Abstract
Background
Amino acids (AAs) and acylcarnitines play a key role in metabolic disease and can be used as biomarkers of various diseases such as malignancies, type 2 diabetes (T2D), insulin resistance, and cardiovascular diseases, therefore, designing an accurate and simple laboratory method that simultaneously measure both groups of substances, could improve the process of analytes quantification. In this research, a flow injection tandem mass spectrometry (FI-MS/MS) method for simultaneous measurement of AAs and acylcarnitines in addition to results of validation is explained.
Methods
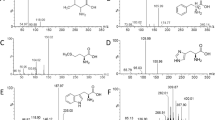
Samples were mixed with internal standards and after derivatization (with butanolic-HCL), AAs, and acylcarnitines were quantified by tandem mass spectrometry (SCIEX API 3200). Analytical performance studies were designed based on the Clinical and Laboratory Standards Institute (CLSI) guidelines including precision, accuracy, linearity, and limit of detection-quantification (LOD-LOQ) experiments. Samples from patients with T2D in different stages of kidney disease were also analyzed to ensure the clinical usage of the method.
Results
Performance evaluation of the method demonstrated adequate results. The mean of estimated inter-assay precision (reported as a coefficient variation) for AAs and acylcarnitines were less than 8.7% and 12.3%, the estimated mean bias was below 8.8% and 10.2% respectively. LOD of analytes ranged between 0.6–10 μmol per liter (μmol/L) for AAs and 0.02-1 μmol/L for acylcarnitines. LOQ analytes showed a range of 2–25 μmol/L and 0.05–5 μmol/L for AAs and carnitine/acylcarnitines respectively. In diabetic patients sample analysis, a significant increase in acylcarnitines (C2, C4, C5DC, C6, C8, C10, C14) and citrulline with a significant decrease in valine were seen in patients with severely increased albuminuria.
Conclusion
FI-MS/MS method with pre-injection derivatization with butanolic-HCL can be used for concurrent measurement of AAs and carnitine/acylcarnitines in a short time and it satisfies the analytical performance requirements. This method is applied for AAs and carnitine/acylcarnitines measurement in patient with T2DM and results show some of the acylcarnitines and AAs can be involved in diabetic nephropathy development.
Similar content being viewed by others
Data availability
The dataset supporting the conclusions of this article is available and can be accessed by contacting the corresponding author.
References
Bujak R, Struck-Lewicka W, Markuszewski MJ, Kaliszan R. Metabolomics for laboratory diagnostics. J Pharm Biomed Anal. 2015;113:108–20.
Newgard CB. Metabolomics and metabolic diseases: where do we stand? Cell Metab. 2017;25(1):43–56.
Wu G. Amino acids: metabolism, functions, and nutrition. Amino Acids. 2009;37(1):1–17.
Gannon NP, Schnuck JK, Vaughan RA. BCAA metabolism and insulin sensitivity–Dysregulated by metabolic status? Mol Nutr Food Res. 2018;62(6):1700756.
Nie C, He T, Zhang W, Zhang G, Ma X. Branched chain amino acids: beyond nutrition metabolism. Int J Mol Sci. 2018;19(4):954.
Bene J, Márton M, Mohás M, Bagosi Z, Bujtor Z, Oroszlán T, et al. Similarities in serum acylcarnitine patterns in type 1 and type 2 diabetes mellitus and in metabolic syndrome. Ann Nutr Metab. 2013;62(1):80–5.
Mansour FR, Wei W, Danielson ND. Separation of carnitine and acylcarnitines in biological samples: a review. Biomed Chromatogr. 2013;27(10):1339–53.
Mingrone G. Carnitine in type 2 diabetes. Ann N Y Acad Sci. 2004;1033(1):99–107.
Ahmed AT, MahmoudianDehkordi S, Bhattacharyya S, Arnold M, Liu D, Neavin D, et al. Acylcarnitine metabolomic profiles inform clinically-defined major depressive phenotypes. J Affect Disord. 2020;264:90–7.
Jones LL, McDonald DA, Borum PR. Acylcarnitines: role in brain. Prog Lipid Res. 2010;49(1):61–75.
