Abstract
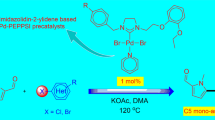
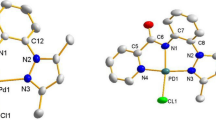
The present work describes the synthesis of palladium(II) complex with non-chelating 4-pyridylselenolate ligand as [PdCl(4-SeC5H4N)(PPh3)2]. The catalyst was found to be active for aminocarbonylation reaction of aryl and hetero aryl iodides with primary amines using carbon monoxide a simple and efficient C1 source. The catalytic system was optimized with respect to various reaction parameters to give excellent yield of desired products. The milder reaction conditions and wild functional groups tolerance enhance its practical applicability.

Similar content being viewed by others
References
Beller M (2006) Catalytic carbonylation reaction. Springer-Verlag, Berlin
Wu X-F, Neumann H, Beller M (2013) Synthesis of heterocycles via Palladium-Catalyzed carbonylations. Chem Rev 113:1–35
Zeni G, Larock RC (2004) Synthesis of Heterocycles via Palladium π-Olefin and π-Alkyne chemistry. Chem Rev 104:2285–2309
Khumtaveeporn K, Alper H (1995) Transition metal mediated carbonylative ring expansion of heterocyclic compounds. Acc Chem Res 28:414–422
Gadge ST, Bhanage BM (2014) Recent developments in palladium catalysed carbonylation reactions. RSC Adv 4:10367–10389
Friis SD, Lindhardt AT, Skrydstrup T (2016) The development and application of two-chamber reactors and carbon monoxide precursors for safe carbonylation reactions. Acc Chem Res 49:594–605
Colquhoun HM, Thompson DJ, Twigg MV (1991) Carbonylation, direct synthesis of carbonyl compounds. Plenum Press, New York
Beller M, Wu XF (2013) Transition metal catalyzed carbonylation reactions: carbonylative activation of C–X bonds. Springer, Heidelberg
Wu X-F, Neumann H, Beller M (2011) Palladium-catalyzed carbonylative coupling reactions between Ar-X and carbon nucleophiles. Chem Soc Rev 40:4986–5009
Surry DS, Buchwald SL (2011) Dialkylbiaryl phosphines in Pd-catalyzed amination: a user’s guide. Chem Sci 2:27–50
The role of metals and ligands in organic hydroformylation
Balakrishna MS, Reddy VS, Krishnamurthy SS, Burckette St Laurent JCTR, Nixon JF (1994) Coordination chemistry of diphosphinoamine and cyclodiphosphazane ligands. Coord Chem Rev 129:1–90
Kumar A, Rao GK, Saleem F, Singh AK (2012) Organoselenium ligands in catalysis. Dalton Trans 41:11949–11977
Ritch JS, Charette BJ (2016) An experimental and computational comparison of phosphorus- and selenium-based ligands for catalysis. Can J Chem 94:1–6
Kedarnath G, Jain VK (2013) Pyridyl and pyrimidyl chalcogen (Se and Te) compounds: a family of multi utility molecules. Coord Chem Rev 257:1409–1435
Paluru DK, Dey S, Wadawale A, Maity DK, Bhuvanesh N, Jain VK (2015) Structural variation in [PdX2{RE(CH2)nNMe2}] (E = Se, Te; X = Cl, OAc) complexes: experimental results, computational analysis, and catalytic activity in suzuki coupling reactions. Eur J Inorg Chem 2015:397–407
Sharma KN, Joshi H, Prakash O, Sharma AK, Bhaskar R, Singh AK (2015) Pyrazole–Stabilized Dinuclear Palladium(II) Chalcogenolates Formed by Oxidative Addition of Bis [2-(4-bromopyrazol-1-yl)ethyl] Dichalcogenides to Palladium (II)–Tailoring of Pd–S/Se Nanoparticles. Eur J Inorg Chem 29:4829–4838
Vivekananda KV, Dey S, Adawale A, Bhuvanesh N, Jain VK (2013) Syntheses of Pd(II)/Pt(II) complexes with non-chelatingn 4-pyridylselenolate ligand ranging from mononuclear to macrocyclic structures and their utility as catalysts in Suzuki C–C coupling reaction. Dalton Trans 42:14158–14167
Pattabiraman VR, Bode JW (2011) Rethinking amide bond synthesis. Nature 480:471
Castanho M, Santos N (2011) Peptide drug discovery and development. Wiley, Weinheim
