Abstract
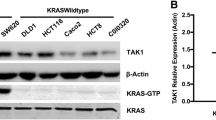
KRAS mutation in colorectal cancer (CRC) activates transforming growth factor-β (TGF-β)-activated kinase 1 (TAK1) to promote tumor progression. In the current study, we explored the potential effect of LYTAK1, a novel TAK1 inhibitor, against KRAS mutant CRC cells in vitro and in vivo. We found that LYTAK1 dose-dependently inhibited KRAS mutant CRC cell (HT-29 and SW-620 lines) growth, and induced cell cycle G1-S arrest. Further, LYTAK1 activated apoptosis in HT-29 cells and SW-620 cells, and apoptosis inhibitors almost reversed LYTAK1-mediated growth inhibition. While in KRAS wild-type (WT) CRC cell lines (DLD-1 and HCT-116), LYTAK1 had almost no effect on cell growth, cell cycle progression, or cell apoptosis. In KRAS mutant HT-29 cells and SW-260 cells, LYTAK1 blocked TAK1 activation or phosphorylation at Thr-184/187. Activation of nuclear factor κB (NF-κB) in these cells, detected by phosphorylations of p65 and IκB kinase α (IKKα) as well as expression of NF-κB-regulated gene cyclin D1, was significantly inhibited by LYTAK1. Further, LYTAK1 treatment resulted in downregulation of β-catenin and Wnt response gene Axin 2, indicating Wnt inactivation. In vivo, oral LYTAK1 significantly inhibited HT-29 xenograft growth in nude mice. Together, these results show that LYTAK1 inhibits KRAS mutant CRC cell growth both in vitro and in vivo. LYTAK1 might be investigated as a novel agent against CRC with KRAS mutation.




Similar content being viewed by others
References
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9–29.
Westwood M, van Asselt T, Ramaekers B, Whiting P, Joore M, Armstrong N, et al. Kras mutation testing of tumours in adults with metastatic colorectal cancer: a systematic review and cost-effectiveness analysis. Health Technol Assess. 2014;18:1–132.
Arrington AK, Heinrich EL, Lee W, Duldulao M, Patel S, Sanchez J, et al. Prognostic and predictive roles of KRAS mutation in colorectal cancer. Int J Mol Sci. 2012;13:12153–68.
Siddiqui AD, Piperdi B. KRAS mutation in colon cancer: a marker of resistance to EGFR-I therapy. Ann Surg Oncol. 2010;17:1168–76.
Fakih MM. Kras mutation screening in colorectal cancer: from paper to practice. Clin Colorectal Cancer. 2010;9:22–30.
Rinehart J, Adjei AA, Lorusso PM, Waterhouse D, Hecht JR, Natale RB, et al. Multicenter phase II study of the oral MEK inhibitor, CI-1040, in patients with advanced non-small-cell lung, breast, colon, and pancreatic cancer. J Clin Oncol. 2004;22:4456–62.
Sakurai H. Targeting of TAK1 in inflammatory disorders and cancer. Trends Pharmacol Sci. 2012;33:522–30.
Martin SE, Wu ZH, Gehlhaus K, Jones TL, Zhang YW, Guha R, et al. Rnai screening identifies TAK1 as a potential target for the enhanced efficacy of topoisomerase inhibitors. Curr Cancer Drug Targets. 2011;11:976–86.
Melisi D, Xia Q, Paradiso G, Ling J, Moccia T, Carbone C, et al. Modulation of pancreatic cancer chemoresistance by inhibition of TAK1. J Natl Cancer Inst. 2011;103:1190–204.
Singh A, Sweeney MF, Yu M, Burger A, Greninger P, Benes C, et al. TAK1 inhibition promotes apoptosis in KRAS-dependent colon cancers. Cell. 2012;148:639–50.
Ray DM, Myers PH, Painter JT, Hoenerhoff MJ, Olden K, Roberts JD. Inhibition of transforming growth factor-beta-activated kinase-1 blocks cancer cell adhesion, invasion, and metastasis. Br J Cancer. 2012;107:129–36.
Chen MB, Wei MX, Han JY, Wu XY, Li C, Wang J, et al. Microrna-451 regulates AMPK/mTORC1 signaling and fascin1 expression in HT-29 colorectal cancer. Cell Signal. 2014;26:102–9.
Di Nicolantonio F, Arena S, Tabernero J, Grosso S, Molinari F, Macarulla T, et al. Deregulation of the PI3k and KRAS signaling pathways in human cancer cells determines their response to everolimus. J Clin Invest. 2010;120:2858–66.
Wang S, Liu Z, Wang L, Zhang X. NF-kappab signaling pathway, inflammation and colorectal cancer. Cell Mol Immunol. 2009;6:327–34.
Vaiopoulos AG, Athanasoula K, Papavassiliou AG. NF-kappab in colorectal cancer. J Mol Med (Berl). 2013;91:1029–37.
Guttridge DC, Albanese C, Reuther JY, Pestell RG, Baldwin Jr AS. NF-kappab controls cell growth and differentiation through transcriptional regulation of cyclin D1. Mol Cell Biol. 1999;19:5785–99.
McConnell BB, Yang VW. The role of inflammation in the pathogenesis of colorectal cancer. Curr Color Cancer Rep. 2009;5:69–74.
Ishizuka M, Nagata H, Takagi K, Kubota K. Influence of inflammation-based prognostic score on mortality of patients undergoing chemotherapy for far advanced or recurrent unresectable colorectal cancer. Ann Surg. 2009;250:268–72.
Kraus S, Arber N. Inflammation and colorectal cancer. Curr Opin Pharmacol. 2009;9:405–10.
Itzkowitz SH, Yio X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004;287:G7–G17.
Li Q, Verma IM. NF-kappab regulation in the immune system. Nat Rev Immunol. 2002;2:725–34.
Bienz M, Clevers H. Linking colorectal cancer to Wnt signaling. Cell. 2000;103:311–20.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (81372433 and 81472920), the Natural Science Foundation of Jiangsu Province of China (BK20131149), and the Suzhou Administration of Science & Technology (SYS201360)
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jundong Zhou, Bing Zheng, and Jiansong Ji are co-first authors.
Rights and permissions
About this article
Cite this article
Zhou, J., Zheng, B., Ji, J. et al. LYTAK1, a novel TAK1 inhibitor, suppresses KRAS mutant colorectal cancer cell growth in vitro and in vivo. Tumor Biol. 36, 3301–3308 (2015). https://doi.org/10.1007/s13277-014-2961-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2961-2