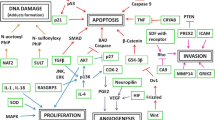
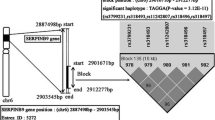
Abstract
Background and Objectives
To investigate a potential association between single-nucleotide polymorphisms (SNPs) and haplotypes at the TNFA-LTA locus and the development of oral cancer in an Indian population.
Materials and Methods
In this study, 150 oral precancer/cancer samples (50 precancer and 100 cancer), along with an equal number of control samples, were genotyped. Six SNPs at the TNF-LTA locus (i.e., −238G/A, −308G/A, −857C/T, −863C/A, −1031T/C, and +252A/G) were analyzed by use of a polymerase chain reaction–restriction fragment length polymorphism method, the assay was validated by sequencing 10 % of samples.
Results
The allelic frequencies of TNFA and LTA SNPs were found to be significantly associated with the risk of oral cancer and precancerous lesions in comparison with controls (P < 0.0003). Further haplotypic analysis showed that two haplotypes (ATCTGG and ACACGG) served as risk haplotypes for oral cancer. These haplotypes were also found to be significantly and positively associated with lifestyle habits (tobacco chewing P = 0.04, odds ratio [OR] 3.4) and socioeconomic status (P = 0.01, OR 3.4). We noticed an increased percentage of risk haplotypes correlating with the aggressiveness of oral cancer. The percentages of risk haplotypes were found to be threefold higher in precancer and fourfold higher in advanced stages of oral cancer in comparison with controls.
Conclusion
Five SNPs at the TNF-LTA locus (i.e., −308G>A, −857C>T, −863C>A, −1031T>C, and +252A>G) were found to be associated with the development of oral cancer. Two haplotypes (ATCTGG and ACACGG) emerged as major risk haplotypes for oral carcinoma progression and were also found to be associated with lifestyle factors and clinical aggressiveness. These findings make the TNF-LTA locus a suitable candidate for a future biomarker, which may be used either for early detection or for helping to improve treatment efficacy and effectiveness.
Similar content being viewed by others
References
Fitzmaurice C, Dicker D, Pain A, Hamavid H, Moradi-Lakeh M, MacIntyre MF, et al. The global burden of cancer 2013. JAMA Oncol. 2015;1(4):505–27.
Williams HK. Molecular pathogenesis of oral squamous carcinoma. Mol Pathol. 2000;53(4):165–72.
Jayalekshmi PA, Gangadharan P, Akiba S, Nair RR, Tsuji M, Rajan B. Tobacco chewing and female oral cavity cancer risk in Karunagappally cohort, India. Br J Cancer. 2009;100(5):848–52.
Mehta FS, Bhonsle RB, Murti PR, Daftary DK, Gupta PC, Pindborg JJ. Central papillary atrophy of the tongue among bidi smokers in India: a 10-year study of 182 lesions. J Oral Pathol Med. 1989;18(8):475–80.
Raabe T, Bukrinsky M, Currie RA. Relative contribution of transcription and translation to the induction of tumor necrosis factor-alpha by lipopolysaccharide. J Biol Chem. 1998;273(2):974–80.
Gupta R, Sharma SC, Das SN. Association of TNF-alpha and TNFR1 promoters and 3’ UTR region of TNFR2 gene polymorphisms with genetic susceptibility to tobacco-related oral carcinoma in Asian Indians. Oral Oncol. 2008;44(5):455–63.
Kohaar I, Tiwari P, Kumar R, Nasare V, Thakur N, Das BC, et al. Association of single nucleotide polymorphisms (SNPs) in TNF-LTA locus with breast cancer risk in Indian population. Breast Cancer Res Treat. 2009;114(2):347–55.
Yang CM, Hou YY, Chiu YT, Chen HC, Chu ST, Chi CC, et al. Interaction between tumour necrosis factor-alpha gene polymorphisms and substance use on risk of betel quid-related oral and pharyngeal squamous cell carcinoma in Taiwan. Arch Oral Biol. 2011;56(10):1162–9.
Vairaktaris E, Yapijakis C, Serefoglou Z, Avgoustidis D, Critselis E, Spyridonidou S, et al. Gene expression polymorphisms of interleukins-1 beta, -4, -6, -8, -10, and tumor necrosis factors -alpha, -beta: regression analysis of their effect upon oral squamous cell carcinoma. J Cancer Res Clin Oncol. 2008;134(8):821–32.
Yapijakis C, Serefoglou Z, Vylliotis A, Nkenke E, Derka S, Vassiliou S, et al. Association of polymorphisms in tumor necrosis factor alpha and beta genes with increased risk for oral cancer. Anticancer Res. 2009;29(6):2379–86.
