Abstract
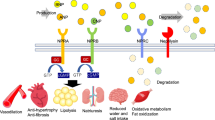
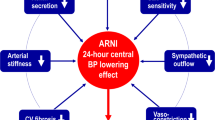
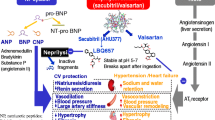
The burden of cardiovascular disease (CVD) is continuously and progressively raising worldwide. Essential hypertension is a major driver of cardiovascular events, including coronary artery disease, myocardial infarction, ischemic stroke and congestive heart failure. This latter may represent the final common pathway of different cardiovascular diseases, and it is often mediated by progressive uncontrolled hypertension. Despite solid advantages derived from effective and sustained blood pressure control, and the widespread availability of effective antihypertensive medications, the vast majority of the more than 1 billion hypertensive patients worldwide continue to have uncontrolled hypertension. Among various factors that may be involved, the abnormal activation of neurohormonal systems is one consistent feature throughout the continuum of cardiovascular diseases. These systems may initiate biologically meaningful “injury responses”. However, their sustained chronic overactivity often may induce and maintain the progression from hypertension towards congestive heart failure. The renin–angiotensin–aldosteron system, the sympathetic nervous system and the endothelin system are major neurohormonal stressor systems that are not only able to elevate blood pressure levels by retaining water and sodium, but also to play a role in the pathophysiology of cardiovascular diseases. More recently, the angiotensin receptor neprilysin inhibitor (ARNI) represents a favourable approach to inhibit neutral endopeptidase (NEP) and suppress the RAAS via blockade of the AT1 receptors, without the increased risk of angioedema. LCZ696, the first-in-class ARNI, has already demonstrated BP lowering efficacy in patients with hypertension, in particular with respect to systolic blood pressure levels, improved cardiac biomarkers, cardiac remodelling and prognosis in patients with heart failure. This manuscript will briefly overview the main pathophysiological and therapeutic aspects of ARNI in the clinical management of hypertension and heart failure.
Similar content being viewed by others
References
Kearney PM, Whelton M, Reynolds K, et al. Global burden of hypertension: analysis of worldwide data. Lancet. 2005;365:217–23.
Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338:b1665.
Wolf-Maier K, Cooper RS, Kramer H, et al. Hypertension treatment and control in five European countries, Canada, and the United States. Hypertension. 2004;43:10–7.
Goldsmith SR. Interactions between the sympathetic nervous system and the RAAS in heart failure. Curr Heart Fail Rep. 2004;1:45–50.
Parati G, Esler M. The human sympathetic nervous system: its relevance in hypertension and heart failure. Eur Heart J. 2012;33:1058–66.
Volpe M, Cosentino F, Tocci G, et al. Antihypertensive therapy in diabetes: the legacy effect and RAAS blockade. Curr Hypertens Rep. 2011;13:318–24.
Marchesi C, Paradis P, Schiffrin EL. Role of the renin-angiotensin system in vascular inflammation. Trends Pharmacol Sci. 2008;29:367–74.
von Lueder TG, Sangaralingham SJ, Wang BH, et al. Renin-angiotensin blockade combined with natriuretic peptide system augmentation: novel therapeutic concepts to combat heart failure. Circ Heart Fail. 2013;6:594–605.
Burnett JC Jr, Granger JP, Opgenorth TJ. Effects of synthetic atrial natriuretic factor on renal function and renin release. Am J Physiol. 1984;247:F863–6.
Wada A, Tsutamoto T, Matsuda Y, et al. Cardiorenal and neurohumoral effects of endogenous atrial natriuretic peptide in dogs with severe congestive heart failure using a specific antagonist for guanylate cyclase-coupled receptors. Circulation. 1994;89:2232–40.
Stevens SM Jr, Kem WR, Prokai L. Investigation of cytolysin variants by peptide mapping: enhanced protein characterization using complementary ionization and mass spectrometric techniques. Rapid Commun Mass Spectrom. 2002;16:2094–101.
Cataliotti A, Boerrigter G, Costello-Boerrigter LC, et al. Brain natriuretic peptide enhances renal actions of furosemide and suppresses furosemide-induced aldosterone activation in experimental heart failure. Circulation. 2004;109:1680–5.
Publication Committee for the VI. Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA. 2002;287:1531–40.
