Abstract
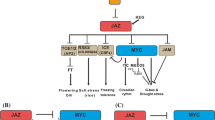
Amongst all the phytohormones, jasmonates are most important signaling molecule associated with plant defense against herbivores as they activate the expression of both direct and indirect defenses. Jasmonates are produced at insect-infested local sites and either jasmonate itself or some Jasmonic Acid (JA)-elicited compound travels to systemic leaves and elicits defense response. Upon herbivory, receptors/sensors of the host plant recognize Herbivory Associated Molecular Patterns (HAMPs) and early signaling events start with the involvement of Mitogen Activated Protein Kinases (MAPKs). JA biosynthesis is initiated in the chloroplast with the hydrolysis of chloroplast membrane lipids by phospholipases releasing free α-linolenic acid (α-LA), which gets converted to 12-oxo-phytodienoic acid (OPDA). OPDA is subsequently transported to peroxisomes where JA is produced by β-oxidation and it conjugates with isoleucine by JA amino acid synthetase (JAR) enzymes. Once high levels of JA-Isoleucine (Ile) are achieved, JA-induced response is instigated by SCF complex (a complex consisting of Skp1, Cullin-1 and F-box protein). JA-Ile forms a complex to form JA-Ile-COI1 (Coronatine insensitive Protein1) that recognizes a target protein Jasmonate ZIM domain (JAZ1), which is ubiquitinated and subsequently subjected to proteasomal degradation releasing the repression of MYC2, thereby activating expression of JA responsive genes. Further, both Novel Interactor of JAZ (NINJA) and Topless (TPL) proteins function as negative regulators of jasmonate responses. Although there has been remarkable progress in recent years, many open questions remain to be answered regarding jasmonate signaling during herbivory.

Similar content being viewed by others
References
Agrawal AA (2011) Current trends in the evolutionary ecology of plant defence: plant defence theory. Funct. Ecol. 25:420–432. doi : 10.1111/j.1365-2435.2010.01796.x
Andreou A, Brodhun F, Feussner I (2009) Biosynthesis of oxylipins in non-mammals. Prog Lipid Res 48:148–170. doi:10.1016/j.plipres.2009.02.002
Arimura GI, Matsui K, Takabayashi J (2009) Chemical and molecular ecology of herbivore-induced plant volatiles: proximate factors and their ultimate functions. Plant Cell Physiol 50:911–923. doi:10.1093/pcp/pcp030
Bell E, Creelman R, Mullet J (1995) A chloroplast lipoxygenase is required for wound-induced jasmonic acid accumulation in Arabidopsis. Proc Natl Acad Sci U S A 92:8675–8679
Bennett RN, Wallsgrove RM (1994) Secondary metabolites in plant defence mechanisms. New Phytol 127:617–633. doi:10.1111/j.1469-8137.1994.tb02968.x
Berenbaum MR, Zangerl AR (2008) Facing the future of plant-insect interaction research: le retour à la “raison d'être”. Plant Physiol 146:804–811. doi:10.1104/pp.107.113472
Berger S, Bell E, Mullet JE (1996) Two methyl jasmonate-insensitive mutants show altered expression of AtVSP in response to methyl jasmonate and wounding. Plant Physiol 111:525–531. doi:10.1104/pp.111.2.525
Brash AR, Baertschi SW, Ingram CD, Harris TM (1988) Isolation and characterization of natural allene oxides: unstable intermediates in the metabolism of lipid hydroperoxides. Proc Natl Acad Sci U S A 85(10):3382–3386
Browse J (2009a) Jasmonate passes muster: a receptor and targets for the defensormone. Annul Rev Plant Biol 60:183–205. doi:10.1146/annurev.arplant.043008.092007
