Abstract
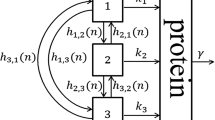
Fluctuating environments pose tremendous challenges to bacterial populations. It is observed in numerous bacterial species that individual cells can stochastically switch among multiple phenotypes for the population to survive in rapidly changing environments. This kind of phenotypic heterogeneity with stochastic phenotype switching is generally understood to be an adaptive bet-hedging strategy. Mathematical models are essential to gain a deeper insight into the principle behind bet-hedging and the pattern behind experimental data. Traditional deterministic models cannot provide a correct description of stochastic phenotype switching and bet-hedging, and traditional Markov chain models at the cellular level fail to explain their underlying molecular mechanisms. In this paper, we propose a nonlinear stochastic model of multistable bacterial systems at the molecular level. It turns out that our model not only provides a clear description of stochastic phenotype switching and bet-hedging within isogenic bacterial populations, but also provides a deeper insight into the analysis of multidimensional experimental data. Moreover, we use some deep mathematical theories to show that our stochastic model and traditional Markov chain models are essentially consistent and reflect the dynamic behavior of the bacterial system at two different time scales. In addition, we provide a quantitative characterization of the critical state of multistable bacterial systems and develop an effective data-driven method to identify the critical state without resorting to specific mathematical models.

Article PDF
Similar content being viewed by others
References
Kussell, E. and Leibler, S. (2005) Phenotypic diversity, population growth, and information in fluctuating environments. Science, 309, 2075–2078
Smits, W. K., Kuipers, O. P. and Veening, J. W. (2006) Phenotypic variation in bacteria: the role of feedback regulation. Nat. Rev. Microbiol., 4, 259–271
Dubnau, D. and Losick, R. (2006) Bistability in bacteria. Mol. Microbiol., 61, 564–572
Avery, S. V. (2006) Microbial cell individuality and the underlying sources of heterogeneity. Nat. Rev. Microbiol., 4, 577–587
Dhar, N. and McKinney, J. D. (2007) Microbial phenotypic heterogeneity and antibiotic tolerance. Curr. Opin. Microbiol., 10, 30–38
Lu, T., Shen, T., Bennett, M. R., Wolynes, P. G. and Hasty, J. (2007) Phenotypic variability of growing cellular populations. Proc. Natl. Acad. Sci. USA, 104, 18982–18987
Veening, J. W., Smits, W. K. and Kuipers, O. P. (2008) Bistability, epigenetics, and bet-hedging in bacteria. Annu. Rev. Microbiol., 62, 193–210
Fraser, D. and Kaern, M. (2009) A chance at survival: gene expression noise and phenotypic diversification strategies. Mol. Microbiol., 71, 1333–1340
Jablonka, E. and Raz, G. (2009) Transgenerational epigenetic inheritance: prevalence, mechanisms, and implications for the study of heredity and evolution. Q. Rev. Biol., 84, 131–176
Snijder, B. and Pelkmans, L. (2011) Origins of regulated cell-to-cell variability. Nat. Rev. Mol. Cell Biol., 12, 119–125
Mao, J., Blanchard, A. E. and Lu, T. (2014) Slow and steady wins the race: a bacterial exploitative competition strategy in fluctuating environments. ACS Synth. Biol. doi: 10.1021/sb4002008
Rulands, S., Jahn, D. and Frey, E. (2014) Specialization and bet hedging in heterogeneous populations. Phys. Rev. Lett., 113, 108102
Acar, M., Mettetal, J. T. and van Oudenaarden, A. (2008) Stochastic switching as a survival strategy in fluctuating environments. Nat. Genet., 40, 471–475