Guasch-Ferré M, Zheng Y, Ruiz-Canela M, Hruby A, Martínez-González MA, Clish CB, et al. Plasma acylcarnitines and risk of cardiovascular disease: effect of Mediterranean diet interventions. Am J Clin Nutr. 2016;103(6):1408–16.
Kalim S, Clish CB, Wenger J, Elmariah S, Yeh RW, Deferio JJ, et al. A plasma long-chain acylcarnitine predicts cardiovascular mortality in incident dialysis patients. J Am Heart Assoc. 2013;2(6):e000542.
Xu W, Zhong C, Zou C, Wang B, Zhang N. Analytical methods for amino acid determination in organisms. Amino Acids. 2020:1–18.
de Sousa C, English NR, Stacey TE, Chalmers RA. Measurement of L-carnitine and acylcarnitines in body fluids and tissues in children and in adults. Clin Chim Acta. 1990;187(3):317–28.
Kerner J, Bieber L. A radioisotopic-exchange method for quantitation of short-chain (acid-soluble) acylcarnitines. Anal Biochem. 1983;134(2):459–66.
Bieber L, Lewin L. [13] Measurement of carnitine and O-acylcarnitines. Methods in enzymology. 72: Elsevier; 1981. p. 276–87.
Sargent M. Guide to achieving reliable quantitative LC-MS measurements. RSC analytical methods committee 2013.
Awad H, Khamis MM, El-Aneed A. Mass spectrometry, review of the basics: ionization. Appl Spectrosc Rev. 2015;50(2):158–75.
Banerjee S, Mazumdar S. Electrospray ionization mass spectrometry: a technique to access the information beyond the molecular weight of the analyte. International journal of analytical chemistry. 2012;2012:1–40.
El-Aneed A, Cohen A, Banoub J. Mass spectrometry, review of the basics: electrospray, MALDI, and commonly used mass analyzers. Appl Spectrosc Rev. 2009;44(3):210–30.
Lane C. Mass spectrometry-based proteomics in the life sciences. Cellular and Molecular Life Sciences CMLS. 2005;62(7–8):848–69.
Nanita SC, Kaldon LG. Emerging flow injection mass spectrometry methods for high-throughput quantitative analysis. Anal Bioanal Chem. 2016;408(1):23–33.
Sarvin B, Lagziel S, Sarvin N, Mukha D, Kumar P, Aizenshtein E, Shlomi T Fast and sensitive flow-injection mass spectrometry metabolomics by analyzing sample-specific ion distributions. Nat Commun 2020;11(1):1–11, 3186.
Gaugler SC, Rykl J, Cebolla VL. Validation of an automated extraction procedure for amino acids and acylcarnitines for use with tandem mass spectrometry for newborn screening. 2019.
Ni J, Xu L, Li W, Wu L. Simultaneous determination of thirteen kinds of amino acid and eight kinds of acylcarnitine in human serum by LC–MS/MS and its application to measure the serum concentration of lung cancer patients. Biomed Chromatogr. 2016;30(11):1796–806.
Teav T, Gallart-Ayala H, van der Velpen V, Mehl F, Henry H, Ivanisevic J. Merged targeted quantification and untargeted profiling for comprehensive assessment of acylcarnitine and amino acid metabolism. Anal Chem. 2019;91(18):11757–69.
Corso G, Paglia G, Garofalo D, D'apolito O. Neutral loss analysis of amino acids by desorption electrospray ionization using an unmodified tandem quadrupole mass spectrometer. Rapid Communications in Mass Spectrometry: An International Journal Devoted to the Rapid Dissemination of Up-to-the-Minute Research in Mass Spectrometry. 2007;21(23):3777–84.
Sharma G, Attri SV, Behra B, Bhisikar S, Kumar P, Tageja M, et al. Analysis of 26 amino acids in human plasma by HPLC using AQC as derivatizing agent and its application in metabolic laboratory. Amino Acids. 2014;46(5):1253–63.
Rebane R, Oldekop M-L, Herodes K. Comparison of amino acid derivatization reagents for LC–ESI-MS analysis. Introducing a novel phosphazene-based derivatization reagent. J Chromatogr B. 2012;904:99–106.
de Puit M, Ismail M, Xu X. LCMS analysis of fingerprints, the amino acid profile of 20 donors. J Forensic Sci. 2014;59(2):364–70.