Browse J (2009b) The power of mutants for investigating jasmonate biosynthesis and signaling. Phytochem 70:1539–1546. doi:10.1016/j.phytochem.2009.08.004
Browse J, Howe GA (2008) New weapons and a rapid response against insect attack. Plant Physiol 146:832–838. doi:10.1104/pp.107.115683
Bruinsma M, Posthumus MA, Mumm R, Mueller MJ, van Loon JJA, Dicke M (2009) Jasmonic acid-induced volatiles of Brassica oleracea attract parasitoids: effects of time and dose, and comparison with induction by herbivores. J Exp Bot 60:2575–2587. doi:10.1093/jxb/erp101
Chini A, Fonseca S, Fernández G, Adie B, Chico JM, Lorenzo O, García-Casado G, López-Vidriero I, Lozano FM, Ponce MR, et al. (2007) The JAZ family of repressors is the missing link in jasmonate signalling. Nature 448(7154):666–671. doi:10.1038/nature06006
Chung HS, Koo AJK, Gao X, Jayanty S, Thines B, Jones AD, Howe GA (2008) Regulation and function of Arabidopsis JASMONATE ZIM-domain genes in response to wounding and herbivory. Plant Physiol 146:952–964. doi:10.1104/pp.107
Dicke M, Baldwin IT (2010) The evolutionary context for herbivore-induced plant volatiles: beyond the ‘cry for help’. Trends Plant Sci 15:167–175. doi:10.1016/j.tplants
Dickens JC (2006) Plant volatiles moderate response to aggregation to pheromone in Colorado potato beetle. J Appl Entomol 130:26–31. doi:10.1111/j.1439-0418.2005.01014.x
Dombrecht B, Xue GP, Sprague SJ, Kirkegaard JA, Ross JJ, Reid JB, Fitt GP, Sewelam N, Schenk PM, Manners JM, et al. (2007) MYC2 differentially modulates diverse jasmonate-dependent functions in Arabidopsis. Plant Cell 19:2225–2245. doi:10.1105/tpc.106.048017
Durrant WE, Dong X (2004) Systemic acquired resistance. Annu Rev Phytopathol 42:185–209
Farmer EE, Ryan CA (1990) Interplant communication: air- borne methyl jasmonate induces synthesis of proteinase inhibitors in plant leaves. Proc Natl Acad Sci U S A 87:7713–7716. doi:10.1073/pnas.87.19.7713
Farmer EE, Ryan CA (1992) Octadecanoid precursors of jasmonic acid activate the synthesis of wound inducible proteinase inhibitors. Plant Cell 4:129–134. doi:10.1105/tpc.4.2.129
Feussner I, Wasternack C (2002) The lipoxygenase pathway. Annual Review in Plant Biology 53:275–297. doi:10.1146/annurev.arplant.53.100301.135248
Feys BJF, Benedetti CE, Penfold CN, Turner JG (1994) Arabidopsis mutants selected for resistance to the phytotoxin coronatine are male sterile, insensitive to methyl jasmonate, and resistant to a bacterial pathogen. Plant Cell 6:751–759. doi:10.1105/tpc.6.5.751
Franceschi VR, Grimes HD (1991) Induction of soybean vegetative storage proteins and anthocyanins by low level atmospheric methyl jasmonate. Proc Natl Acad Sci U S A 88:6745–6749
Gao LL, Anderson JP, Klingler JP, Nair RM, Edwards OR, Singh KB (2007) Involvement of the octadecanoid pathway in Bluegreen aphid resistance in Medicago truncatula. Mol Plant-Microbe Interact 20:82–93
Gatehouse JA (2002) Plant resistance towards insect herbivores: a dynamic interaction. New Phytol. 56:145–169. doi:10.1046/j.1469-8137.2002.00519.x
Glauser G, Dubugnon L, Mousavi SAR, Rudaz S, Wolfender J-L, Farmer EE (2009) Velocity estimates for signal propagation leading to systemic jasmonic acid accumulation in wounded Arabidopsis. J Biol Chem 284:34506–34513. doi:10.1074/jbc.M109.061432
