Abstract
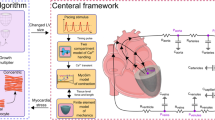
Although image-based methods like MRI are well-developed, numerical simulation can help to understand human heart function. This function results from a complex interplay of biochemistry, structural mechanics, and blood flow. The complexity of the entire system often causes one of the three parts to be neglected, which limits the truth to reality of the reduced model. This paper focuses on the interaction of myocardial stress distribution and ventricular blood flow during diastole and systole in comparison to a simulation of the same patient-specific geometry with a given wall movement (Spiegel, Strömungsmechanischer Beitrag zur Planung von Herzoperationen, 2009). The orthotropic constitutive law proposed by Holzapfel et al. (Philos. Trans. R. Soc. Lond. Ser. A, 367:3445–3475, 2009) was implemented in a finite element package to model the passive behavior of the myocardium. Then, this law was modified for contraction. Via the ALE method, the structural model was coupled to a flow model which incorporates blood rheology and the circulatory system (Oertel, Prandtl—Essentials of Fluid Mechanics, 3rd edn, Springer Science + Business Media, 2010; Oertel et al., Modelling the Human Cardiac Fluid Mechanics, 3rd edn, Universitätsverlag Karlsruhe, 2009). Comparison reveals a good quantitative and qualitative agreement with respect to fluid flow. The motion of the myocardium is consistent with physiological observations. The calculated stresses and the distribution are within the physiological range and appear to be reasonable. The coupled model presented contains many features essential to cardiac function. It is possible to calculate wall stresses as well as the characteristic ventricular fluid flow. Based on the simulations we derive two characteristics to assess the health state quantitatively including solid and fluid mechanical aspects.






Similar content being viewed by others
References
de Vecchi, A., D. A. Nordsletten, R. Razavi, G. Greil, and N. P. Smith. Patient specific fluid-structure ventricular modelling for integrated cardiac care. Med. Biol. Eng. Comput. 51(11):1261–1270, 2013.
Demirdzic, I., and M. Peric. Space conservation law in finite volume calculations of fluid flow. Int. J. Numer. Methods Fluids 8(9):1037–1050, 1988.
Demirdzic, I., and M. Peric. Finite volume method for prediction of fluid flow in arbitrarily shaped domains with moving boundaries. Int. J. Numer. Methods Fluids 10(7):771–790, 1990.
Dokos, S., B. H. Smaill, A. A. Young, and I. J. LeGrice. Shear properties of passive ventricular myocardium. Am. J. Physiol. Heart Circ. Physiol. 283:H2650–H2659, 2002.
Formaggia, L., A. Quarteroni, and A. Veneziani. Cardiovascular mathematics. Berlin: Springer, 2009.
Fraunhofer Institute for Algorithms and Scientific Computing (SCAI). MpCCI 4.3 Documentation, 2014. (http://www.mpcci.de/fileadmin/mpcci/download/MpCCI-4.3.0/doc/pdf/MpCCIdoc.pdf).
Fung, Y. C. Mathematical representation of the mechanical properties of the heart muscle. J. Biomech. 3:381–404, 1970.
Fung, Y. C. Biomechanics—Mechanical Properties of Living Tissues (2nd ed.). New York: Springer, 1993.
Fung, Y. C. Biomechanics—Circulation (2nd ed.). New York: Springer, 1997.
Göktepe, S., S. N. S. Acharya, J. Wong, and E. Kuhl. Computational modeling of passive myocardium. Int. J. Numer. Methods Biomech. Eng. 27:1–12, 2011.
Guccione, J. M., G. S. Kassab, and M. B. Ratcliffe (eds.). Computational Cardiovascular Mechanics—Modeling and Applications in Heart Failure. Berlin: Springer, 2010.
Holzapfel, G. A., and R. W. Ogden. Constitutive modelling of passive myocardium: a structurally based framework for material characterization. Philos. Trans. R. Soc. Lond. Ser. A 367:3445–3475, 2009.
Humphrey, J. D., and F. C. Yin. A new constitutive formulation for characterizing the mechanical behavior of soft tissues. Biophys. J. 52(4):563–570, 1987.
Hunter, P. J., A. D. McCulloch, P. M. Nielsen, and B. H. Smaill. A finite element model of passive ventricular mechanics. In: Computational Methods in Bioengineering, edited by R. L. Spilker, and B. R. Simon. New York: ASME, 1988.
Jones, T. N., and D. N. Metaxas. Patient-specific analysis of left ventricular blood flow. In: Medical Image Computing and Computer-Assisted Intervention—MICCAI98, Vol. 1496, edited by W. Wells, A. Colchester, and S. Delp. Lecture Notes in Computer Science, Berlin: Springer, 1998, pp. 156–166.
Krittian, S. Modellierung der kardialen Strömung-Struktur-Wechselwirkung: Implicit coupling for KaHMo FSI. PhD thesis, Universiät Karlsruhe (TH), 2009.
Krittian, S., U. Janoske, H. Oertel, and T. Böhlke. Partitioned fluid–solid coupling for cardiovascular blood flow: left-ventricular fluid mechanics. Ann. Biomed. Eng. 38(4):1426–1441, 2010.