Kramer IR, Lucas RB, Pindborg JJ, Sobin LH. Definition of leukoplakia and related lesions: an aid to studies on oral precancer. Oral Surg Oral Med Oral Pathol. 1978;46(4):518–39.
Bjartveit K, Tverdal A. Health consequences of smoking 1–4 cigarettes per day. Tob Control. 2005;14(5):315–20.
Dufour MC. What is moderate drinking? Defining “drinks” and drinking levels. Alcohol Res Health. 1999;23(1):5–14.
Bairwa M, Rajput M, Sachdeva S. Modified Kuppuswamy’s socioeconomic scale: social researcher should include updated income criteria, 2012. Indian J Community Med. 2012;38(3):185–6.
Sambrook J, Fritsh EF, Maniatis T. Molecular cloning: a laboratory manual. New York: Cold Spring Harbor Laboratory Press; 1989.
Barrett JC, Fry B, Maller J, Daly MJ. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics. 2005;21(2):263–5.
Azmy IA, Balasubramanian SP, Wilson AG, Stephenson TJ, Cox A, Brown NJ, et al. Role of tumour necrosis factor gene polymorphisms (−308 and −238) in breast cancer susceptibility and severity. Breast Cancer Res. 2004;6(4):R395–400.
Guo W, Wang N, Li Y, Zhang JH. Polymorphisms in tumor necrosis factor genes and susceptibility to esophageal squamous cell carcinoma and gastric cardiac adenocarcinoma in a population of high incidence region of North China. Chin Med J (Engl). 2005;118(22):1870–8.
Saito S, Kasai Y, Nomoto S, Fujiwara M, Akiyama S, Ito K, et al. Polymorphism of tumor necrosis factor in esophageal, gastric or colorectal carcinoma. Hepatogastroenterology. 2001;48(38):468–70.
Kroeger KM, Carville KS, Abraham LJ. The −308 tumor necrosis factor-alpha promoter polymorphism effects transcription. Mol Immunol. 1997;34(5):391–9.
Liu CJ, Wong YK, Chang KW, Chang HC, Liu HF, Lee YJ. Tumor necrosis factor-alpha promoter polymorphism is associated with susceptibility to oral squamous cell carcinoma. J Oral Pathol Med. 2005;34(10):608–12.
Messer G, Spengler U, Jung MC, Honold G, Blomer K, Pape GR, et al. Polymorphic structure of the tumor necrosis factor (TNF) locus: an NcoI polymorphism in the first intron of the human TNF-beta gene correlates with a variant amino acid in position 26 and a reduced level of TNF-beta production. J Exp Med. 1991;173(1):209–19.
Kohaar I, Thakur N, Salhan S, Batra S, Singh V, Sharma A, et al. TNFalpha-308G/A polymorphism as a risk factor for HPV associated cervical cancer in Indian population. Cell Oncol. 2007;29(3):249–56.
Jang WH, Yang YI, Yea SS, Lee YJ, Chun JH, Kim HI, et al. The −238 tumor necrosis factor-alpha promoter polymorphism is associated with decreased susceptibility to cancers. Cancer Lett. 2001;166(1):41–6.
Gallagher G, Eskdale J, Oh HH, Richards SD, Campbell DA, Field M. Polymorphisms in the TNF gene cluster and MHC serotypes in the West of Scotland. Immunogenetics. 1997;45(3):188–94.
Singh PK, Bogra J, Chandra G, Ahmad MK, Gupta R, Kumar V, et al. Association of TNF-alpha (−238 and −308) promoter polymorphisms with susceptibility of oral squamous cell carcinoma in North Indian population. Cancer Biomark. 2014;15(2):125–31.
Liu L, Yang X, Chen X, Kan T, Shen Y, Chen Z, et al. Association between TNF-alpha polymorphisms and cervical cancer risk: a meta-analysis. Mol Biol Rep. 2011;39(3):2683–8.
Higuchi T, Seki N, Kamizono S, Yamada A, Kimura A, Kato H, et al. Polymorphism of the 5′-flanking region of the human tumor necrosis factor (TNF)-alpha gene in Japanese. Tissue Antigens. 1998;51(6):605–12.
Kaijzel EL, Bayley JP, van Krugten MV, Smith L, van de Linde P, Bakker AM, et al. Allele-specific quantification of tumor necrosis factor alpha (TNF) transcription and the role of promoter polymorphisms in rheumatoid arthritis patients and healthy individuals. Genes Immun. 2001;2(3):135–44.
Uglialoro AM, Turbay D, Pesavento PA, Delgado JC, McKenzie FE, Gribben JG, et al. Identification of three new single nucleotide polymorphisms in the human tumor necrosis factor-alpha gene promoter. Tissue Antigens. 1998;52(4):359–67.