Gosset V, Harmel N, Gobel C, Francis F, Haubruge E, Wathelet JP, du Jardin P, Feussner I, Fauconnier ML (2009) Attacks by a piercing-sucking insect myzus persicae sultzer) or a chewing insect (leptinotarsa decemlineata say) on potato plants (Solanum tuberosum L.) induce differential changes in volatile compound release and oxylipin synthesis. J Exp Bot 60:1231–1240
Green TR, Ryan CA (1972) Wound-induced proteinase inhibitor in plant leaves: a possible defense mechanism against insects. Science 175:776–777
Gulsen O, Eickhoff T, Heng-Moss T, Shearman R, Baxendale F, Sarath G, Lee D (2010) Characterization of peroxidase changes in resistant and susceptible warm season turf grasses challenged by blissus occiduus. Arthropod-Plant Interact. 4:45–55. doi:10.1007/s11829-010-9086-3
Gundlach H, Müller MJ, Kutchan TM, Zenk MH (1992) Jasmonic acid is a signal transducer in elicitor induced plant cell cultures. Proc Natl Acad Sci U S A 89:2389–2393
Halitschke R, Baldwin IT (2003) Antisense LOX expression increases herbivore performance by decreasing defense responses and inhibiting growth-related transcriptional reorganization in nicotiana attenuata. Plant J 36:794–807. doi:10.1046/j.1365-313X.2003.01921.x
Hamberg M, Fahlstadius P (1990) Allene oxide cyclase: a new enzyme in plant lipid metabolism. Arch Biochem Biophys 276:518–526. doi:10.1016/0003-9861(90)90753-L
Heil M (2008) Indirect defence via tritrophic interactions. New Phytol 178:41–61. doi:10.1111/j.1469-8137.2007.02330.x
Hofmann E, Zerbe P, Schaller F (2006) The crystal structure of Arabidopsis thaliana allene oxide cyclase: insights into the oxylipin cyclization reaction. Plant Cell 18:3201–3217. doi:10.1105/tpc.106.043984
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. doi:10.1146/annurev.arplant.59.032607.092825
Howe GA, Lightner J, Browse J, Ryan CA (1996) An octadecanoid pathway mutant (JL5) of tomato is compromised in signaling for defense against insect attack. Plant Cell 8(11):2067–2077. doi:10.1105/tpc.8.11.2067
Hyun Y, Choi S, Hwang H-J, Yu J, Nam S-J, Ko J, Park J-Y, Seo YS, Kim EY, Ryu SB, Kim WT, Lee Y-H, Kang H, Lee I (2008) Cooperation and functional diversification of two closely related galactolipase genes for jasmonate biosynthesis. Dev Cell 14:183–192. doi:10.1016/j.devcel.2007.11.010
Ishiguro S, Kawai-Oda A, Ueda J, Nishida I, Okada K (2001) The DEFECTIVE IN ANTHER DEHISCENCE1 gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 13:2191–2209. doi:10.1105/tpc.010192
Jianqiang W, Baldwin IT (2010) New insights into plant responses to the attack from insect herbivores. Annu Rev Genet 44:1–24. doi:10.1146/annurev-genet-102209-163500
Jones CG, Hopper RF, Coleman JS, Krischik VA (1993) Control of systemically induced herbivore resistance by plant vascular architecture. Oecologia 93:452–456. doi:10.1007/BF00317892
Kang J-H, Wang L, Giri A, Baldwin IT (2006) Silencing threonine deaminase and JAR4 in nicotiana attenuata impairs jasmonic acid-isoleucine-mediated defenses against manduca sexta. Plant Cell 18:3303–3320. doi:10.1105/tpc.106.041103
Katsir L, Schilmiller AL, Staswick PE, He SY, Howe GA (2008) COI1 is a critical component of a receptor for jasmonate and the bacterial virulence factor coronatine. Proc Natl Acad Sci U S A 105:7100–7105. doi:10.1073/pnas.0802332105