Salathé, M., van Cleve, J. and Feldman, M. W. (2009) Evolution of stochastic switching rates in asymmetric fitness landscapes. Genetics, 182, 1159–1164
Leisner, M., Stingl, K., Frey, E. and Maier, B. (2008) Stochastic switching to competence. Curr. Opin. Microbiol., 11, 553–559
Gaál, B., Pitchford, J. W. and Wood, A. J. (2010) Exact results for the evolution of stochastic switching in variable asymmetric environments. Genetics, 184, 1113–1119
Libby, E., Rainey, P. B. (2011) Exclusion rules, bottlenecks and the evolution of stochastic phenotype switching. P. Roy. Soc. B-Biol. Sci., 278, 3574–3583
Rainey, P. B., Beaumont, H. J., Ferguson, G. C., Gallie, J., Kost, C., Libby, E. and Zhang, X. X. (2011) The evolutionary emergence of stochastic phenotype switching in bacteria. Microb. Cell Fact., 10,Suppl, 1, S14
Ozbudak, E. M., Thattai, M., Lim, H. N., Shraiman, B. I. and van Oudenaarden, A. (2004) Multistability in the lactose utilization network of Escherichia coli. Nature, 427, 737–740
Süel, G. M., Garcia-Ojalvo, J., Liberman, L. M. and Elowitz, M. B. (2006) An excitable gene regulatory circuit induces transient cellular differentiation. Nature, 440, 545–550
Tsang, J. and van Oudenaarden, A. (2006) Exciting fluctuations: monitoring competence induction dynamics at the single-cell level. Mol. Syst. Biol., 2, 0025
Schultz, D., Ben Jacob, E., Onuchic, J. N. and Wolynes, P. G. (2007) Molecular level stochastic model for competence cycles in Bacillus subtilis. Proc. Natl. Acad. Sci. USA, 104, 17582–17587
Sonenshein, A.L., Hoch, J.A. and Losick R. (2002) Bacillus subtilis and its closest relatives: from genes to cells. Washington: American Society for Microbiology.
Errington, J. (2003) Regulation of endospore formation in Bacillus subtilis. Nat. Rev. Microbiol., 1, 117–126
Morohashi, M., Ohashi, Y., Tani, S., Ishii, K., Itaya, M., Nanamiya, H., Kawamura, F., Tomita, M. and Soga, T. (2007) Model-based definition of population heterogeneity and its effects on metabolism in sporulating Bacillus subtilis. J. Biochem., 142, 183–191
de Jong, I. G., Veening, J. W. and Kuipers, O. P. (2010) Heterochronic phosphorelay gene expression as a source of heterogeneity in Bacillus subtilis spore formation. J. Bacteriol., 192, 2053–2067
Sureka K., Ghosh, B., Dasugpta, A., Basu J., Kundu, M. and Bose Z., (2008) Positive feedback and noise activate the stringent response regulatorrel in mycobacteria. PLoS One 3, e1771.
Gefen, O. and Balaban, N. Q. (2009) The importance of being persistent: heterogeneity of bacterial populations under antibiotic stress. FEMS Microbiol. Rev., 33, 704–717
Ghosh, S., Sureka, K., Ghosh, B., Bose, I., Basu, J. and Kundu, M. (2011) Phenotypic heterogeneity in mycobacterial stringent response. BMC Syst. Biol., 5, 18
Veening, J.W., Stewart, E. J., Berngruber, T.W., Taddei, F., Kuipers, O. P. and Hamoen, L.W. (2008) Bet-hedging and epigenetic inheritance in bacterial cell development. Proc. Natl. Acad. Sci. USA, 105, 4393–4398
Beaumont, H. J., Gallie, J., Kost, C., Ferguson, G. C. and Rainey, P. B. (2009) Experimental evolution of bet hedging. Nature, 462, 90–93
Wolf, D. M., Vazirani, V. V. and Arkin, A. P. (2005) Diversity in times of adversity: probabilistic strategies in microbial survival games. J. Theor. Biol., 234, 227–253
Gupta, P. B., Fillmore, C. M., Jiang, G., Shapira, S. D., Tao, K., Kuperwasser, C. and Lander, E. S. (2011) Stochastic state transitions give rise to phenotypic equilibrium in populations of cancer cells. Cell, 146, 633–644
Zhou, D., Wu, D., Li, Z., Qian, M., and Zhang, M. Q. (2013) Population dynamics of cancer cells with cell state conversions. Quant. Biol., 1, 1–8.
