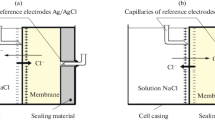
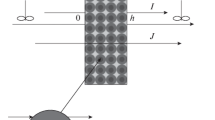
Abstract
We conducted a theoretical study of ion transport through a polar cationic membrane subjected to the action of an electric field. We solved the transport equation without the usual electroneutrality assumption. We investigated the validity of the constant field approximation in the study of ion transport across a membrane and analyzed the case in which the membrane’s charge density is constant. We have shown, by numerical simulations, that the PS model (Sistat and Pourcelly, J Elec Chem 460:53, 1999) does not reproduce the three regimes of polarization curve mode. We also proposed an alternative approach to the problem based on Mean Field approximation. Our approach reproduces qualitatively better the well-known behavior of the polarization curve.





Similar content being viewed by others
References
Ayala-Bribiesca E, Pourcelly G, Bazinet L (2007) Nature identification and morphology characterization of anion-exchange membrane fouling during conventional electrodialysis. J Colloid Interface Sci 308:182–190
Bard AJ, Faulkner LR (2000) Electrochemical methods fundamentals and applications, 2nd edn. John Wiley & Sons Inc, New York
Dalla Costa RF, Rodrigues MAS, Zoppas Ferreira J (1998) Transport of trivalent and hexavalent chromium through different ion-selective membranes in acidic aqueous media. Sep Sci Technol 33(8):1135
Ferreira CA, Casanovas J, Rodrigues MAS, Muller F, Armelin E, Alemán C (2010) Transport of metallicions through polyaniline-containing composite membranes. J Chem Eng Data 55:4801–4807
Moura RCA, Bertuol DA, Ferreira CA, Franco DR, Amado FDR (2012) Study of chromium removal by the electrodialysis of tannery and metal finishing effluents. Int J Chem Eng, Article ID 179312
Noble RD, Stern SA (1995) Membrane separations and technology principles and application. Elsevier, New York, NY
Rodrigues MAS, Dalla Costa RF, Bernardes AM, Zoppas Ferreira J (2001) Influence of ligand exchange on the treatment of trivalent chromium solutions by electrodialysis. Electrochim Acta 47:753
Scott K (1995) Handbook of Industrial Membranes. Elservier Advanced Technology, Oxford
Strathmann H (2005) Electrodialysis and related prosses. In: Noble RD, Stern SA (eds) Membrane Separations Technology-Principles and Applications. Elservier, New York, pp 213–281 (2005)
Vallois C, Sistat P, Roualdès S, Pourcelly G (2003) Separation of H+/Cu2+ cations by electrodialysis using modified proton conducting membranes. Memb Sci 216:13–25
Sistat P, Pourcelly G (1999) Steady-state ion transport through homopolar ion-exchange membranes: an analytical solution of the Nernst–Planck equations for a 1:1 electrolyte under the electroneutrality assumption. J Elec Chem 460:53–62
Vilfan I (1998) Lecture notes in statistical mechanics: the Abdus Salam ICTP diploma programme, academic year 1998–99 : condensed matter physics courses : course on statistical mechanics
Feldberg SW (2000) On the dilemma of the use of the electroneutrality constraint in electrochemical calculations. Elec Chem Commun 2:453
Barry DA, Parlange JY, Prommer H, Cunningham CJ, Stagnitti FM et al (2003) Analytical approximations for real values of the Lambert W-function. Math Comput Simul 53:95–103
Bergman TL, Lavine AS, Incropera FP, Dewitt DP (2011) Fundamentals of heat and mass transfer, 7th edn. Wiley, New York
Buck RP (1984) Kinetics of bulk and interfacial ionic motion: microscopic basis and limits for the Nernst-Planck eqautions appilied to membrane systems. J Membr Sci 17:1–62
Corry B, Kuyucak S, Chung S (2000) Tests of continuum theories as models of ion channels. II. Poisson-Nernst-Planck theory versus brownian dynamics. Biophys J 78:2364–2381
Fíla V, Bouzek K (2008) The effect of convection in the external diffusion layer on the results of a mathematical model of multiple ion transport across an ion-selective membrane. J Appl Electrochem 38:1241–1252
Nikonenko V, Zabolotsky V, Larchet C, Auclair B, Pourcelly G (2002) Mathematical description of ion transport in membrane systems. Desalination 147:369–374
Rohman FS, Aziz N (2008) Steady-state space charge effects in symmetric cells with concentration polarized electrodes. Bull Chem React Eng Catal 3(1–3):3–8
Syganow A, von Kitzing E (1999) (In)validity of the constant field and constant currents assumptions in theories of ion transport. Biophys J 76:768–781
Tanioka A, Matsumoto H, Yamamoto R (2004) Charge effectiveness of sulfonated polymer membranes under low-water-content condition. Sci Technol Adv Mater 5:461–468
Volgin VM, Davydov AD (2005) Ionic transport through ion-exchange and bipolar membranes. J Membr Sci 259:110–121
Wang J et al (2014) A critical review of transport through osmotic membranes. J Membr Sci 454:516–537
Vieira IP, Oliveira AC (2015) Work in progress
Acknowledgments
The authors would like to thank the editor and the referee for the valuable comments on our submitted manuscript. A. C. O. acknowledges FAPEMIG for the partial financial support. F. D. R. A. acknowledges CNPq for the partial financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Technical Editor: Francisco Ricardo Cunha.
Rights and permissions
About this article
Cite this article
Oliveira, A.C., Amado, F.D.R. & Moura, R.C.A. Steady state of ion transport in homopolar ion-exchange membrane: a theoretical study. J Braz. Soc. Mech. Sci. Eng. 38, 1165–1170 (2016). https://doi.org/10.1007/s40430-015-0357-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40430-015-0357-x