Abstract
Purpose
Methicillin-resistant Staphylococcus aureus (MRSA) decolonization is a widely established, though controversial part of many MRSA controlling strategies. The aim of this study was to evaluate our decolonization success rate, identify the risk factors for decolonization failure and determine the optimal duration of follow-up in our low MRSA prevalence setting (2.6 % of isolates).
Methods
Every patient with newly detected MRSA colonization or infection between January 2007 and December 2009 was recruited to the study. The MRSA strategy of our institution (a 700 bed tertiary hospital in eastern Switzerland) consists of a 5-day regimen of nasal mupirocin ointment, chlorhexidin mouth rinse and whole body wash with didecyldimonium chloride. Systemic antibiotics are usually not added to the regimen.
Results
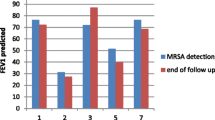
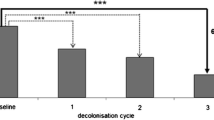
We determined a MRSA decolonization success rate of 65 % (33/51) after a median follow-up of 13 months [i.e. a tripling of the spontaneous clearance rate of 22 % (6/27) in the non-decolonized group]. The most important risk factor for decolonization failure was colonization of the respiratory tract [odds risk (OR) 9.1, 95 % confidence interval (CI) 1.2–66.7], as well as isolation of MRSA spa-type 002 ([R 5.8, 95 % CI 1.0–33.3). Of all the episodes of MRSA recurrence, 88 % (14/16) were detected within 270 days after decolonization.
Conclusion
High MRSA decolonization success rates can be achieved without the routine use of oral antibiotics. A time period of 1 year after decolonization seems to be a reasonable duration of follow-up in our setting. We encourage other institutions to take into account local MRSA epidemiology (e.g. predominance of certain subtypes) for the management of MRSA patients.



Similar content being viewed by others
References
Huang S, Platt R. Risk of methicillin-resistant Staphylococcus aureus infection after previous infection or colonization. Clin Infect Dis. 2003;36:281–5.
Boyce JM. MRSA patients: proven methods to treat colonization and infection. J Hosp Infect. 2001;48[Suppl A]:S9–14.
Loeb M, Main C, Walker-Dilks C, Eady A. Antimicrobial drugs for treating methicillin-resistant Staphylococcus aureus colonization. Cochrane Database Syst Rev. 2003;(4):CD003340.
Lucet JC, Regnier B. Screening and decolonization: does methicillin-susceptible Staphylococcus aureus hold lessons for methicillin-resistant S. aureus? Clin Infect Dis. 2010;51(5):585–90.
Robicsek A, Beaumont JL, Thomson RB Jr, Govindarajan G, Peterson LR. Topical therapy for methicillin-resistant Staphylococcus aureus colonization: impact on infection risk. Infect Control Hosp Epidemiol. 2009;30:623–32.
Dow G, Field D, Mancuso M, Allard J. Decolonization of methicillin-resistant Staphylococcus aureus during routine hospital care: efficacy and long-term follow-up. Can J Infect Dis Med Microbiol. 2010;21(1):38–44.
Ammerlaan HS, Kluytmans JA, Wertheim HF, Nouwen JL, Bonten MJ. Eradication of methicillin-resistant Staphylococcus aureus carriage: a systematic review. Clin Infect Dis. 2009;48:922–30.
Buehlmann M, Frei R, Fenner L, Dangel M, Fluckiger U, Widmer AF. Highly effective regimen for decolonization of methicillin-resistant Staphylococcus aureus carriers. Infect Control Hosp Epidemiol. 2008;29(6):510–6.
Harbarth S, Liassine N, Dharan S, Herrault P, Auckenthaler R, Pittet D. Risk factors for persistent carriage of methicillin-resistant Staphylococcus aureus. Clin Infect Dis. 2000;31(6):1380–5.
Institut für Klinische Mikrobiologie und Immunologie (IKMI) Antibiotika-Empfindlichkeit der wichtigsten Gram-positiven Bakterien und Gram-negativen Non-Enterobacteriaceae. St. Gallen. 2007. Available at: www.ikmi.ch.
Verhoef J, Beaujean D, Blok H, Baars A, Meyler A, van der Werken C, Weersink A. A Dutch approach to methicillin-resistant Staphylococcus aureus. Eur J Clin Microbiol Infect Dis. 1999;18(7):461–6.
Witteck A, Rettenmund G, Schlegel M. MRSA admission screening in a low prevalence setting—much ado about nothing? Swiss Med Wkly. 2011;7(141):w13217.
Mathews AA, Thomas M, Appalaraju B, Jayalakshmi J. Evaluation and comparison of tests to detect methicillin-resistant Staphylococcus aureus. Indian J Pathol Microbiol. 2010;53(1):79–82.
Brown DF, Edwards DI, Hawkey PM, et al. Guidelines for the laboratory diagnosis and susceptibility testing of methicillin-resistant Staphylococcus aureus (MRSA). J Antimicrob Chemother. 2005;56(6):1000–8.