Karmakar, R. and Bose, I. (2007) Positive feedback, stochasticity and genetic competence. Phys. Biol., 4, 29–37
Mitrophanov, A. Y. and Groisman, E. A. (2008) Positive feedback in cellular control systems. BioEssays, 30, 542–555
Mantzaris, N. V. (2007) From single-cell genetic architecture to cell population dynamics: quantitatively decomposing the effects of different population heterogeneity sources for a genetic network with positive feedback architecture. Biophys. J., 92, 4271–4288
Vellela, M. and Qian, H. (2009) Stochastic dynamics and nonequilibrium thermodynamics of a bistable chemical system: the Schlögl model revisited. J. R. Soc. Interface, 6, 925–940
Qian, H. and Bishop, L. M. (2010) The chemical master equation approach to nonequilibrium steady-state of open biochemical systems: linear single-molecule enzyme kinetics and nonlinear biochemical reaction networks. Int. J. Mol. Sci., 11, 3472–3500
Qian, H. (2011) Nonlinear stochastic dynamics of mesoscopic homogeneous biochemical reaction systems-an analytical theory. Nonlinearity, 24, R19–R49
Ge, H. and Qian, H. (2011) Non-equilibrium phase transition in mesoscopic biochemical systems: from stochastic to nonlinear dynamics and beyond. J. R. Soc. Interface, 8, 107–116
Qian, H. (2012) Cooperativity in cellular biochemical processes: noise-enhanced sensitivity, fluctuating enzyme, bistability with nonlinear feedback, and other mechanisms for sigmoidal responses. Annu. Rev. Biophys., 41, 179–204
Qian, H. and Ge, H. (2012) Mesoscopic biochemical basis of isogenetic inheritance and canalization: stochasticity, nonlinearity, and emergent landscape. Mol Cell Biomech., 9, 1–30
McAdams, H. H. and Arkin, A. (1997) Stochastic mechanisms in gene expression. Proc. Natl. Acad. Sci. USA, 94, 814–819
Elowitz, M. B., Levine, A. J., Siggia, E. D. and Swain, P. S. (2002) Stochastic gene expression in a single cell. Science, 297, 1183–1186
Ozbudak, E. M., Thattai, M., Kurtser, I., Grossman, A. D. and van Oudenaarden, A. (2002) Regulation of noise in the expression of a single gene. Nat. Genet., 31, 69–73
Paulsson, J. (2004) Summing up the noise in gene networks. Nature, 427, 415–418
Kaern, M., Elston, T. C., Blake, W. J. and Collins, J. J. (2005) Stochasticity in gene expression: from theories to phenotypes. Nat. Rev. Genet., 6, 451–464
Raser, J. M. and O’Shea, E. K. (2005) Noise in gene expression: origins, consequences, and control. Science, 309, 2010–2013
Cai, L., Friedman, N. and Xie, X. S. (2006) Stochastic protein expression in individual cells at the single molecule level. Nature, 440, 358–362
Yu, J., Xiao, J., Ren, X., Lao, K. and Xie, X. S. (2006) Probing gene expression in live cells, one protein molecule at a time. Science, 311, 1600–1603
Xie, X. S., Choi, P. J., Li, G. W., Lee, N. K. and Lia, G. (2008) Singlemolecule approach to molecular biology in living bacterial cells. Annu. Rev. Biophys., 37, 417–444
Sanchez, A., Choubey, S. and Kondev, J. (2013) Regulation of noise in gene expression. Annu. Rev. Biophys., 42, 469–491
Freidlin, M. I., Szücs, J. and Wentzell, A. D. (2012) Random perturbations of dynamical systems, 3rd ed. New York: Springer-Verlag
Kim, D., Rath, O., Kolch, W. and Cho, K. H. (2007) A hidden oncogenic positive feedback loop caused by crosstalk between Wnt and ERK pathways. Oncogene, 26, 4571–4579
Kellershohn, N. and Laurent, M. (2001) Prion diseases: dynamics of the infection and properties of the bistable transition. Biophys. J., 81, 2517–2529
Olivieri, E. and Vares, M. E. (2005) Large deviations and metastability. UK: Cambridge University Press.
