Abstract
Background and Objectives
The aim of this study was to evaluate the cost effectiveness of novel oral anticoagulants (NOACs) for stroke prevention among atrial fibrillation (AF) patients by incorporating Taiwanese demographic information derived from a population-based database, the National Health Insurance Research Database (NHIRD), into cost-effectiveness analysis.
Methods
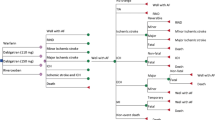
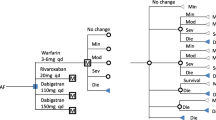
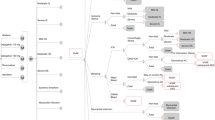
From 1 January to 31 December 2012, 98,213 AF patients were selected from the NHIRD database. A Markov model was constructed that combined published secondary data with the Taiwan NHIRD to compare the cost and incremental cost effectiveness of apixaban 5 mg twice daily, dabigatran 110 or 150 mg twice daily, rivaroxaban 20 mg once daily, and warfarin.
Results
The lifetime costs of warfarin, dabigatran 110 mg, dabigatran 150 mg, rivaroxaban 20 mg, and apixaban 5 mg were US$10,660, US$13,693, US$13,426, US$13,455, US$15,965, respectively. Apixaban resulted in an incremental cost effectiveness of US$39,351, US$27,039, US$41,298, and US$48,896 per quality-adjusted life-year (QALY) compared with warfarin, dabigatran 110 mg, dabigatran 150 mg, and rivaroxaban 20 mg, respectively. In Monte-Carlo analyses, apixaban 5 mg, rivaroxaban 20 mg, warfarin, and dabigatran 110 mg were cost effective in 83, 10.4, 7, and 0.8%, respectively, of the simulations using a willingness-to-pay (WTP) threshold of US$50,000 per QALY.
Conclusions
Apixaban was more cost effective than warfarin, dabigatran, and rivaroxaban for stroke prevention in patients with AF. Among the anticoagulant therapies, the WTP threshold of apixaban was about US$50,000 per QALY gained. These cost-effectiveness estimations provide useful information to aid clinical decision making in stroke prevention for AF patients.




Similar content being viewed by others
References
Chien KL, Su TC, Hsu HC, et al. Atrial fibrillation prevalence, incidence and risk of stroke and all-cause death among Chinese. Int J Cardiol. 2010;139:173–80.
Benjamin EJ, Wolf PA, D’Agostino RB, et al. Impact of atrial fibrillation on the risk of death: the Framingham Heart Study. Circulation. 1998;98:946–52.
Krahn AD, Manfreda J, Tate RB, et al. The natural history of atrial fibrillation: incidence, risk factors, and prognosis in the Manitoba Follow-Up Study. Am J Med. 1995;98:476–84.
Go AS, Hylek EM, Phillips KA, et al. Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention: the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA. 2001;285:2370–5.
Schnabel RB, Yin X, Gona P, et al. 50 year trends in atrial fibrillation prevalence, incidence, risk factors, and mortality in the Framingham Heart Study: a cohort study. Lancet. 2015;386(9989):154–62.
Lee CH, Liu PY, Tsai LM, et al. Characteristics of hospitalized patients with atrial fibrillation in Taiwan: a nationwide observation. Am J Med. 2007;120:819.e1–7.
Kannel WB, Benjamin EJ. Status of the epidemiology of atrial fibrillation. Med Clin North Am. 2008;92:17–40, ix.
Fuster V, Ryden LE, Cannom DS, et al. 2011 ACCF/AHA/HRS focused updates incorporated into the ACC/AHA/ESC 2006 guidelines for the management of patients with atrial fibrillation: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Circulation. 2011;123:e269–367.
January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130:e199–267.
Chen CY, Huang YB, Tzu-Chi Lee C. Epidemiology and disease burden of ischemic stroke in Taiwan. Int J Neurosci. 2013;123:724–31.
Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med. 2009;361:1139–51.
Lin LJ, Cheng MH, Lee CH, et al. Compliance with antithrombotic prescribing guidelines for patients with atrial fibrillation–a nationwide descriptive study in Taiwan. Clin Ther. 2008;30:1726–36.
Harrington AR, Armstrong EP, Nolan PE Jr, et al. Cost-effectiveness of apixaban, dabigatran, rivaroxaban, and warfarin for stroke prevention in atrial fibrillation. Stroke. 2013;44:1676–81.
Lip GYH, Kongnakorn T, Phatak H, et al. Cost-effectiveness of apixaban against other novel oral anticoagulants (NOACs) for stroke prevention in atrial fibrillation patients [abstract no. P563]. Eur Heart J. 2012;33:54.
Dorian P, Kongnakorn T, Phatak H, et al. Cost-effectiveness of apixaban vs. current standard of care for stroke prevention in patients with atrial fibrillation. Eur Heart J. 2014;35:1897–906.
Fernandez Avila Y, Garcia KC, Garrido Lecca S, et al. Cost-effectiveness of apixaban versus other new oral anticoagulants and warfarin for stroke prevention in atrial fibrillation in Venezuela [abstract no. PCV14]. Value Health. 2015;18:A829.
Tanaka S, Preto MC, Bernardino G, et al. Cost-effectiveness of apixaban versus other noacs and warfarin, during hospitalization in the private Brazilian health system [abstract no. PCV17]. Value Health. 2015;18:A830.
Yeh MJ, Chang HH. National health insurance in Taiwan [letter]. Health Aff (Millwood). 2015;34:1067.
