Abstract
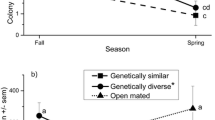
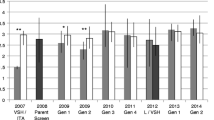
In two tests, honey bee colonies of different origins were sampled monthly to detect possible differential infection with Nosema ceranae; colony sizes and queen status were monitored quarterly. One experiment used queens crossed with drones of the same type obtained from colonies which had previously exhibited high and low infections. A second experiment used queens from ten commercial sources. No clear genotypic (P = 0.682) or phenotypic (P = 0.623) differences in infection were evident. Colony deaths and supersedures did not relate significantly with infection except for deaths of colonies in the autumn (P = 0.02). Significant effects on colony growth were found in all seasons: average 3-month decreases in population ranged from 0.4 to 1.4 frames of bees per million N. ceranae per bee. These results confirm that N. ceranae can be involved in weakening of colonies even in warm climates and suggest that breeding for resistance may require more intense selection, larger base populations, or different screening methods.


Similar content being viewed by others
References
Anderson, D.L., Giacon, H. (1992) Reduced pollen collection by honey bee (Hymenoptera: Apidae) colonies infected with Nosema apis and sacbrood virus. Ann. Entomol. Soc. Amer. 85, 47–51
Antúnez, K., Martín-Hernández, R., Prieto, L., Meana, A., Zunino, P., Higes, M. (2009) Immune suppression in the honey bee (Apis mellifera) following infection by Nosema ceranae (Microsporidia). Environ. Microbiol. 11, 2284–2290
Bourgeois, A.L., Rinderer, T.E., Beaman, L.D., Danka, R.G. (2010) Genetic detection and quantification of Nosema apis and N. ceranae in the honey bee. J. Invertebr. Pathol. 103, 53–58
Bourgeois, L., Beaman, L.D., Rinderer, T.E. (2011) Preservation and processing methods for molecular genetic detection and quantification of Nosema ceranae. Sci. Bee Cult. 3, 1–5
Bourgeois, L., Rinderer, T.E., Sylvester, H.A., Holloway, B., Oldroyd, B.P. (2012) Patterns of Apis mellifera infestation by Nosema ceranae support the parasite hypothesis for the evolution of extreme polyandry in eusocial insects. Apidologie 43, 539–548
Chaimanee, V., Chantawannakul, P., Chen, Y., Evans, J.D., Pettis, J.C. (2012) Differential expression of immune genes of adult honey bee (Apis mellifera) after inoculated by Nosema ceranae. J. Insect Physiol. 58, 1090–1095
Chen, Y., Evans, J.D., Smith, I.B., Pettis, J.S. (2008) Nosema ceranae is a long-present and wide-spread microsporidian infestation of the European honey bee (Apis mellifera) in the United States. J. Invertebr. Pathol. 97, 186–188
Eischen, F.A., Graham, H., Rivera, R. (2010) Almond pollen collection by honey bee colonies heavily infected with Nosema ceranae. Proc. Amer. Bee Res. Conf. Amer Bee J. 150, 500
Eischen, F.A., Graham, H., Rivera, R. (2012) Impact of Nosema ceranae on honey bee colonies: A 14 month study. Proc. Amer. Bee Res. Conf. Amer Bee J. 152, 401
Genersch, E., von der Ohe, W., Kaatz, H., Schroeder, A., Otten, C., Büchler, R., Berg, S., Ritter, W., Mühlen, W., Gisder, S., Meixner, M., Liebig, G., Rosenkranz, P. (2010) The German bee monitoring project: a long term study to understand periodically high winter losses of honey bee colonies. Apidologie 41, 332–352
Guzmán-Novoa, E., Eccles, L., Calvete, Y., Mcgowan, J., Kelly, P.G., Correa-Benítez, A. (2010) Varroa destructor is the main culprit for the death and reduced populations of overwintered honey bee (Apis mellifera) colonies in Ontario, Canada. Apidologie 41, 443–450
Harbo, J.H., Hoopingarner, R.A. (1997) Honey bees (Hymenoptera: Apidae) in the United States that express resistance to Varroa jacobsoni (Mesostigmata: Varroidae). J Econ. Ent. 90, 893–898
Higes, M., García-Palencia, P., Martıín-Hernández, R., Meana, A. (2007) Experimental infection of Apis mellifera honeybees with Nosema ceranae (Microsporidia). J. Invertebr. Pathol. 94, 211–217