Colucci WS, Elkayam U, Horton DP, et al. Intravenous nesiritide, a natriuretic peptide, in the treatment of decompensated congestive heart failure. Nesiritide Study Group. N Engl J Med. 2000;343:246–53.
Hobbs RE, Mills RM, Young JB. An update on nesiritide for treatment of decompensated heart failure. Expert Opin Investig Drugs. 2001;10:935–42.
Boerrigter G, Burnett JC Jr. Recent advances in natriuretic peptides in congestive heart failure. Expert Opin Investig Drugs. 2004;13:643–52.
Wang TJ, Larson MG, Levy D, et al. Impact of obesity on plasma natriuretic peptide levels. Circulation. 2004;109:594–600.
Sackner-Bernstein JD, Kowalski M, Fox M, et al. Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA. 2005;293:1900–5.
Sackner-Bernstein JD, Skopicki HA, Aaronson KD. Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure. Circulation. 2005;111:1487–91.
Kerr MA, Kenny AJ. The molecular weight and properties of a neutral metallo-endopeptidase from rabbit kidney brush border. Biochem J. 1974;137:489–95.
Malfroy B, Swerts JP, Guyon A, et al. High-affinity enkephalin-degrading peptidase in brain is increased after morphine. Nature. 1978;276:523–6.
Turner AJ, Tanzawa K. Mammalian membrane metallopeptidases: NEP, ECE, KELL, and PEX. FASEB J. 1997;11:355–64.
Stephenson SL, Kenny AJ. The hydrolysis of alpha-human atrial natriuretic peptide by pig kidney microvillar membranes is initiated by endopeptidase-24.11. Biochem J. 1987;243:183–7.
Stephenson SL, Kenny AJ. Metabolism of neuropeptides. Hydrolysis of the angiotensins, bradykinin, substance P and oxytocin by pig kidney microvillar membranes. Biochem J. 1987;241:237–47.
Vijayaraghavan J, Scicli AG, Carretero OA, et al. The hydrolysis of endothelins by neutral endopeptidase 24.11 (enkephalinase). J Biol Chem. 1990;265:14150–5.
Shima M, Seino Y, Torikai S, et al. Intrarenal localization of degradation of atrial natriuretic peptide in isolated glomeruli and cortical nephron segments. Life Sci. 1988;43:357–63.
Graf K, Koehne P, Grafe M, et al. Regulation and differential expression of neutral endopeptidase 24.11 in human endothelial cells. Hypertension. 1995;26:230–5.
Dussaule JC, Stefanski A, Bea ML, et al. Characterization of neutral endopeptidase in vascular smooth muscle cells of rabbit renal cortex. Am J Physiol. 1993;264:F45–52.
Standeven KF, Hess K, Carter AM, et al. Neprilysin, obesity and the metabolic syndrome. Int J Obes (Lond). 2011;35:1031–40.
Ferro CJ, Spratt JC, Haynes WG, et al. Inhibition of neutral endopeptidase causes vasoconstriction of human resistance vessels in vivo. Circulation. 1998;97:2323–30.
Burnett JC Jr. Vasopeptidase inhibition: a new concept in blood pressure management. J Hypertens Suppl. 1999;17:S37–43.
Cataliotti A, Boerrigter G, Chen HH, et al. Differential actions of vasopeptidase inhibition versus angiotensin-converting enzyme inhibition on diuretic therapy in experimental congestive heart failure. Circulation. 2002;105:639–44.
Hoover T, Lippmann M, Grouzmann E, et al. Angiotensin converting enzyme inhibitor induced angio-oedema: a review of the pathophysiology and risk factors. Clin Exp Allergy. 2010;40:50–61.
Adam A, Cugno M, Molinaro G, et al. Aminopeptidase P in individuals with a history of angio-oedema on ACE inhibitors. Lancet. 2002;359:2088–9.
Gu J, Noe A, Chandra P, et al. Pharmacokinetics and pharmacodynamics of LCZ696, a novel dual-acting angiotensin receptor-neprilysin inhibitor (ARNi). J Clin Pharmacol. 2010;50:401–14.
Ruilope LM, Dukat A, Bohm M, et al. Blood-pressure reduction with LCZ696, a novel dual-acting inhibitor of the angiotensin II receptor and neprilysin: a randomised, double-blind, placebo-controlled, active comparator study. Lancet. 2010;375:1255–66.