Rubrichi S, Rognoni C, Sacchi L, et al. Graphical representation of life paths to better convey results of decision models to patients. Med Decis Making. 2015;35:398–402.
Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med. 2011;365:981–92.
Lee S, Monz BU, Clemens A, et al. Representativeness of the dabigatran, apixaban and rivaroxaban clinical trial populations to real-world atrial fibrillation patients in the United Kingdom: a cross-sectional analysis using the General Practice Research Database. BMJ Open. 2012;2:e001768.
Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med. 2011;365:883–91.
Connolly SJ, Ezekowitz MD, Yusuf S, et al. Newly identified events in the RE-LY trial. N Engl J Med. 2010;363:1875–6.
Connolly SJ, Eikelboom J, Joyner C, et al. Apixaban in patients with atrial fibrillation. N Engl J Med. 2011;364:806–17.
Gage BF, Cardinalli AB, Owens DK. The effect of stroke and stroke prophylaxis with aspirin or warfarin on quality of life. Arch Intern Med. 1996;156:1829–36.
Sullivan PW, Ghushchyan V. Preference-Based EQ-5D index scores for chronic conditions in the United States. Med Decis Making. 2006;26:410–20.
Pignone M, Earnshaw S, Pletcher MJ, et al. Aspirin for the primary prevention of cardiovascular disease in women: a cost-utility analysis. Arch Intern Med. 2007;167:290–5.
Sullivan PW, Lawrence WF, Ghushchyan V. A national catalog of preference-based scores for chronic conditions in the United States. Med Care. 2005;43:736–49.
Tengs TO, Lin TH. A meta-analysis of quality-of-life estimates for stroke. Pharmacoeconomics. 2003;21:191–200.
National Health Insurance Administration. Taiwan diagnosis-related group payment system. Ministry of Health and Welfare; Taipei City, Taiwan, R.O.C. 2015.
Zhou ZQ, Hu DY, Chen J, et al. An epidemiological survey of atrial fibrillation in China [in Chinese]. Zhonghua Nei Ke Za Zhi. 2004;43:491–4.
Ohsawa M, Okayama A, Okamura T, et al. Mortality risk attributable to atrial fibrillation in middle-aged and elderly people in the Japanese general population: nineteen-year follow-up in NIPPON DATA80. Circ J. 2007;71:814–9.
Kannel WB, Wolf PA, Benjamin EJ, et al. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol. 1998;82:2N–9N.
Coyle D, Coyle K, Cameron C, et al. Cost-effectiveness of new oral anticoagulants compared with warfarin in preventing stroke and other cardiovascular events in patients with atrial fibrillation. Value Health. 2013;16:498–506.
Yu ST, Chang HY, Yao KP, Lin YH, Hurng BS. The reliability and validity of EQ-5D in Taiwan: results from 2009 National Health Interview Survey. Zhu-Nan, Taiwan, R.O.C. 2010.
Kamel H, Easton JD, Johnston SC, et al. Cost-effectiveness of apixaban vs warfarin for secondary stroke prevention in atrial fibrillation. Neurology. 2012;79:1428–34.
Tanahashi N, Hori M, Matsumoto M, et al. Rivaroxaban versus warfarin in Japanese patients with nonvalvular atrial fibrillation for the secondary prevention of stroke: a subgroup analysis of J-ROCKET AF. J Stroke Cerebrovasc Dis. 2013;22:1317–25.
Kamae I, Hashimoto Y, Koretsune Y, et al. Cost-effectiveness analysis of apixaban against warfarin for stroke prevention in patients with nonvalvular atrial fibrillation in Japan. Clin Ther. 2015;37:2837–51.
Krejczy M, Harenberg J, Marx S, et al. Comparison of cost-effectiveness of anticoagulation with dabigatran, rivaroxaban and apixaban in patients with non-valvular atrial fibrillation across countries. J Thromb Thrombolysis. 2014;37:507–23.
Owens DK. Interpretation of cost-effectiveness analyses. J Gen Intern Med. 1998;13:716–7.
Acknowledgements
The authors thank the National Health Research Institutes (NHRI) for providing the National Health Insurance Research Database (NHIRD). The study data were partly retrieved from the NHIRD, which is managed by the National Health Insurance Administration (NHIA), Ministry of Health and Welfare, and maintained by the NHRI in Taiwan. The results, interpretation, and conclusions of this paper do not represent the opinions of the NHIA, Ministry of Health and Welfare, or the NHRI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was sponsored by Pfizer (Taiwan), grant number DN-PC-100-015.
Conflict of interest
Neither of the authors is an employee of Pfizer (Taiwan). Chieh-Yu Liu is a full-time associate professor in the Department of Midwifery and Women Health Care, National Taipei University of Nursing and Health Sciences, and has received research grants from Pfizer Taiwan Inc. Hui-Chun Chen is a graduate student in the Department of Nursing, National Taipei University of Nursing and Health Sciences.
Ethical approval
Due to the nature of secondary data analysis and using previously collected and anonymous data, all data analysis procedures conducted in this paper were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and this study was classified as being in the Exempt Review category.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, CY., Chen, HC. Cost-Effectiveness Analysis of Apixaban, Dabigatran, Rivaroxaban, and Warfarin for Stroke Prevention in Atrial Fibrillation in Taiwan. Clin Drug Investig 37, 285–293 (2017). https://doi.org/10.1007/s40261-016-0487-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-016-0487-7