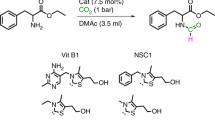
Abstract
The catalyst-free oxidation of various pyridine derivatives and tertiary amines to their corresponding N-oxides with 1,1,2,2-tetrahydroperoxy-1,2-diphenylethane as an efficient oxidant has been developed. The methodology proved to tolerate a number of functional groups. The reactions proceeded smoothly under solvent-free and mild conditions at room temperature. All the products were easily extracted from the reaction mixtures in excellent yields.
Graphical abstract
The catalyst-free oxidation of various pyridine derivatives and tertiary amines to their corresponding N-oxides with 1,1,2,2-tetrahydroperoxy-1,2-diphenylethane as an efficient oxidant has been developed. The methodology proved to tolerate a number of functional groups. The reactions proceeded smoothly under solvent-free and mild conditions at room temperature. All the products were easily extracted from the reaction mixtures in excellent yields.




Similar content being viewed by others
References
C. Copéret, H. Adolfsson, VKh Tinh-Alfredo, A.K. Yudin, K.B. Sharpless, J. Org. Chem. 63, 1740 (1998)
K. Imamura, K. Tano, S. Kobeppn, S. Jpn. Kokai Tokkyo Koho, JP Patent 1087811 (1998)
C.J. Oconnor, E. Sinn, R.L. Carlin, Inorg. Chem. 16, 3314 (1977)
A. Albini, Synthesis 263 (1993)
A.V. Malkov, M. Bell, F. Castelluzzo, P. Kocovsky, Org. Lett. 7, 3219 (2005)
R. Sarma, A. Karmakar, J.B. Baruah, Inorg. Chim. Acta 361, 2081 (2008)
C. Coperet, H. Adolfsson, K.B. Sharpless, Chem. Commun 16, 1565 (1997)
K.M. Kim, K.H Chung, J.N. Kim, E.K. Ryu, Synthesis 283 (1993)
J. Balzarini, M. Stevens, G. Andrei et al., Helv. Chim. Acta 85, 296 (2002)
J. Balzarini, M. Stevens, E. De Clereq, D. Schols, C. Panecouque, J. Antimicrob. Chemoth 55, 135 (2005)
M. Stevens, C. Pannecouque, E. De Clercq et al., Antimicrob. Agents Ch. 47, 2951 (2003)
D. Swern, Organic Peroxides, vol. 2 (Wiley, New York, 1971), pp. 355–533
G.B. Payne, P.H. Deming, P.H. Williams, J. Org. Chem. 26, 651 (1961)
G.B. Payne, J. Org. Chem. 26, 668 (1961)
D.C. Edwards, Tetrahedron Lett. 7, 4767 (1966)
E. Ochiai, Chem. Commun. 28 (1971)
P. Brougham, M.S. Cooper, D.A. Cummerson, H. Heaney, N. Thompson, Synthesis 1015 (1987)
B.H.R. Derek, O. Nubar, V. Bernard, Tetrahedron Lett. 44, 7385 (1988)
S. Daya, M. Kol, S. Rozen, Synthesis 1427 (1999)
R. Nesi, D. Giomi, S. Papaleo, S. Bracci, P. Dapporto, Synthesis 884 (1988)
R. Nesi, D. Giomi, S. Papaleo, S. Bracci, P. Dapporto, J. Org. Chem. 54, 706 (1989)
R. Nesi, D. Giomi, S. Papaleo, M. Corti, J. Org. Chem. 55, 1227 (1990)
R. Nesi, D. Giomi, S. Papaleo, S. Turchi, J. Org. Chem. 57, 3713 (1992)
A.W. Chucholowski, S. Uhlendorf, Tetrahedron Lett. 31, 1949 (1990)
R.A. Sheldon, Chemtech. 24, 38 (1994)
P.T. Anastas, J.C. Warner, Green chemistry: theory and practice (Oxford University Press, Oxford, 1998)
J.H. Clark, Green Chem. 1, 1 (1999)
K. Takabe, T. Yamada, T. Katagiri, Chem. Lett. 11, 1987 (1982)
R.W. Murray, K. Iyanar, J. Org. Chem. 61, 8099 (1996)
L. Goti, Nannelli. Tetrahedron Lett. 37, 6027 (1996)
M. Ferrer, F. Sanchez-Baeza, A. Messeguer, A. Diez, M. Rubiralta, J. Chem. Soc., Chem. Commun. 293 (1995)
M. Ferrer, F. Sanchez-Baeza, A. Messeguer, Tetrahedron 53, 15877 (1997)
G. Dyker, B. Holzer, Tetrahedron 55, 12557 (1999)
W. Adam, D. Golsch, Angew. Chem. Int. Ed. Engl. 32, 737 (1993)
D. Azarifar, K. Khosravi, F. Soleimanei, Synthesis 15, 2553 (2009)
D. Azarifar, K. Khosravi, F. Soleimanei, Molecules 15, 1433 (2010)
D. Azarifar, K. Khosravi, J. Iran. Chem. Soc. 8, 1006 (2011)
D. Azarifar, Z. Najminejad, K. Khosravi, Synth. Commun. 43, 826 (2013)
D. Azarifar, O. Badalkhani, K. Khosravi, Y. Abbasi, J. Adv. Chem. 11, 3452 (2015)
D. Azarifar, B. Mahmoudi, K. Khosravi, J. Adv. Chem. 11, 3547 (2015)
D. Azarifar, K. Khosravi, Z. Najminejad, J. Iran. Chem. Soc. 10, 979 (2013)
D. Azarifar, K. Khosravi, Z. Najminejad, K. Soleimani, J. Iran. Chem. Soc. 9, 321 (2012)
D. Azarifar, K. Khosravi, Eur. J. Chem. 1, 15 (2010)
D. Azarifar, K. Khosravi, Synlett 2755 (2010)
D. Azarifar, Z. Najminejad, Synlett 1377 (2013)
D. Azarifar, S.M. Khatami, Z. Najminejad, J. Iran. Chem. Soc. 11, 587 (2014)
D. Azarifar, K. Khosravi, Z. Najminejad, K. Soleimani, Heterocycles 81, 2855 (2010)
D. Azarifar, M. Golbaghi, M. Pirveisian, Z. Najminejad, J. Adv. Chem. 10, 308 (2014)
Z. Zhu, J.H. Espenson, J. Org. Chem. 60, 7728 (1995)
S.L. Jain, B. Sain, Chem. Commun. 10, 1040 (2002)
S.L. Jain, J.K. Joseph, B. Sain, Synlett 16, 2661 (2006)
Y. Ding, W. Zhao, W. Song, Z. Zhang, B. Ma, Green Chem. 13(6), 1486 (2011)
Acknowledgments
The authors wish to thank the Research Council of Bu-Ali Sina University for financial support to carry out this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Azarifar, D., Mahmoudi, B. Catalyst-free and selective oxidation of pyridine derivatives and tertiary amines to corresponding N-oxides with 1,2-diphenyl-1,1,2,2-tetrahydroperoxyethane. J IRAN CHEM SOC 13, 645–651 (2016). https://doi.org/10.1007/s13738-015-0776-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0776-8