Fleisch F, Oechslin EC, Gujer AR, Ritzler E, Imhof A, Ruef C, Reinhart WH. Transregional spread of a single clone of methicillin-resistant Staphylococcus aureus between groups of drug users in Switzerland. Infection. 2005;33(4):273–7.
Fenner L, Widmer AF, Dangel M, Frei R. Distribution of spa types among meticillin-resistant Staphylococcus aureus isolates during a 6 year period at a low-prevalence University Hospital. J Med Microbiol. 2008;57(Pt 5):612–6.
Ammerlaan HS, Kluytmans JA, Berkhout H, Buiting A, de Brauwer EI, van den Broek PJ, van Gelderen P, Leenders SA, Ott A, Richter C, Spanjaard L, Spijkerman IJ, van Tiel FH, Voorn GP, Wulf MW, van Zeijl J, Troelstra A, MRSA Eradication Study Group. Eradication of carriage with methicillin-resistant Staphylococcus aureus: effectiveness of a national guideline. J Antimicrob Chemother. 2011;66(10):2409–17.
Grundmann H, Aanensen DM, van den Wijngaard CC, Spratt BG, Harmsen D, European Staphylococcal Reference Laboratory Working Group. Geographic distribution of Staphylococcus aureus causing invasive infections in Europe: a molecular-epidemiological analysis. PLoS Med. 2010;7(1):e1000215.
Pan A, Lee A, Cooper B, Chalfine A, Daikos G, Garilli S, Malhotra-Kumar S, Martinez JA, Patroni A, Harbarth S, MOSAR-04 Study Team. Risk factors for previously unknown methicillin-resistant Staphylococcus aureus (MRSA) carriage on admission to 13 surgical wards in Europe. BMC Proc. 2011;5[Suppl 6]:O85.
Tiemersma EW, Bronzwaer SL, Lyytikäinen O, Degener JE, Schrijnemakers P, Bruinsma N, Monen J, Witte W, European Antimicrobial Resistance Surveillance System Participants. Methicillin-resistant Staphylococcus aureus in Europe, 1999–2002. Emerg Infect Dis. 2004;10(9):1627–34.
Hudson LO, Murphy CR, Spratt BG, Enright MC, Terpstra L, Gombosev A, Hannah P, Mikhail L, Alexander R, Moore DF, Huang SS. Differences in methicillin-resistant Staphylococcus aureus (MRSA) strains isolated from pediatric and adult patients from hospitals in a large California County. J Clin Microbiol. 2012;50(3):573–9.
Vindel A, Cuevas O, Cercenado E, Marcos C, Bautista V, Castellares C, Trincado P, Boquete T, Pérez-Vázquez M, Marín M, Spanish Group for the Study of Staphylococcus. Methicillin-resistant Staphylococcus aureus in Spain: molecular epidemiology and utility of different typing methods. J Clin Microbiol. 2009;47(6):1620–7.
Larsson AK, Gustafsson E, Nilsson AC, Odenholt I, Ringberg H, Melander E. Duration of methicillin-resistant Staphylococcus aureus colonization after diagnosis: a four-year experience from southern Sweden. Scand J Infect Dis. 2011;43(6–7):456–62.
Ammerlaan HS, Kluytmans JA, Berkhout H, Buiting A, de Brauwer EI, van den Broek PJ, van Gelderen P, Leenders SA, Ott A, Richter C, Spanjaard L, Spijkerman IJ, van Tiel FH, Voorn GP, Wulf MW, van Zeijl J, Troelstra A, MRSA Eradication Study Group. Eradication of carriage with methicillin-resistant Staphylococcus aureus: determinants of treatment failure. J Antimicrob Chemother. 2011;66(10):2418–24.
Simor AE, Phillips E, McGeer A, Konvalinka A, Loeb M, Devlin HR, Kiss A. Randomized controlled trial of chlorhexidine gluconate for washing, intranasal mupirocin, and rifampin and doxycycline versus no treatment for the eradication of methicillin-resistant Staphylococcus aureus colonization. Clin Infect Dis. 2007;44(2):178–85.
Marschall J, Muehlemann K. Duration of MRSA-carriage, according to risk factors for acquisition. Infect Control Hosp Epidemiol. 2006;27(11):1206–12.
Acknowledgments
The MRSA diagnostic procedures were performed at the Institute of Clinical Microbiology and Immunology (IKMI) in St. Gallen, Switzerland. PFGE was performed at the Laboratory of Hospital Epidemiology, University Hospital of Zurich, Switzerland. Spa-typing was performed at the Institute for Medical Microbiology (Department of Biomedicine) in Basel, Switzerland.
Conflict of interest
There was no financial support and there is no potential conflict of interest for any of the contributing authors.
Ethical approval
No formal informed consent was obtained from patients as the evaluation was considered within the overall quality control system instituted at the hospital.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kohler, P., Bregenzer-Witteck, A., Rettenmund, G. et al. MRSA decolonization: success rate, risk factors for failure and optimal duration of follow-up. Infection 41, 33–40 (2013). https://doi.org/10.1007/s15010-012-0290-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s15010-012-0290-1