Kusaka H, Sueta D, Koibuchi N, et al. LCZ696, angiotensin II receptor-neprilysin inhibitor, ameliorates high-salt-induced hypertension and cardiovascular injury more than valsartan alone. Am J Hypertens. 2015 [Epub ahead of print].
Kario K, Sun N, Chiang FT, et al. Efficacy and safety of LCZ696, a first-in-class angiotensin receptor neprilysin inhibitor, in Asian patients with hypertension: a randomized, double-blind, placebo-controlled study. Hypertension. 2014;63:698–705.
Williams B, Cockcroft JR, Kario K, et al. Rationale and study design of the Prospective comparison of Angiotensin Receptor neprilysin inhibitor with Angiotensin receptor blocker MEasuring arterial sTiffness in the eldERly (PARAMETER) study. BMJ Open. 2014;4:e004254.
Masson S, Latini R, Anand IS, et al. Direct comparison of B-type natriuretic peptide (BNP) and amino-terminal proBNP in a large population of patients with chronic and symptomatic heart failure: the Valsartan Heart Failure (Val-HeFT) data. Clin Chem. 2006;52:1528–38.
Mangiafico S, Costello-Boerrigter LC, Andersen IA, et al. Neutral endopeptidase inhibition and the natriuretic peptide system: an evolving strategy in cardiovascular therapeutics. Eur Heart J. 2013;34:886–93c.
Solomon SD, Zile M, Pieske B, et al. The angiotensin receptor neprilysin inhibitor LCZ696 in heart failure with preserved ejection fraction: a phase 2 double-blind randomised controlled trial. Lancet. 2012;380:1387–95.
Tatoulis J. Coronary revascularization in heart failure-old lessons relearnt: patient outcomes are paramount in the stenting era. Eur J Cardiothorac Surg. 2015;47:322–3.
McMurray JJ, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med. 2014;371:993–1004.
Packer M, McMurray JJ, Desai AS, et al. Angiotensin receptor neprilysin inhibition compared with enalapril on the risk of clinical progression in surviving patients with heart failure. Circulation. 2015;131:54–61.
Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145–53.
Fox KM. Investigators EUtOrocewPiscAd. Efficacy of perindopril in reduction of cardiovascular events among patients with stable coronary artery disease: randomised, double-blind, placebo-controlled, multicentre trial (the EUROPA study). Lancet. 2003;362:782–8.
Volpe M, Rubattu S, Burnett J Jr. Natriuretic peptides in cardiovascular diseases: current use and perspectives. Eur Heart J. 2014;35:419–25.
Squire IB, O’Brien RJ, Demme B, et al. N-terminal pro-atrial natriuretic peptide (N-ANP) and N-terminal pro-B-type natriuretic peptide (N-BNP) in the prediction of death and heart failure in unselected patients following acute myocardial infarction. Clin Sci (Lond). 2004;107:309–16.
Makikallio AM, Makikallio TH, Korpelainen JT, et al. Natriuretic peptides and mortality after stroke. Stroke. 2005;36:1016–20.
Kasama S, Furuya M, Toyama T, et al. Effect of atrial natriuretic peptide on left ventricular remodelling in patients with acute myocardial infarction. Eur Heart J. 2008;29:1485–94.
Kasama S, Toyama T, Hatori T, et al. Effects of intravenous atrial natriuretic peptide on cardiac sympathetic nerve activity and left ventricular remodeling in patients with first anterior acute myocardial infarction. J Am Coll Cardiol. 2007;49:667–74.
Kitakaze M, Asakura M, Kim J, et al. Human atrial natriuretic peptide and nicorandil as adjuncts to reperfusion treatment for acute myocardial infarction (J-WIND): two randomised trials. Lancet. 2007;370:1483–93.
Chen HH, Martin FL, Gibbons RJ, et al. Low-dose nesiritide in human anterior myocardial infarction suppresses aldosterone and preserves ventricular function and structure: a proof of concept study. Heart. 2009;95:1315–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volpe, M., Tocci, G., Battistoni, A. et al. Angiotensin II Receptor Blocker Neprilysin Inhibitor (ARNI): New Avenues in Cardiovascular Therapy. High Blood Press Cardiovasc Prev 22, 241–246 (2015). https://doi.org/10.1007/s40292-015-0112-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-015-0112-5