Kempema LA, Cui XP, Holzer FM, Walling LL (2007) Arabidopsis transcriptome changes in response to phloem-feeding silverleaf whitefly nymphs. Similarities and distinctions in responses to aphids. Plant Physiol 143:849–865
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144. doi:10.1126/science.291.5511.2141
Kessler A, Baldwin IT (2002) Plant responses to insect herbivory: the emerging molecular analysis. Annu Rev Plant Biol 53:299–328. doi:10.1146/annurev.arplant.53.100301.135207
Kost C, Heil M (2008) The defensive role of volatile emission and extrafloral nectar secretion for lima bean in nature. J Chem Ecol 34(1):2–13. doi:10.1007/s10886-007-9404-0
Kuśnierczyk A, Winge P, Midelfart H, Armbruster WS, Rossiter JT, Bones AM (2007) Transcriptional responses of Arabidopsis thaliana ecotypes with different glucosinolate profiles after attack by polyphagous myzus persicae and oligophagous brevicoryne brassicae. J Exp Bot 58:2537–2552
Laudert D, Hennig P, Stelmach BA, Müller A, Andert L, Weiler EW (1997) Analysis of 12-oxo-phytodienoic acid enantiomers in biological samples by capillary gas chromatography-mass spectrometry using cyclodextrin stationary phases. Anal Biochem 246:211–217. doi:10.1006/abio.1997.2012
Li L, Li C, Lee GI, Howe GA (2002) Distinct roles for jasmonate synthesis and action in the systemic wound response of tomato. Proc Natl Acad Sci U S A 99:6416–6421. doi:10.1073/pnas.072072599
Li C, Liu G, Xu C, Lee GI, Bauer P, Ling HQ, Ganal MW, Howe GA (2003) The tomato suppressor of prosystemin-mediated responses2 gene encodes a fatty acid desaturase required for the biosynthesis of jasmonic acid and the production of a systemic wound signal for defense gene expression. Plant Cell 15:1646–1661. doi:10.1105/tpc.012237
Li L, Zhao Y, McCaig BC, Wingerd BA, Wang J, Whalon ME, Pichersky E, Howe GA (2004) The tomato homolog of CORONATINE-INSENSITIVE1 is required for the maternal control of seed maturation, jasmonate-signaled defense responses, and glandular trichome development. Plant Cell 16:126–143. doi:10.1105/tpc.017954
Li C, Schilmillera AL, Liu G, Lee GI, Jayanty S, Sageman C, Vrebalov J, Giovannoni JJ, Yagie K, Kobayashi Y, Howe GA (2005) Role of β-oxidation in jasmonate biosynthesis and systemic wound signaling in tomato. Plant Cell 17:971–986. doi:10.1105/tpc.104.029108
Lorenzo O, Solano R (2005) Molecular players regulating the jasmonate signalling network. Curr Opin Plant Biol 8:532–540. doi:10.1016/j.pbi.2005.07.003
Lorenzo O, Chico JM, Sanchez-Serrano JJ, Solano R (2004) Jasmonate-insensitive1 encodes a MYC transcription factor essential to discriminate between different jasmonate-regulated defense responses in Arabidopsis. Plant Cell 16:1938–1950. doi:10.1105/tpc.022319
Ludwig AA, Romeis T, Jones JDG (2004) CDPK-mediated signalling pathways: specificity and cross-talk. J Exp Bot 55:181–188. doi:10.1093/jxb/erh008
Maffei M, Bossi S, Spiteller D, Mithöfer A, Boland W (2004) Effects of feeding spodoptera littoralis on lima bean leaves. I. Membrane potentials, intracellular calcium variations, oral secretions, and regurgitate components. Plant Physiol 134:1752–1762. doi:10.1104/pp.103.034165
Maffei M, Mithöfer A, Arimura GI, Uchtenhagen H, Bossi S, Bertea CM, Cucuzza LS, Novero M, Volpe V, Quadro S, Boland W (2006) Effects of feeding spodoptera littoralis on lima bean leaves. III Membrane Depolarization and Involvement of Hydrogen Peroxide. Plant Physiol 140:1022–1035. doi:10.1104/pp.105.071993
