors. Drug Dev Res 2002;56: 335–346.
Dineley KT. Beta-amyloid peptide—nicotinic acetylcholine receptor interaction: the two faces of health and disease. Front Biosci 2007;12: 5030–5038.
Arneric SP, Holladay MW, Sullivan JP. Cholinergic channel modulators as a novel therapeutic strategy for Alzheimer’s disease. Exp Opin Invest Drugs 1996;5;79–100.
Bontempi B, Whelan KT, Risbrough VB, et al. SIB-1553A, (+/−)-4-[[2-(l-methyl-2-pyrrolidinyl)ethyl]thio] phenol hydrochloride, a subtype-selective ligand for nicotinic acetylcholine receptors with putative cognitive-enhancing properties: effects on working and reference memory performances in aged rodents and nonhuman primates. J Pharmacol Exp Ther 2001;299: 297–306.
Bencherif M, Byrd G, Caldwell WS, et al. Pharmacological characterization of RJR-2403: a nicotinic agonist with potential therapeutic benefit in the treatment of Alzheimer’s disease. CNS Drug Rev 1997;3: 325–345.
Kern WR. The brain alpha7 nicotinic receptor may be an important therapeutic target for the treatment of Alzheimer’s disease: studies with DMXBA (GTS-21). Behav Brain Res 2000;113: 169–181.
Daly JW. Nicotinic agonists, antagonists, and modulators from natural sources Cell Molec Neurobiol 2005: 25: 512–552.
Bitner RS, Bunnelle WH, Anderson DJ, et al. Broad-spectrum efficacy across cognitive domains by alpha7 nicotinic acetylcholine receptor agonism correlates with activation of ERK1/2 and CREB phosphorylation pathways. J Neurosci 2007;27: 10578–10587.
Coyle J, Kershaw P. Galantamine, a cholinesterase inhibitor that allostericallty modulates nicotinic receptors: effects on the course of Alzheimer’s disease. Biolog Psych 2002;49: 289–299.
Nitsch RM, Slack BE, Wurtman RJ, Growdon JH. Release of Alzheimer amyloid precursor derivatives stimulated by activation of muscarinic acetylcholine receptors. Science 1992;58: 304–307.
Wolf BA, Wertkin AM, Jolly YC, et al. Muscarinic regulation of Alzheimer’s disease amyloid precursor protein secretion and amyloid beta-protein production in human neuronal NT2N cells. J Biol Chem 1995;270: 4916–4922.
Haring R, Fisher A, Marciano D, et al. Mitogen-activated protein kinase-dependent and protein kinase C-dependent pathways link the Ml muscarinic receptor to amyloid precursor protein secretion. J Neurochem 1998;71: 2094–2103.
Pittel Z, Heldman E, Barg J, Haring R, Fisher A. Muscarinic control of amyloid precursor protein secretion in rat cerebral cortex and cerebellum. Brain Res 1996;742: 299–304.
Müller DM, Mendla K, Farber SA, Nitsch RM. Muscarinic Ml receptor agonists increase the secretion of the amyloid precursor protein ectodomain. Life Sci 1997;60: 985–991.
Davis AA, Fang G, Huang X, Wess J, Levey AI. Muscarinic receptor regulation of amyloid precursor protein processing in primary neuronal cultures. Neurodegener Dis 2007;4(suppl 1): 97.
Racchi M, Mazzucchelli M, Porrello E, Lanni C, Govoni S. Acetylcholinesterase inhibitors: novel activities of old molecules. Pharmacol Res 2004;50: 441–451.
Mori F, Lai CC, Fusi F, Giacobini E. Cholinesterase inhibitors increase secretion of APPs in rat brain cortex. Neuroreport 1995; 6: 633–636.
Pakaski M, Rakonczay Z, Kasa P. Reversible and irreversible acetylcholinesterase inhibitors cause changes in neuronal amyloid precursor protein processing and protein kinase C level in vitro. Neurochem Int 2001;38: 219–226.
Lahiri DK, Fallow MR. Differential effect of tacrine and physostigmine on the secretion of the beta-amyloid precursor protein in cell lines. J Mol Neurosci 1996;7: 41–49.
Kim SH, Kim YK, Jeong SJ, et al. Enhanced release of secreted form of Alzheimer’s amyloid precursor protein from PC12 cells by nicotine. Mol Pharmacol 1997;52: 430–436.
