Abstract
Background and Objectives
We aimed to study the impact of size, maturation and cytochrome P450 2D6 (CYP2D6) genotype activity score as predictors of intravenous tramadol disposition.
Methods
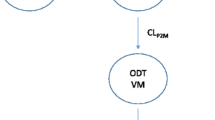
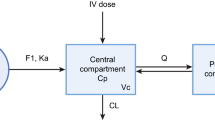
Tramadol and O-desmethyl tramadol (M1) observations in 295 human subjects (postmenstrual age 25 weeks to 84.8 years, weight 0.5–186 kg) were pooled. A population pharmacokinetic analysis was performed using a two-compartment model for tramadol and two additional M1 compartments. Covariate analysis included weight, age, sex, disease characteristics (healthy subject or patient) and CYP2D6 genotype activity. A sigmoid maturation model was used to describe age-related changes in tramadol clearance (CLPO), M1 formation clearance (CLPM) and M1 elimination clearance (CLMO). A phenotype-based mixture model was used to identify CLPM polymorphism.
Results
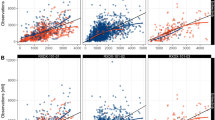
Differences in clearances were largely accounted for by maturation and size. The time to reach 50 % of adult clearance (TM50) values was used to describe maturation. CLPM (TM50 39.8 weeks) and CLPO (TM50 39.1 weeks) displayed fast maturation, while CLMO matured slower, similar to glomerular filtration rate (TM50 47 weeks). The phenotype-based mixture model identified a slow and a faster metabolizer group. Slow metabolizers comprised 9.8 % of subjects with 19.4 % of faster metabolizer CLPM. Low CYP2D6 genotype activity was associated with lower (25 %) than faster metabolizer CLPM, but only 32 % of those with low genotype activity were in the slow metabolizer group.
Conclusions
Maturation and size are key predictors of variability. A two-group polymorphism was identified based on phenotypic M1 formation clearance. Maturation of tramadol elimination occurs early (50 % of adult value at term gestation).






Similar content being viewed by others
References
Grond S, Sablotzki A. Clinical pharmacology of tramadol. Clin Pharmacokinet. 2004;43(13):879–923.
Reeves RR, Burke RS. Tramadol: basic pharmacology and emerging concepts. Drugs Today. 2008;44(11):827–36.
Ardakani YH, Rouini MR. Pharmacokinetics of tramadol and its three main metabolites in healthy male and female volunteers. Biopharm Drug Dispos. 2007;28(9):527–34.
Gong L, Stamer UM, Tzvetkov MV, et al. PharmGKB summary: tramadol pathway. Pharmacogenet Genomics. 2014;24(7):374–80.
Allegaert K, Anderson BJ, Verbesselt R, et al. Tramadol disposition in the very young: an attempt to assess in vivo cytochrome P-450 2D6 activity. Br J Anaesth. 2005;95(2):231–9.
Allegaert K, van den Anker JN, de Hoon JN, et al. Covariates of tramadol disposition in the first months of life. Br J Anaesth. 2008;100(4):525–32.
Pedersen RS, Damkier P, Brosen K. Enantioselective pharmacokinetics of tramadol in CYP2D6 extensive and poor metabolizers. Eur J Clin Pharmacol. 2006;62(7):513–21.
Stamer UM, Lehnen K, Höthker F, et al. Impact of CYP2D6 genotype on postoperative tramadol analgesia. Pain. 2003;105(1–2):231–8.
Murthy BV, Pandya KS, Booker PD, et al. Pharmacokinetics of tramadol in children after i.v. or caudal epidural administration. Br J Anaesth. 2000;84(3):346–9.
Garrido MJ, Habre W, Rombout F, et al. Population pharmacokinetic/pharmacodynamic modelling of the analgesic effects of tramadol in pediatrics. Pharm Res. 2006;23(9):2014–23.
Bressolle F, Rochette A, Khier S, et al. Population pharmacokinetics of the two enantiomers of tramadol and O-demethyl tramadol after surgery in children. Br J Anaesth. 2009;102(3):390–9.
Lintz W, Barth H, Osterloh G, et al. Bioavailability of enteral tramadol formulations. 1st communication: capsules. Arzneimittelforschung. 1986;36(8):1278–83.
Lintz W, Barth H, Becker R, et al. Pharmacokinetics of tramadol and bioavailability of enteral tramadol formulations. 2nd communication: drops with ethanol. Arzneimittelforschung. 1998;48(5):436–45.
Lintz W, Barth H, Osterloh G, et al. Pharmacokinetics of tramadol and bioavailability of enteral tramadol formulations. 3rd communication: suppositories. Arzneimittelforschung. 1998;48(9):889–99.
Lintz W, Beier H, Gerloff J. Bioavailability of tramadol after i.m. injection in comparison to i.v. infusion. Int J Clin Pharmacol Ther. 1999;37(4):175–83.
Lintz W, Becker R, Gerloff J, et al. Pharmacokinetics of tramadol and bioavailability of enteral tramadol formulations. 4th communication: drops (without ethanol). Arzneimittelforschung. 2000;50(2):99–108.
Stamer UM, Musshoff F, Kobilay M, et al. Concentrations of tramadol and O-desmethyltramadol enantiomers in different CYP2D6 genotypes. Clin Pharmacol Ther. 2007;82(1):41–7.
Abdel-Rahman SM, Leeder JS, Wilson JT, et al. Concordance between tramadol and dextromethorphan parent/metabolite ratios: the influence of CYP2D6 and non-CYP2D6 pathways on biotransformation. J Clin Pharmacol. 2002;42(1):24–9.