Oldekop M-L, Rebane R, Herodes K. Dependence of matrix effect on ionization polarity during LC–ESI–MS analysis of derivatized amino acids in some natural samples. European Journal of Mass Spectrometry. 2017;23(5):245–53.
Casetta B, Tagliacozzi D, Shushan B, Federici G. Development of a method for rapid quantitation of amino acids by liquid chromatography-tandem mass spectrometry (LC-MSMS) in plasma. Clin Chem Lab Med. 2000;38(5):391–401.
Miller MJ, Cusmano-Ozog K, Oglesbee D, Young S. Laboratory analysis of acylcarnitines. Update: a technical standard of the American College of Medical Genetics and Genomics (ACMG). Genetics in Medicine. 2020;2020:1–10.
Li S, Gao D, Jiang Y. Function, detection and alteration of acylcarnitine metabolism in hepatocellular carcinoma. Metabolites. 2019;9(2):36.
Harder U, Koletzko B, Peissner W. Quantification of 22 plasma amino acids combining derivatization and ion-pair LC–MS/MS. J Chromatogr B. 2011;879(7–8):495–504.
Carling RS, John K, Churchus R, Turner C, Dalton RN. Validation of a rapid, comprehensive and clinically relevant amino acid profile by underivatised liquid chromatography tandem mass spectrometry. Clinical Chemistry and Laboratory Medicine (CCLM). 2020;58(5):758–68.
Perich C, Minchinela J, Ricós C, Fernández-Calle P, Alvarez V, Doménech MV, et al. Biological variation database: structure and criteria used for generation and update. Clinical Chemistry and Laboratory Medicine (CCLM). 2015;53(2):299–305.
Westgard Q. Biological variation database, and quality specifications for imprecision, bias and total error (desirable and minimum). The 2014 update. 2014.
Lau T, Owen W, Yu YM, Noviski N, Lyons J, Zurakowski D, et al. Arginine, citrulline, and nitric oxide metabolism in end-stage renal disease patients. J Clin Invest. 2000;105(9):1217–25.
Mai M, Tönjes A, Kovacs P, Stumvoll M, Fiedler GM, Leichtle AB. Serum levels of acylcarnitines are altered in prediabetic conditions. PLoS One. 2013;8(12):e82459.
Rahimi N, Razi F, Nasli-Esfahani E, Qorbani M, Shirzad N, Larijani B. Amino acid profiling in the gestational diabetes mellitus. Journal of Diabetes & Metabolic Disorders. 2017;16(1):13.
Sun L, Liang L, Gao X, Zhang H, Yao P, Hu Y, et al. Early prediction of developing type 2 diabetes by plasma acylcarnitines: a population-based study. Diabetes Care. 2016;39(9):1563–70.
Chuang W-H, Arundhathi A, Lu C, Chen C-C, Wu W-C, Susanto H, et al. Altered plasma acylcarnitine and amino acid profiles in type 2 diabetic kidney disease. Metabolomics. 2016;12(6):108.
Goek O-N, Döring A, Gieger C, Heier M, Koenig W, Prehn C, et al. Serum metabolite concentrations and decreased GFR in the general population. Am J Kidney Dis. 2012;60(2):197–206.
Fouque D, Holt S, Guebre-Egziabher F, Nakamura K, Vianey-Saban C, Hadj-Aïssa A, et al. Relationship between serum carnitine, acylcarnitines, and renal function in patients with chronic renal disease. J Ren Nutr. 2006;16(2):125–31.
Funding
This study is funded by Endocrinology and Metabolism Research Institute.
Author information
Authors and Affiliations
Contributions
All authors participated and approved the paper before submission. FR planned the study, NN, AK, and ShH did the analytical examination and analyzed the result, AS contributed in optimization and supervision of systems, BA and SE participated in the study design, PE contributed in technical issues, statistical analysis and preparation of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Endocrinology and Metabolism Research Institute, Tehran University of Medical Sciences.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Esmati, P., Najjar, N., Emamgholipour, S. et al. Mass spectrometry with derivatization method for concurrent measurement of amino acids and acylcarnitines in plasma of diabetic type 2 patients with diabetic nephropathy. J Diabetes Metab Disord 20, 591–599 (2021). https://doi.org/10.1007/s40200-021-00786-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40200-021-00786-3