Lahiri DK, Fallow MR, Hintz N, et al. Cholinesterase inhibitors, β-amyloid precursor protein and amyloid β-peptides in Alzheimer’s disease. Acta Neurol Scand 2000;176(suppl): 60–67.
Grimes CA, Jope RS. The multifaceted roles of glycogen synthase kinase-3β in cellular signaling. Prog Neurobiol 2001;65: 391–426.
De Sarno P, Bijur GN, Zmijewska AA, Li X, Jope RS. In vivo regulation of GSK3 phosphorylation by cholinergic and NMDA receptors. Neurobiol Aging 2006;27: 413–422.
Balaram Y, Lymaye AR, Levey AI, Srinivasan S. GSK-3β and Alzheimer’s disease: pathological and therapeutic significance. Cell Mol Life Sci 2006;63: 1226–1235.
Leroy K, Yilmaz Z, Brion JP. Increased level of active GSK-3beta in Alzheimer’s disease and accumulation in argyrophilic grains and in neurones at different stages of neurofibrillary degeneration. Neuropathol Appl Neurobiol 2007;33: 43–55.
Sadot E, Gurwitz D, Barg J, Behar R, Ginzburg I, Fisher A. Activation of M1-muscarinic acetylcholine receptor regulates tau phosphorylation in transfected PC12 cells. J Neurochem 1996; 66: 877–880.
Forlenza OV, Spink JM, Dayanandan R, Anderton BH, Olesen OF, Lovestone S. Muscarinic agonists reduce τ phosphorylation in non-neuronal cells via GSK-3β inhibition and in neurons. J Neural Transm 2000;107: 1201–1212.
Genis I, Fisher A, Michaelson DM. Site-specific dephosphorylation of tau in apolipoprotein E-deficient and control mice by M1 muscarinic agonist treatment. J Neurochem 1999;12: 206–213.
Fisher A, Kealler E, Bons N. Cognitive and behavioral improvements in the aged primate Microcebus murinus following one year treatment with the M1 muscarinic agonist, AF150(S). World Conference on AD: Washington DC, July 9–13, 2000.
Hellstrom-Lindahl E, Moore H, Nordberg A. Increased levels of τ protein in SH-SY5Y cells after treatment with cholinesterase inhibitors and nicotinic agonists. J Neurochem 2000;74: 777–784.
Wang H-Y, Li W, Benedetti NJ, Lee DHS. Alpha 7 nicotinic acetylcholine receptors mediate beta-amyloid peptide-induced tau protein phosphorylation. J Biol Chem 2003;278: 31547–31553.
Dineley K T, Westerman M, Bui D, et al. Beta-amyloid activates the mitogen activated protein kinase cascade via hippocampal alpha7 nicotinic acetylcholine receptors: in vitro and in vivo mechanisms related to Alzheimer’s disease. J Neurosci 2001; 4125–4133.
Mattson MP. Central role of oxyradicals in the mechanism of amyloid β-peptide cytotoxicity. Alzheimer’s Dis Rev 1997;2: 1–14.
Gu Z, Zhong P, Yan Z. Activation of muscarinic receptors inhibits beta-amyloid peptide-induced signaling in cortical slices. J Biol Chem 2003;278: 17546–17556.
Fisher A, Brandeis R, Bar-Ner N, et al. AF150(S) and AF267B: Ml muscarinic agonists as innovative therapies for Alzheimer’s disease. J Mol Neurosci 2002;19: 145–153.
Farias GG, Godoy JA, Hernandez F, Avila J, Fisher A, Inestrosa NC. M1 muscarinic receptor activation protects neurons from beta-amyloid toxicity. A role for Wnt signaling pathway. Neurobiol Dis 2004;17: 337–348.
De Ferrari GV, Inestrosa NC. Wnt signaling function in Alzheimer’s disease. Brain Res Rev 2000;33: 1–12.
Mudher A, Lovestone S. Alzheimer’s disease-do tauists and baptists finally shake hands? Trends Neurosci 2002;25: 22–26.
Kihara T, Shimohama S, Sawada H, et al. Nicotinic receptor stimulation protects neurons against β-amyloid toxicity. Ann Neurol 1997;42: 159–163.
Kihara T, Shimohama S, Urushitani M, et al. Stimulation of α4β2 nicotinic acetylcholine receptors inhibits β-amyloid toxicity. Brain Res 1998;792: 331–334.