Krittian, S., T. Schenkel, U. Janoske, and H. Oertel. Partitioned fluid-solid coupling for cardiovascular blood flow: validation study of pressure-driven fluid-domain deformation. Ann. Biomed. Eng. 38(8):2676–2689, 2010.
LeGrice, I. J., B. H. Smaill, L. Z. Chai, S. G. Edgar, J. B. Gavin, and P. J. Hunter. Laminar structure of the heart: ventricular myocyte arrangement and connective tissue architecture in the dog. Am. J. Physiol. 269:H571–H582, 1995.
LeGrice, I. J., P. J. Hunter, and B. H. Smaill. Laminar structure of the heart: a mathematical model. Am. J. Physiol. Heart Circ. Physiol. 272:H2466–H2476, 1997.
Liepsch, D., G. Thurston, and M. Lee. Viscometric studies simulating blood-like fluids and their applications in models of arterial branches. Biorheology 28:39–52, 1991.
Mühlhausen, M.-P. Strömung-struktur-gekoppelte Modellierung und Simulation des menschlichen Herzens. PhD thesis, Karlsruher Institut für Technologie (KIT), 2012.
Nordsletten, D. A., S. A. Niederer, M. P. Nash, P. J. Hunter, and N. P. Smith. Coupling multi-physics models to cardiac mechanics. Progr. Biophys. Mol. Biol. 2009.
Nordsletten, D. A., M. McCormick, P. J. Kilner, P. J. Hunter, D. Kay, and N. P. Smith. Fluid-solid coupling for the investigation of diastolic and systolic human left ventricular function. Int. J. Numer. Methods Biomed. Eng. 27:1017–1039, 2011.
Oertel, H. (ed.). Prandtl—Essentials of Fluid Mechanics (3rd ed.). New York: Springer Science + Business Media, 2010.
Oertel, H., and S. Krittian. Modelling the Human Cardiac Fluid Mechanics (4th ed.). Karlsruhe: KIT Scientific Publishing, 2012.
Oertel, H., S. Krittian, and K. Spiegel. Modelling the Human Cardiac Fluid Mechanics (3rd ed.). Karlsruhe: Universitätsverlag Karlsruhe, 2009.
Pedrizzetti, G., and K. Perktold. Cardiovascular Fluid Mechanics. Number 446 in CISM International Centre for Mechanical Sciences. Wien: Springer, 2003.
Perschall, M. Numerische Untersuchung des Wellenpumpenkonzeptes und der mechanischen Herzunterstützung. PhD thesis, Karlsruher Institut für Technologie (KIT), 2010. (http://digbib.ubka.uni-karlsruhe.de/volltexte/1000016087).
Peskin, C. S. Flow patterns around heart valves: a numerical method. J. Comput. Phys. 10:252–271, 1972.
M. Reik. Simulation der Strömungsstruktur im menschlichen Herzen. PhD thesis, Universität Karlsruhe (TH), 2007.
Schenkel, T., M. Malve, M. Reik, M. Markl, B. Jung, and H. Oertel. Mri-based cfd analysis of flow in a human left ventricle: methodology and application to a healthy heart. Ann. Biomed. Eng. 37:503–515, 2009.
Spiegel, K. Strömungsmechanischer Beitrag zur Planung von Herzoperationen. PhD thesis, Universität Karlsruhe (TH), 2009.
Streeter, D. D., and D. L. Bassett. An engineering analysis of myocardial fiber orientation in pig’s left ventricle in systole. Anat. Record 155:503–511, 1966.
Streeter, D. D., H. M. Spotnitz, D. P. Patel, J. Ross, and E. H. Sonnenblick. Fiber orientation in the canine left ventricle during diastole and systole. Circ. Res. 24:339–347, 1969.
Tay, W. B., Y. H. Tseng, L. Y. Lin, and W. Y. Tseng. Towards patient-specific cardiovascular modeling system using the immersed boundary technique. BioMed. Eng. OnLine 10:52, 2011.
Wang, H. M., X. Y. Luo, H. Gao, R. W. Ogden, B. E. Griffith, C. Berry, and T. J. Wang. A modified Holzapfel-Ogden law for a residually stressed finite strain model of the human left ventricle in diastole. Biomech Model Mechanobiol 13(1):99–113, 2014.
Watanabe, H., S. Sugiura, and T. Hisada. The looped heart does not save energy by maintaining the momentum of blood flowing in the ventricle. Am. J. Physiol. Heart Circ. Physiol. 294:H2191–H2196, 2008.
Conflict of interest
Mark-Patrick Mühlhausen, Uwe Janoske, and Herbert Oertel declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Ajit P. Yoganathan oversaw the review of this article.
Rights and permissions
About this article
Cite this article
Muehlhausen, MP., Janoske, U. & Oertel, H. Implicit Partitioned Cardiovascular Fluid–Structure Interaction of the Heart Cycle Using Non-newtonian Fluid Properties and Orthotropic Material Behavior. Cardiovasc Eng Tech 6, 8–18 (2015). https://doi.org/10.1007/s13239-014-0205-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-014-0205-7