Mao YB, Cai WJ, Wang JW, Hong GJ, Tao XY, Wang LJ, Huang YP, Chen XY (2007) Silencing a cotton bollworm P450 monooxygenase gene by plant-mediated RNAi impairs larval tolerance of gossypol. Nat Biotechnol 25:1307–1313. doi:10.1038/nbt1352
McConn M, Browse J (1996) The critical requirement for linolenic acid is pollen development, not photosynthesis, in an Arabidopsis mutant. Plant Cell 8:403–416. doi:10.1105/tpc.8.3.403
McConn M, Creelman RA, Bell E, Mullet JE, Browse J (1997) Jasmonate is essential for insect defense in Arabidopsis. Proc Natl Acad Sci U S A 94:5473–5477
McGurl B, Pearce G, Orozco-Cardenas M, Ryan CA (1992) Structure, expression, and antisense inhibition of the systemin precursor gene. Science 255:1570–1573. doi:10.1126/science.1549783
Meindl T, Boller T, Felix G (1998) The plant wound hormone systemin binds with the N-terminal part to its receptor but needs the C-terminal part to activate it. Plant Cell. 10:1561–1570. doi:10.1105/tpc.10.9.1561
Narvaez-Vasquez J, Florin-Christensen J, Ryan CA (1999) Positional specificity of a phospholipase-a activity induced by wounding, systemin, and oligosaccharide elicitors in tomato leaves. Plant Cell 11:2249–2260. doi:10.1105/tpc.11.11.2249
Orians CM, Pomerleau J, Ricco R (2000) Vascular architecture generates fine scale variation in systemic induction of proteinase inhibitors in tomato. J Chem Ecol 26:471–485. doi:10.1023/A:1005469724427
Orozco-Cardenas ML, Ryan CA (2002) Nitric oxide negatively modulates wound signaling in tomato plants. Plant Physiol 130:487–493. doi:10.1104/pp.008375
Orozco-Cardenas ML, Narvaez-Vasquez J, Ryan CA (2001) Hydrogen peroxide acts as a second messenger for the induction of defense genes in tomato plants in response to wounding, systemin, and methyl jasmonate. Plant Cell 13:179–191. doi:10.1105/tpc.13.1.179
Ozawa R, Arimura G, Takabayashi J, Shimoda T, Nishioka T (2000) Involvement of jasmonate- and salicylate-related signaling pathways for the production of specific herbivore-induced volatiles in plants. Plant Cell Physiol. 41:391–398. doi:10.1093/pcp/41.4.391
Park JH, Halitschke R, Kim HB, Baldwin IT, Feldmann KA, Feyereisen R (2002) A knock-out mutation in allene oxide synthase results in male sterility and defective wound signal transduction in Arabidopsis due to a block in jasmonic acid biosynthesis. Plant J 31:1–12. doi:10.1046/j.1365-313X.2002.01328.x
Park SJ, Huang YH, Ayoubi P (2006) Identification of expression profiles of sorghum genes in response to greenbug phloem-feeding using cDNA subtraction and microarray analysis. Planta 223:932–947
Paschold A, Halitschke R, Baldwin IT (2007) Co(i)-ordinating defenses: NaCOI1 mediates herbivore induced resistance in nicotiana attenuata and reveals the role of herbivore movement in avoiding defenses. Plant J Cell Mol Biol 51:79–91. doi:10.1111/j.1365-313X.2007.03119.x
Pauwels L, Inzé D, Goossens A (2009) Jasmonate-inducible gene: what does it mean? Trends Plant Sci 14:87–91. doi:10.1016/j.tplants.2008.11.005
Pauwels L, Barbero GF, Geerinck J, Tilleman S, Grunewald W, Peŕez AC, Chico JM, Bossche RV, Sewell J, Gil E, et al. (2010) NINJA connects the co-repressor TOPLESS to jasmonate signalling. Nature 464:788–791. doi:10.1038/nature08854
Reymond P, Bodenhausen N, Van Poecke RM, Krishnamurthy V, Dicke M, Farmer EE (2004) A conserved transcript pattern in response to a specialist and a generalist herbivore. Plant Cell 16:3132–3147. doi:10.1105/tpc.104.026120