Liu, R., Li, M.Y., Liu, Z.P., Wu, J.R., Chen, L.N. and Aihara, K. (2012) Identifying critical transitions and their leading biomolecular networks in complex diseases. Sci. Rep., 2, 813
Dai, L., Vorselen, D., Korolev, K. S. and Gore, J. (2012) Generic indicators for loss of resilience before a tipping point leading to population collapse. Science, 336, 1175–1177
Angeli, D., Ferrell, J. E. Jr. and Sontag, E. D. (2004) Detection of multistability, bifurcations, and hysteresis in a large class of biological positive-feedback systems. Proc. Natl. Acad. Sci. USA, 101, 1822–1827
Gouzé, J. L. (1998) Positive and negative circuits in dynamical systems. J. Biol. Syst., 6, 11–15
Snoussi, E. H. (1998) Necessary conditions for multistationarity and stable periodicity. J. Biol. Syst., 6, 3–9
Cinquin, O. and Demongeot, J. (2002) Positive and negative feedback: striking a balance between necessary antagonists. J. Theor. Biol., 216, 229–241
Gillespie, D. T. (1992) A rigorous derivation of the chemical master equation. Physica A, 188, 404–425
Kurtz, T. G. (1970) Solutions of ordinary differential equations as limits of pure jump Markov processes. J. Appl. Probab., 7, 49–58
Kurtz, T. G. (1971) Limit theorems for sequences of jump Markov processes approximating ordinary differential processes. J. Appl. Probab., 8, 344–356
Kurtz, T. G. (1972) The relationship between stochastic and deterministic models for chemical reactions. J. Chem. Phys., 57, 2976–2978
Gillespie, D. T. (2000) The chemical Langevin equation. J. Chem. Phys., 113, 297–306
Rao, C. V., Wolf, D. M. and Arkin, A. P. (2002) Control, exploitation and tolerance of intracellular noise. Nature, 420, 231–237
Wilkinson, D. J. (2009) Stochastic modelling for quantitative description of heterogeneous biological systems. Nat. Rev. Genet., 10, 122–133
Meister, A., Du, C., Li, Y. H., Wong, W. H. (2014) Modeling stochastic noise in gene regulatory systems. Quant. Biol., 2, 1–29
Yao, G., Lee, T. J., Mori, S., Nevins, J. R. and You, L. (2008) A bistable Rb-E2F switch underlies the restriction point. Nat. Cell Biol., 10, 476–482
Ferrell, J. E. Jr and Machleder, E. M. (1998) The biochemical basis of an all-or-none cell fate switch in Xenopus oocytes. Science, 280, 895–898
Xiong, W. and Ferrell, J. E. Jr. (2003) A positive-feedback-based bistable ‘memory module’ that governs a cell fate decision. Nature, 426, 460–465
Corson, F. and Siggia, E. D. (2012) Geometry, epistasis, and developmental patterning. Proc. Natl. Acad. Sci. USA, 109, 5568–5575
Yan, L., Yang, M., Guo, H., Yang, L., Wu, J., Li, R., Liu, P., Lian, Y., Zheng, X., Yan, J., et al. (2013) Single-cell RNA-Seq profiling of human preimplantation embryos and embryonic stem cells. Nat. Struct. Mol. Biol., 20, 1131–1139
Eissing, T., Conzelmann, H., Gilles, E. D., Allgöwer, F., Bullinger, E. and Scheurich, P. (2004) Bistability analyses of a caspase activation model for receptor-induced apoptosis. J. Biol. Chem., 279, 36892–36897
Spencer, S. L., Gaudet, S., Albeck, J. G., Burke, J. M. and Sorger, P. K. (2009) Non-genetic origins of cell-to-cell variability in TRAIL-induced apoptosis. Nature, 459, 428–432
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Jia, C., Qian, M., Kang, Y. et al. Modeling stochastic phenotype switching and bet-hedging in bacteria: stochastic nonlinear dynamics and critical state identification. Quant Biol 2, 110–125 (2014). https://doi.org/10.1007/s40484-014-0035-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40484-014-0035-5