Larock RC (1999) Comprehensive organic transformations, 2nd edn. Wiley, New York
Chen YH, Zhang YH, Zhang HJ, Liu DZ, Gu M, Li JY, Wu F, Zhu XZ, Li J, Nan FJ (2006) Design, synthesis, and biological evaluation of isoquinoline-1,3,4-trione derivatives as potent caspase-3 inhibitors. J Med Chem 49:1613–1623
Slee DH, Laslo KL, Elder JH, Ollmann IR, Gustchina A, Kervinen J, Zdanov A, Wlodawer A, Wong CH (1995) Selectivity in the inhibition of HIV and FIV Protease: inhibitory and mechanistic studies of pyrrolidine-containing. alpha.-Keto amide and hydroxyethylamine core structures. J Am Chem Soc 117:11867
Allen CL, Williams JM (2011) Metal-catalysed approaches to amide bond formation. J Chem Soc Rev 40:3405–3415
Valeur E, Bradley M (2009) Amide bond formation: beyond the myth of coupling reagents. Chem Soc Rev 38:606–631
Gooben LJ, Huang L, Arndt M, Gooben K, Heydt H (2015) Late transition metal-catalyzed hydroamination and hydroamidation. Chem Rev 115:2596
Yedage SL, D’Silva DS, Bhanage BM (2015) MnO2 catalyzed formylation of amines and transamidation of amides under solvent-free conditions. RSC Adv 5:80441
Sawant DN, Wagh YS, Bhatte KD, Bhanage BM (2011) Palladium-catalyzed carbon-monoxide-free aminocarbonylation of aryl halides using N-substituted formamides as an amide source. J Org Chem 76:5489
Qureshi ZS, Revankar SA, Khedkar MV, Bhanage BM (2012) Aminocarbonylation of aryl iodides with primary and secondary amines in aqueous medium using polymer supported palladium-N-heterocyclic carbine complex as an efficient and heterogeneous recyclable catalyst. Catal Today 198:148–153
Schoenberg A, Heck RF (1974) Palladium-catalyzed amidation of aryl, heterocyclic, and vinylic halides. J Org Chem 39:3327–3330
Martinelli JR, Clark TP, Watson DA, Munday RH, Buchwald SL (2007) Palladium-catalyzed aminocarbonylation of aryl chlorides at atmospheric pressure: the dual role of sodium phenoxide. Angew Chem Int Ed 46:8460–8463
Skoda-Foldes R, Takacs E, Horvath J, Tuba Z, Kollar L (2003) Palladium-catalysed aminocarbonylation of steroidal 17-iodo-androst-16-ene derivatives in N, N′-dialkyl-imidazolium-type ionic liquids. Green Chem 5:643–645
Khedkar MV, Sasaki T, Bhanage BM (2013) Immobilized palladium metal-containing ionic liquid-catalyzed alkoxycarbonylation, phenoxycarbonylation, and aminocarbonylation reactions. ACS Catal 3:287–293
Tambade PJ, Patil YP, Bhanage BM (2009) Palladium bis(2,2,6,6-tetramethyl-3,5-heptanedionate) catalyzed alkoxycarbonylation and aminocarbonylation reactions. Appl Organometal Chem 23:235–240
Mane RS, Bhanage BM (2016) Pd/C-Catalyzed aminocarbonylation of aryl iodides via oxidative C–N bond activation of tertiary amines to tertiary amides. J Org Chem 81:1223–1228
Tambade PJ, Patil YP, Bhanushali MJ, Bhanage BM (2008) Pd(OAc)2-Catalyzed aminocarbonylation of aryl iodides with aromatic or aliphatic amines in water. Synthesis 15:2347–2352
Boduszek B, Gancarz R (1996) Pyridine-4-selenenyl bromides as new reagents for selenenylation of olefins. J Prakt Chem 338:186–189
Dey S, Jain VK, Knodler A, Klein A, Kaim W, Zalis S (2002) Structural basis for unusually long-wavelength charge transfer transitions in complexes [MCl(ECH2CH2NMe2)(PR3)] (E = Te, Se; M = Pt, Pd): experimental results and TD-DFT calculations. Inorg Chem 41:2864–2870
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chavan, S.P., Dey, S., Jain, V.K. et al. Palladium(II) Complex of 4-Pyridylselenolate Ligand: An Efficient Catalyst for Aminocarbonylation of Aryl and Hetero Aryl Iodides with Primary Amines. Proc. Natl. Acad. Sci., India, Sect. A Phys. Sci. 86, 581–587 (2016). https://doi.org/10.1007/s40010-016-0291-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40010-016-0291-z