Svensson AL, Nordberg A. Tacrine and donepezil attenuate the neurotoxic effect of Aβ(25–35) in rat PC12 cells. Neuroreport 1998;9: 1519–1522.
Beach TG, Walker DG, Potter PE, Sue LI, Fisher A. Reduction of cerebrospinal fluid amyloid beta after systemic administration of M1 muscarinic agonists. Brain Res 2001;905: 220–223.
Beach TG, Walker DG, Sue LI, et al. Immunotoxin lesion of the cholinergic nucleus basalis causes Aβ deposition: towards a physiologic animal model of Alzheimer’s disease. Curr Med Chem-Immun Endoc & Metab Agents 2003;3: 233–243.
Oddo S, Caccamo A, Shepherd JD, et al. Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron 2003;39: 409–421.
Bons N, Rieger F, Drudhomme D, Fisher A, Krause K-H. Microcebus murinus: a useful primate model for human cerebral aging and Alzheimer’s disease? Genes Brain Behav 2005;5: 120–130.
Nordberg A, Hellstrom-Lindhal, Lee M, et al. Chronic nicotine treatment reduces β-amyloidosis in the brain of a mouse model of Alzhemer’s disease (APPsw). J Neurochem 2002;81: 655–658.
Oddo S, Caccamo A, Green KN, et al. Chronic nicotine administration exacerbates tau pathology in a transgenic model of Alzheimer’s disease. Proc Natl Acad Sci U S A 2005; 102: 3046–3051.
Liu L, Ikonen S, Heikkinen T, et al. The effects of long-term treatment with metrifonate, a cholinesterase inhibitor, on cholinergic activity, amyloid pathology, and cognitive function of APP and PS1 doubly transgenic mice. Exp Neurol 2002;173: 196–204.
Capsoni S, Giannotta S, Stebel M, et al. Ganstigmine and donepezil improve neurodegeneration in AD11 antinerve growth factor transgenic mice. Am J Alzheimers Dis Other Demen 2004;19: 153–160.
Van Dam D, Coen K, De Deyn PP. Cognitive evaluation of disease-modifying efficacy of donepezil in the APP23 mouse model for Alzheimer’s disease. Psychopharmacology (Berl) 2008;197: 37–43.
Ren K, Thinschmidt J, Liu J, et al. The alpha7 nicotinic receptor agonist 4OH-GTS-21 protects axotomized septohippocampal cholinergic neurons in wild type but not amyloid-overexpressing transgenic mice. Neuroscience 2007;145: 314–322.
Nitsch RM, Deng M, Tennis M, Schenfield D, Growdon JH. The selective muscarinic M1 agonist AF102B decreases levels of total Aβ in cerebrospinal fluid of patients with Alzheimer’s disease. Ann Neurol 2000;48: 913–918.
Hock C, Madallena A, Raschig A, et al. Treatment with the selective muscarinic Ml agonist talsaclidine decreases cerebrospinal fluid levels of Aβ42 in patients with Alzheimer’s disease. Amyloid 2003;10: 1–6.
Pameti L, Amici S, Lanari A. Cerebrospinal fluid levels of biomarkers and activity of acetylcholinesterase (AChE) and butyrylcholinesterase in Alzheimers’s disease patients before and after treatment with different AChE inhibitors. Neurolog Sci 2002;23(suppl 2): s95-s96.
Ballard CG, Chalmers KA, Todd C, et al. Cholinesterase inhibitors reduce cortical Abeta in dementia with Lewy bodies. Neurology 2007;68: 1726–1729.
Perry EK, Kilford L, Lees AJ, Bum DJ, Perry RH. Increased Alzheimer pathology in Parkinson’s disease is associated with antimuscarinic drugs. Ann Neurol 2003;54: 235–38.
Giacobini E, Becker RE. One hundred years after the discovery of Alzheimer’s disease. A turning point for therapy? J Alzheimers Dis 2007;12: 37–52.
Murphy MF, Wilcke SJ, Graham EA, Kurtz NM. First in man experience with NGX267, a selective M1 agonist for disease modification and symptomatic treatment of Alzheimer’s disease. Neurodegenerative Dis 2007;4(suppl 1): 98.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fisher, A. Cholinergic treatments with emphasis on M1 muscarinic agonists as potential disease-modifying agents for Alzheimer’s disease. Neurotherapeutics 5, 433–442 (2008). https://doi.org/10.1016/j.nurt.2008.05.002
Issue Date:
DOI: https://doi.org/10.1016/j.nurt.2008.05.002