Sanders PM, Lee PY, Biesgen C, Boone JD, Beals TP, Weiler EW, Goldberg RB (2000) The Arabidopsis DELAYED DEHISCENCE1 gene encodes an enzyme in the jasmonic acid synthesis pathway. Plant Cell 12:1041–1061. doi:10.1105/tpc.12.7.1041
Santner A, Estelle M (2009) Recent advances and emerging trends in plant hormone signaling. Nature 459:1071–1078. doi:10.1038/nature08122
Scheer JM, Clarence A, Jr R (2002) The systemin receptor SR160 from lycopersicon peruvianum is a member of the LRR receptor kinase family. Proc Natl Acad Sci U S A 99:9585–9590. doi:10.1073/pnas.132266499
Schilmiller AL, Koo AJK, Howe GA (2007) Functional diversification of acyl-coenzyme a oxidases in jasmonic acid biosynthesis and action. Plant Physiol 143:812–824. doi:10.1104/pp.106.092916
Schittko U, Preston CA, Baldwin IT (2000) Eating the evidence? Manduca sexta larvae cannot disrupt specific jasmonate induction in nicotiana attenuata by rapid consumption. Planta 210:343–346. doi:10.1007/PL00008143
Schmelz EA, Alborn RT, Tumlinson JR (2001) The influence of intact-plant and excised-leaf bioassay designs on volicitin- and jasmonic acid-induced sesquiterpene volatiles release in Zea mays. Planta 214:171–179. doi:10.1007/s004250100603
Schommer C, Palatnik JF, Aggarwal P, Chételat A, Cubas P, Farmer EE, Nath U, Weigel D (2008) Control of jasmonate biosynthesis and senescence by miR319 targets. PLoS Biol 6:e230. doi:10.1371/journal.pbio.0060230
Seo S, Katou S, Seto H, Gomi K, Ohashi Y (2007) The mitogen-activated protein kinases WIPK and SIPK regulate the levels of jasmonic and salicylic acids in wounded tobacco plants. Plant J 49:899–909. doi:10.1111/j.1365-313X.2006.03003x
Sheard LB, Tan X, Mao H, Withers J, Ben-Nissan G, Hinds TR, Kobayashi Y, Hsu FF, Sharon M, Browse J, He SY, Rizo J, Howe GA, Zheng N (2010) Jasmonate perception by inositol-phosphate-potentiated COI1 JAZ co-receptor. Nature 468:400–405. doi:10.1038/nature09430
Shivaji R, Camas A, Ankala A, Engelberth J, Tumlinson JH, Williams WeéééP, Wilkinson JR, Luthe DS (2010) Plants on constant alert: elevated levels of jasmonic acid and jasmonate-induced transcripts in caterpillar-resistant maize. J. Chem. Ecol. 36: 179–191. doi : 10.1007/s10886-010-9752-z
Singh A, Singh IK, Verma PK (2008) Differential transcript accumulation in cicer arietinum L. in response to a chewing insect helicoverpa armigera and defence regulators correlate with reduced insect performance. J. Exp. Bot 59(9):2379–2392. doi:10.1093/jxb/ern111
Stankovic B, Davies E (1997) Intercellular communication in plants: electrical stimulation of proteinase inhibitor gene expression in tomato. Planta 202:402–406. doi:10.1007/s004250050143
Staswick PE, Su W, Howell SH (1992) Methyl jasmonate inhibition of root growth and induction of a leaf protein are decreased in an Arabidopsis thaliana mutant. Proc Natl Acad Sci U S A 89:6837–6840
Staswick PE, Tiryaki I, Rowe ML (2002) Jasmonate response locus JAR1 and several related Arabidopsis genes encode enzymes of the firefly luciferase superfamily that show activity on jasmonic, salicylic, and indole-3-acetic acids in an assay for adenylation. Plant Cell 14:1405–1415. doi:10.1105/tpc.000885