Lee SG, Kim B, Yook JH, Oh ST, Lee I, Song K. TNF/LTA polymorphisms and risk for gastric cancer/duodenal ulcer in the Korean population. Cytokine. 2004;28(2):75–82.
Yang JJ, Ko KP, Cho LY, Shin A, Gwack J, Chang SH, et al. The role of TNF genetic variants and the interaction with cigarette smoking for gastric cancer risk: a nested case–control study. BMC Cancer. 2009;9:238.
Grutters JC, Sato H, Pantelidis P, Lagan AL, McGrath DS, Lammers JW, et al. Increased frequency of the uncommon tumor necrosis factor −857T allele in British and Dutch patients with sarcoidosis. Am J Respir Crit Care Med. 2002;165(8):1119–24.
Hashibe M, Brennan P, Chuang SC, Boccia S, Castellsague X, Chen C, et al. Interaction between tobacco and alcohol use and the risk of head and neck cancer: pooled analysis in the International Head and Neck Cancer Epidemiology Consortium. Cancer Epidemiol Biomarkers Prev. 2009;18(2):541–50.
Joshi MS, Verma Y, Gautam AK, Parmar G, Lakkad BC, Kumar S. Cytogenetic alterations in buccal mucosa cells of chewers of areca nut and tobacco. Arch Oral Biol. 2010;56(1):63–7.
Ko YC, Huang YL, Lee CH, Chen MJ, Lin LM, Tsai CC. Betel quid chewing, cigarette smoking and alcohol consumption related to oral cancer in Taiwan. J Oral Pathol Med. 1995;24(10):450–3.
Lee SS, Yang SF, Ho YC, Tsai CH, Chang YC. The upregulation of metallothionein-1 expression in areca quid chewing-associated oral squamous cell carcinomas. Oral Oncol. 2008;44(2):180–6.
Estruch R, Sacanella E, Badia E, Antunez E, Nicolas JM, Fernandez-Sola J, et al. Different effects of red wine and gin consumption on inflammatory biomarkers of atherosclerosis: a prospective randomized crossover trial. Effects of wine on inflammatory markers. Atherosclerosis. 2004;175(1):117–23.
Haddy N, Sass C, Maumus S, Marie B, Droesch S, Siest G, et al. Biological variations, genetic polymorphisms and familial resemblance of TNF-alpha and IL-6 concentrations: STANISLAS cohort. Eur J Hum Genet. 2005;13(1):109–17.
Hsu HJ, Chang KL, Yang YH, Shieh TY. The effects of arecoline on the release of cytokines using cultured peripheral blood mononuclear cells from patients with oral mucous diseases. Kaohsiung J Med Sci. 2001;17(4):175–82.
Jeng JH, Wang YJ, Chiang BL, Lee PH, Chan CP, Ho YS, et al. Roles of keratinocyte inflammation in oral cancer: regulating the prostaglandin E2, interleukin-6 and TNF-alpha production of oral epithelial cells by areca nut extract and arecoline. Carcinogenesis. 2003;24(8):1301–15.
Lambert C, McCue J, Portas M, Ouyang Y, Li J, Rosano TG, et al. Acrolein in cigarette smoke inhibits T-cell responses. J Allergy Clin Immunol. 2005;116(4):916–22.
Volpato S, Pahor M, Ferrucci L, Simonsick EM, Guralnik JM, Kritchevsky SB, et al. Relationship of alcohol intake with inflammatory markers and plasminogen activator inhibitor-1 in well-functioning older adults: the Health, Aging, and Body Composition study. Circulation. 2004;109(5):607–12.
Mian MF, Lauzon NM, Stampfli MR, Mossman KL, Ashkar AA. Impairment of human NK cell cytotoxic activity and cytokine release by cigarette smoke. J Leukoc Biol. 2008;83(3):774–84.
Acknowledgments
We gratefully acknowledge our patients, their relatives, and clinicians for their support and cooperation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
KB, PS, US, SH, SB, AP, VS, AS, AS, PA, MB, BDB, and RM have no conflict of interest to report.
Funding
This study was funded by grant no. DST SR/SO/HS/0041/2011 from the Government of India to MB and core funds from ICPO (ICMR), Noida, India.
Ethical approval and informed consent
The study procedures were approved by the institutional ethical committee (approval no. ICPO/IEC/P-003/2011), Noida, India. Informed consent was obtained from all participating individuals.
Additional information
K. Bandil and P. Singhal contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bandil, K., Singhal, P., Sharma, U. et al. Impacts of TNF-LTA SNPs/Haplotypes and Lifestyle Factors on Oral Carcinoma in an Indian Population. Mol Diagn Ther 20, 469–480 (2016). https://doi.org/10.1007/s40291-016-0215-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-016-0215-2