Abstract
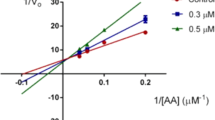
A new group of 3-substituted-5H-benzo[5,6][1,4]thiazino[3,2-e][1,2,4]triazines was designed, synthesized and evaluated as inhibitors of 15-lipoxygenase (15-LO), and the results were compared with those of standard ligand 4-methyl-2-(4-methylpiperazin-1-yl)pyrimido[4,5-b][1,4]benzothiazine (4-MMPB). Among the newly designed ligands, compound 9e showed the best IC50 of 15-LO inhibition (IC50 = 38 µM). The docking calculations were performed in MOE software based on the function of force-field scoring, in order to study the interaction of these new compounds and standard ligand with 15-LO. The docking study implied that these ligands have hydrogen bond interaction with the residue of active site of 15-LO.







Similar content being viewed by others
References
D. Patel, S.D. Toliwal, J.V. Patel, K. Jadav, A. Gupte, Y. Patel, J. Appl. Chem. Res. 16, 53 (2011)
D.L. Boger, Chem. Rev. 86, 781 (1986)
D.L. Boger, Tetrahedron 39, 2869 (1986)
W.A. Romanchick, M.M. Joulie, Heterocycles 9, 1631 (1978)
A. Monge, J. Palop, C. Ramizrez, M. Font, E. Fernandez-Alvarez, Eur. J. Med. Chem. 26, 179 (1991)
I.M. Labouta, N.H. Eshba, H.M. Salama, Monatsh. Chem. 119, 591 (1988)
Z.K.A. El-Samii, J. Chem. Tech. Biotech. 53, 143 (1992)
M.J. Leach, M.S. Nobls, Eur. Pat. EP0459829 (1991)
J.G. Małecki, B. Machura, A. Świtlicka, Struct. Chem. 22, 77 (2011)
L.C. March, G.S. Bajwa, J. Lee, K. Wasti, M.M. Joullie, J. Med. Chem. 19, 845 (1976)
B.F. Abdel-Wahab, M.F. El-Mansy, R.E. Khidre, J. Iran. Chem. Soc. 10, 1085 (2013)
Z. El-Gendy, J.M. Morsy, H.A. Allimony, W.R. Abdel-Monem, R.M. Abdel-Rahman, Phosphorus Sulphur. 178, 2055 (2003)
M. Ghassemzadeh, B. Rezaeirad, S. Bahemmat, B. Neumüller, J. Iran. Chem. Soc. 9, 285 (2012)
J. Wieczorek, M. Modarski, A. Rykowski, P. Nantka-Nimrski, Arch. Immunol. Ther. Exp. 28, 727 (1980)
D.L. Trepanier, E.R. Wagner, G. Harris, D. Allan, J. Med. Chem. 9, 881 (1966)
K. Srinivas, U. Srinivas, R. Jayathirtha, K. Bhanuprakash, K. Harakishore, U.S.N. Murthy, Bioorg. Med. Chem. Lett. 15, 1121 (2005)
S.D. Toliwal, K. Jadav, B. Bhatt, N. Verma, N. Jha, Arc. Apl. Sci. Res. 1, 344 (2009)
V.L. Rusinov, E.N. Ulomskii, O.N. Chupakhin, M.M. Zubairnov, A.B. Kapustin, N.I. Mitin, M.I. Zhiravetskii, I.A. Vinograd, Pharm. Chem. J. 24, 646 (1990)
A.S. Gupta, T. Bhattacharya, K. Hajela, K. Shankar, S. Ahmad, Pestic. Sci. 16, 65 (1985)
S.S. Smagin, V.E. Bogachev, A.K. Yakubovskii, S.E. Metkalova, T.P. Privol’neva, V.V. Chugunon, E.F. Lavretskaya, Pharmaceut. Chem. J. 9, 222 (1975)
C. Rubat, P. Coudert, S. Marvel, J. Fialip, J.J. Couquelet, J. Pharm. Pharmacol. 49, 1019 (1997)
M. Bakavoli, M. Nikpour, M. Rahimizadeh, M.R. Saberi, H. Sadeghian, Bioorgan. Med. Chem. 15, 2120 (2007)
A.R. Brash, J. Biol. Chem. 274, 23679 (1999)
T. Schewe, Biol. Chem. 383, 365 (2005)
U. Kelavkar, W. Glasgow, T.E. Eling, Curr. Urol. Rep. 3, 207 (2002)
A. Karimian, H. Eshghi, M. Bakavoli, A. Shiri, M. Saadatmandzadeh, T. Asghari, H. Moradi, J. Iran. Chem. Soc. 12, 1501 (2015)
H.G. Johnson, M.L. McNee, F.F. Sun, Am. Rev. Respir. Dis. 131, 917 (1985)
L. Zhao, C.D. Funk, Trends. Cardiovasc. Med. 14, 191 (2004)
M. Iranshahi, A. Jabbari, A. Orafaie, R. Mehri, S. Zeraatkar, T. Ahmadi, M. Alimardani, H. Sadeghian, Eur. J. Med. Chem. 57, 134 (2012)
HyperChem® Release 7, Hypercube Inc. http://www.hyper.com (2002)
U.S.F. Tambunan, I. Kurniawan, A.A. Parikesit, Afr. J. Biotechnol. 12, 4419 (2013)
P.K. Chang, T.L.V. Ulbricht, J. Am. Chem. Soc. 80, 976 (1958)
D.H. Deutsch, US. Patent 3922273 (1975)
P.J. Dudfield, V.D. Le, S.D. Lindell, C.W. Rees, J. Chem. Soc. Perkin Trans. 1, 2929 (1999)
Acknowledgments
We are grateful to Ferdowsi University of Mashhad, Iran, for financial support of this project.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Dedicated to the memory of Professor Mohammad Rahimizadeh.
Rights and permissions
About this article
Cite this article
Mohammadi, A., Eshghi, H., Bakavoli, M. et al. Synthesis of novel 3-substituted-5H-benzo[5,6][1, 4]thiazino[3,2-e][1,2,4]triazines and their 15-lipoxygenase inhibitory activity. J IRAN CHEM SOC 13, 1539–1547 (2016). https://doi.org/10.1007/s13738-016-0870-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-016-0870-6