Stintzi A, Browse J (2000) The Arabidopsis male-sterile mutant, opr3, lacks the 12- oxophytodienoic acid reductase required for jasmonate synthesis. Proc Natl Acad Sci U S A 97:10625–10630. doi:10.1073/pnas.190264497
Stintzi A, Weber H, Reymond P, Browse J, Farmer EE (2001) Plant defense in the absence of jasmonic acid: the role of cyclopentenones. Proc Natl Acad Sci U S A 98:12837–12842. doi:10.1073/pnas.211311098
Stork W, Diezel C, Halitschke R, Galis I, Baldwin IT (2009) An ecological analysis of the herbivory elicited JA burst and its metabolism: plant memory processes and predictions of the moving target model. PLoS One 4:e4697. doi:10.1371/journal.pone.0004697
Sun J-Q, Jiang H-L, Li C-Y (2011) Systemin/jasmonate-mediated systemic defense signaling in tomato Mol. Plant 4(4):607–615. doi:10.1093/mp/ssr008
Suza WP, Staswick PE (2008) The role of JAR1 in jasmonoyl-L: −isoleucine production during Arabidopsis wound response. Planta 227:1221–1232. doi:10.1007/s00425-008-0694-4
Takahashi F, Yoshida R, Ichimura K, Mizoguchi T, Seo S, Yonezawa M, Maruyama K, Yamaguchi-Shinozaki K, Shinozaki K, et al. (2007) The mitogen-activated protein kinase cascade MKK3-MPK6 is an important part of the jasmonate signal transduction pathway in Arabidopsis. The Plant Cell 19:805–818. doi:10.1105/tpc.106.046581
Thaler S (1999) Jasmonate-inducible plant defenses cause increased parasitism of herbivores. Nature 399:686–688. doi:10.1038/21420
Thines B, Katsir L, Melotto M, Niu Y, Mandaokar A, Liu G, Nomura K, He SY, Howe GA, Browse J (2007) JAZ repressor proteins are targets of the SCFCOI1 complex during jasmonate signaling. Nature 448:661–665. doi:10.1038/nature05960
Tian D, Traw MB, Chen JQ, Kreitman M, Bergelson J (2003) Fitness costs of R-gene-mediated resistance in Arabidopsis thaliana. Nature 423:74–77. doi:10.1038/nature01588
Tong X, Qi J, Zhu X, Mao B, Zeng L, Wang B, Li Q, Zhou G, Xu X, Lou Y, He Z (2012) The rice hydroperoxide lyase OsHPL3 functions in defense responses by modulating the oxylipin pathway. Plant J 71:763–775. doi:10.1111/j.1365-313X.2012.05027.x
Ulloa RM, Raíces M, MacIntosh GC, Maldonado S, Téllez-Iñón MT (2002) Jasmonic acid affects plant morphology and calcium-dependent protein kinase expression and activity in Solanum tuberosum. Physiol Plant 115:417–427. doi:10.1034/j.1399-3054.2002.1150312.x
Von MB, der GE v, Schneitz K, Keller B (2002) The Arabidopsis male-sterile mutant dde2-2 is defective in the ALLENE OXIDE SYNTHASE gene encoding one of the key enzymes of the jasmonic acid biosynthesis pathway. Planta 216:187–192. doi:10.1007/s00425-002-0906-2
Wang L, Allmann S, Wu J, Baldwin IT (2008) Comparisons of LIPOXYGENASE3-and JASMONATERESISTANT4/6-silenced plants reveal that jasmonic acid and jasmonic acid-amino acid conjugates play different roles in herbivore resistance of nicotiana attenuata. Plant Physiol 146:904–915. doi:10.1104/pp.107.109264
War AR, Paulraj MG, War MY, Ignacimuthu S (2011) Jasmonic acid- mediated induced resistance in groundnut (arachis hypogaea L.) against helicoverpa armigera (hubner) (Lepidoptera: noctuidae). J Plant Growth Regul 30:512–523. doi:10.1007/s00344-011-9213-0
Wasternack C (2007) Jasmonates: an update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann Bot 100:681–697. doi:10.1093/aob/mcm079
Wasternack C, Hause B (2013) Jasmonates: biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany. Ann Bot 111:1021–1058. doi:10.1093/aob/mct067