Higes, M., Martín-Hernández, R., Botías, C., Garrido-Bailón, E., González-Porto, A.V., Barrios, L., del Nozal, M.J., Bernal, J.L., Jiménez, J.J., García-Palencia, P., Meana, A. (2008) How natural infection by Nosema ceranae causes honeybee colony collapse. Environ. Microbiol. 10, 2659–2669
Huang, Q., Kryger, P., Le Conte, Y., Moritz, R.F.A. (2012) Survival and immune response of drones of a Nosemosis tolerant honey bee strain towards N. ceranae infections. J. Invertebr. Pathol. 109, 297–302
Klee, J., Besana, A.M., Genersch, E., Gisder, S., Nanetti, A., Tam, D.Q., Chinh, T.X., Puerta, F., Ruz, J.M., Kryger, P., Message, D., Hatjina, F., Korpela, S., Fries, I., Paxton, R.J. (2007) Widespread dispersal of the microsporidian Nosema ceranae, an emergent pathogen of the western honey bee, Apis mellifera. J. Invertebr. Pathol. 96, 1–10
Malone, L.A., Giacon, H.A., Newton, M.R. (1995) Comparison of the responses of some New Zealand and Australian honey bees (Apis mellifera L) to Nosema apis Z. Apidologie 26, 495–502
Malone, L.A., Stefanovic, D. (1999) Comparison of the response of two races of honeybees to infection with Nosema apis Zander. Apidologie 30, 375–382
Mayack, C., Naug, D. (2009) Energetic stress in the honeybee Apis mellifera from Nosema ceranae infection. J. Invertebr. Pathol. 100, 185–188
Mayack, C., Naug, D. (2010) Parasitic infection leads to decline in hemolymph sugar levels in honeybee foragers. J. Insect Physiol. 56, 1572–1575
Oliver, R. (2012) Sick bees, Part 16: The “quick squash” method. Am. Bee J. 152, 167–171
Rinderer, T.E., Sylvester, H.A. (1978) Variation in response to Nosema apis, longevity and hoarding behavior in a free-mating population of the honey bee. Ann. Entomol. Soc. Amer. 71, 372–374
Smart, M.D., Sheppard, W.S. (2012) Nosema ceranae in age cohorts of the western honey bee (Apis mellifera). J. Invertebr. Pathol. 109, 148–151
Traver, B.E., Fell, R.D. (2011) Prevalence and infection intensity of Nosema in honey bee (Apis mellifera L.) colonies in Virginia. J. Invertebr. Pathol. 107, 43–49
Traynor, K., Traynor, M. (2008) Bee breeding around the world. Am. Bee J. 148, 135–139
Williams, G.R., Shutler, D., Little, C.M., Burgher-Maclellan, K.L., Rogers, R.E.L. (2010) The microsporidian Nosema ceranae, the antibiotic Fumagilin-B®, and western honey bee (Apis mellifera) colony strength. Apidologie 42, 15–22
Acknowledgments
Lorraine Beaman, Garrett Dodds, David Dodge, Victor Rainey, and Daniel Winfrey assisted with field or laboratory work. Garrett Dodds conducted instrumental inseminations. Debbie Boykin (USDA, ARS) provided statistical advice, and Frank Eischen (USDA, ARS) and Eric Mussen (U. California) provided useful comments on versions of the manuscript. Evergeen Honey Company (Bunkie, LA) provided field colonies and equipment. Partial funding for this project was provided by the Honey Bee Health Improvement Project of the North American Pollinator Protection Campaign.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript editor: David Tarpy
Preuve négative des effets de l’origine génétique des abeilles sur Nosema ceranae , preuve positive des effets de Nosema ceranae sur les abeilles
Apis mellifera / résistance / nosémose / élevage / sélection
Negative Belege für Effekte der genetischen Abstammung der Bienen auf Nosema ceranae , positive Belege für Effekte von Nosema ceranae auf Bienen
Nosema ceranae / Apis mellifera / Honigbienen / Resistenz / Züchtung / Selektion
Rights and permissions
About this article
Cite this article
Villa, J.D., Bourgeois, A.L. & Danka, R.G. Negative evidence for effects of genetic origin of bees on Nosema ceranae, positive evidence for effects of Nosema ceranae on bees. Apidologie 44, 511–518 (2013). https://doi.org/10.1007/s13592-013-0201-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13592-013-0201-1