Blake MJ, Gaedigk A, Pearce RE, et al. Ontogeny of dextromethorphan O- and i-demethylation in the first year of life. Clin Pharmacol Ther. 2007;81(4):510–6.
Becker R, Lintz W. Determination of tramadol in human serum by capillary gas chromatography with nitrogen-selective detection. J Chromatogr. 1986;377:213–20.
Gan SH, Ismail R. Validation of a high-performance liquid chromatography method for tramadol and O-desmethyltramadol in human plasma using solid-phase extraction. J Chromatogr B. 2001;759(2):325–35.
Nobilis M, Kopecky J, Kvetina J, et al. High-performance liquid chromatographic determination of tramadol and its O-desmethylated metabolite in blood plasma: application to a bioequivalence study in humans. J Chromatogr A. 2002;949(1–2):11–22.
KuKanich B, Papich MG. Pharmacokinetics of tramadol and the metabolite O-desmethyltramadol in dogs. J Vet Pharmacol Ther. 2004;27(4):239–46.
Giorgi M, Del Carlo S, Saccomanni G, et al. Pharmacokinetics of tramadol and its major metabolites following rectal and intravenous administration in dogs. N Z Vet J. 2009;57(3):146–52.
Giorgi M, Soldani G, Manera C, et al. Pharmacokinetics of tramadol and its metabolites M1, M2 and M4 in horses following intravenous, immediate release (fasted/fed) and sustained release single dose administration. J Equine Vet Sci. 2007;27(11):481–8.
Holford S, Allegaert K, Anderson BJ, et al. Parent-metabolite pharmacokinetic models for tramadol: tests of assumptions and predictions. J Pharmacol Clin Toxicol. 2014;2(1):1023.
Parke J, Holford NH, Charles BG. A procedure for generating bootstrap samples for the validation of nonlinear mixed-effects population models. Comput Methods Programs Biomed. 1999;59(1):19–29.
Holford NH. The visual predictive check: superiority to standard diagnostic (Rorschach) plots (http://www.page-meeting.org/?abstract=972). PAGE 2005;14:972.
Bergstrand M, Hooker AC, Wallin JE, et al. Prediction-corrected visual predictive checks for diagnosing nonlinear mixed-effects models. AAPS J. 2011;13(2):143–51.
Anderson BJ, Holford NH. Mechanism-based concepts of size and maturity in pharmacokinetics. Annu Rev Pharmacol Toxicol. 2008;48:303–32.
West GB, Brown JH, Enquist BJ. A general model for the origin of allometric scaling laws in biology. Science. 1997;276(5309):122.
Tod M, Lokiec F, Bidault R, et al. Pharmacokinetics of oral acyclovir in neonates and in infants: a population analysis. Antimicrob Agents Chemother. 2001;45(1):150–7.
Anderson BJ, Allegaert K, Holford NH. Population clinical pharmacology of children: general principles. Eur J Pediatr. 2006;165(11):741–6.
Tod M, Jullien V, Pons G. Facilitation of drug evaluation in children by population methods and modelling. Clin Pharmacokinet. 2008;47(4):231–43.
Karlsson MO, Jonsson NE, Wiltse CG, et al. Assumption testing in population pharmacokinetic models: illustrated with an analysis of moxonidine data from congestive heart failure patients. J Pharmacokinet Biopharm. 1998;26(2):207–46.
Rhodin MM, Anderson BJ, Peters AM, et al. Human renal function maturation: a quantitative description using weight and postmenstrual age. Pediatr Nephrol. 2009;24(1):67–76.
Allegaert K, Rochette A, Veyckemans F. Developmental pharmacology of tramadol during infancy: ontogeny, pharmacogenetics and elimination clearance. Paediatr Anaesth. 2011;21(3):266–73.
Anderson BJ, Holford NH. Mechanistic basis of using body size and maturation to predict clearance in humans. Drug Metab Pharmacokinet. 2009;24(1):25–36.
Alvan G, Bechtel P, Iselius L, et al. Hydroxylation polymorphisms of debrisoquine and mephenytoin in European populations. Eur J Clin Pharmacol. 1990;39(6):533–7.
Sachse C, Brockmoller J, Bauer S, et al. Cytochrome P450 2D6 variants in a Caucasian population: allele frequencies and phenotypic consequences. Am J Hum Genet. 1997;60(2):284–95.
Caraco Y. Genes and the response to drugs. N Engl J Med. 2004;351(27):2867–9.
Stamer UM, Stüber F, Muders T, et al. Respiratory depression with tramadol in a patient with renal impairment and CYP2D6 gene duplication. Anesth Analg. 2008;107(3):926–9.
Acknowledgments
The clinical research of Karel Allegaert is supported by the Fund for Scientific Research, Flanders (Belgium) (FWO Vlaanderen, 1800,214N). The clinical research of U. Stamer was supported in part by a grant of the R. Sackler Research Foundation (Germany). We are grateful to Dr. B. Kukanich and Dr. M. Giorgi for access to the data from their studies in dogs, which was essential for us to distinguish the different elimination pathways of tramadol. Horst Beier, co-author of this paper is an employee of Grünenthal, Aachen, Germany, one the manufacturers of tramadol. All other authors have no conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Allegaert, K., Holford, N., Anderson, B.J. et al. Tramadol and O-Desmethyl Tramadol Clearance Maturation and Disposition in Humans: A Pooled Pharmacokinetic Study. Clin Pharmacokinet 54, 167–178 (2015). https://doi.org/10.1007/s40262-014-0191-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-014-0191-9