Wasternack C, Kombrink E (2010) Jasmonates: structural requirements for lipid-derived signals active in plant stress responses and development. ACS Chem Biol 5:63–77. doi:10.1021/cb900269u
Wu J, Baldwin IT (2009) Herbivory-induced signalling in plants: perception and action. Plant Cell Environ 32:1161–1174. doi:10.1111/j.1365-3040.2009.01943.x
Wu J, Hettenhausen C, Meldau S, Baldwin IT (2007) Herbivory rapidly activates MAPK signaling in attacked and unattacked leaf regions but not between leaves of nicotiana attenuata. Plant Cell 19:1096–1122. doi:10.1105/tpc.106.049353
Xie DX, Feys BF, James S, Nieto-Rostro M, Turner JG (1998) COI1: an Arabidopsis gene required for jasmonate-regulated defense and fertility. Science 280:1091–1094. doi:10.1126/science.280.5366.1091
Yan J, Zhang C, Gu M, Bai Z, Zhang W, Qi T, Cheng Z, Peng W, Luo H, Nan F, et al. (2009) The Arabidopsis CORONATINE INSENSITIVE1 protein is a jasmonate receptor. Plant Cell 21:2220–2236. doi:10.1105/tpc.109.065730
Yang D-H, Hettenhausen C, Baldwin IT, Wu J (2012) Silencing nicotiana attenuata calcium-dependent protein kinases, CDPK4 and CDPK5, strongly up-regulates wound- and herbivory-induced jasmonic acid accumulations. Plant Physiol 159:1591–1607. doi:10.1104/pp.112.199018
Zavala JA, Patankar AG, Gase K, Baldwin IT (2004) Constitutive and inducible trypsin proteinase inhibitor production incurs large fitness costs in nicotiana attenuata. Proc Natl Acad Sci U S A 101:1607–1612. doi:10.1073/pnas.0305096101
Zerbe P, Weiler EW, Schaller F (2007) Preparative enzymatic solid phase synthesis of cis(+)-12- oxo phytodienoic acid – physical interaction of AOS and AOC is not necessary. Phytochemistry 68:229–236. doi:10.1016/j.phytochem.2006.10.010
Zhang X, Wu Q, Zhang PJ, Zheng SJ, van Loon JJ, Boland W, David A, Mumm R, Dicke M (2009) Whiteflies interfere with indirect plant defense against spider mites in lima bean. Proc Natl Acad Sci U S A 106:21202–21207
Zhang X, Wu Q, Ren J, Qian W, He S, Huang K, Yu XC, Gao Y, Huang P, An C (2012) Two novel RING-type ubiquitin ligases, RGLG3 and RGLG4, are essential for jasmonate-mediated responses in Arabidopsis. Plant Physiol 160:808–822. doi:10.1104/pp.112.203422
Zhao LY, Chen JL, Cheng DF, Sun JR, Liu Y, Tian Z (2009) Biochemical and molecular characterizations of sitobion avenae – induced wheat defense responses. Crop Prot 28:435–442
Ziegler J, Wasternack C, Hamberg M (1999) On the specificity of allene oxide cyclase. Lipids 34:1005–1015
Zimmermann MR, Maischak H, Mithofer A, Boland W, Felle HH (2009) System potentials, a novel electrical long-distance apoplastic signal in plants, induced by wounding. Plant Physiol 149:1593–1600. doi:10.1104/pp.108.133884
Acknowledgments
Authors acknowledge “Science and Engineering Research Board, Department of Science and Technology New Delhi, India” for the financial support under “Young Scientist Scheme”. We thank Mr. Onkar Nath for assistance in the preparation of the diagram.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest declared.
Rights and permissions
About this article
Cite this article
Singh, A., Singh, S. & Singh, I.K. Recent insights into the molecular mechanism of jasmonate signaling during insect-plant interaction. Australasian Plant Pathol. 45, 123–133 (2016). https://doi.org/10.1007/s13313-015-0392-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-015-0392-1