Abstract
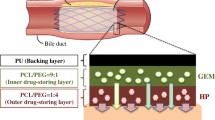
One of the major issues in cholangiocarcinoma (CC) treatment is bile duct occlusion-associated symptoms. As an alternative way of surgical excision of a CC, drug-eluting stents (DESs) have developed to treat both CC and occlusion. Gemcitabine (Gem) is effective for treatment of CC, however, it has limited applicability to use in DES because of its hydrophilic property. To solve these problems, we developed stent coating membrane (PTOA10-Gem) consisted of Gem, oleic acid (OA), and pellethane (PT). PTOA10-Gem membranes were additionally coated with PT to minimize rapid initial burst and washout of water-soluble Gem. OA was incorporated as a tissue penetration enhancer, and successfully achieved enhanced Gem penetration into cancer cell sheet (5.25-fold) and mouse skin (3- fold). Moreover, PTOA10-Gem led to dramatic decrease in tumor volume by 45% for 9 days. These findings suggest that the OA incorporated PT membrane is effective to improve a therapeutic efficacy of hydrophilic drug-eluting stents to treat CC, as well as other non-vascular cancers.

Similar content being viewed by others
References
J.-N. Vauthey and L. H. Blumgart, in Semin. Liver Dis., 14, 109 (1994).
K. Kuwayti, A. H. Baggenstoss, M. H. Stauffer, and J. T. Priestley, Surg. Gynecol. Obstet., 104, 357 (1957).
S. A. Khan, B. R. Davidson, R. Goldin, S. P. Pereira, W. M. C. Rosenberg, S. D. Taylor-Robinson, A. V. Thillainayagam, H. C. Thomas, M. R. Thursz, and H. Wasan, Gut, 51, vi1 (2002).
D. K. Lee, J. Hepatobiliary Pancreat. Surg., 16, 628 (2009).
J. Seo, J. Lee, and K. Na, J. Pharm. Investig., 46, 3171 (2016).
C. Penz, G. V. Kornek, M. Raderer, H. Ulrich-Pur, W. Fiebiger, A. Lenauer, D. Depisch, G. Krauss, B. Schneeweiss, and W. Schneeweiss, Ann. Oncol., 12, 183 (2001).
M. J. Chung, H. Kim, K. S. Kim, S. Park, J. B. Chung, and S. W. Park, J. Gastroenterol. Hepatol., 27, 261 (2012).
P. Gontero and B. Frea, Ann. Oncol., 17, 123 (2006).
W. Plunkett, P. Huang, Y. Z. Xu, V. Heinemann, R. Grunewald, and V. Gandhi, Semin. Oncol., 22, 4 (1995).
B. Pili, C. Bourgaux, F. Meneau, P. Couvreur, and P. M. Ollivon, J. Therm. Anal. Calorim., 98, 19 (2009).
J. W. Lee, S.-G. Yang, and K. Na, Int. J. Pharm., 427, 276 (2012).
L. Z. Benet, D. Kroetz, L. Sheiner, J. Hardman, and L. Limbird, in Goodman and Gilman's the Pharmacological Basis of Therapeutics, McGraw-Hill, New York, 1996, p 3.
G. Arya, M. Vandana, M., S. Acharya, and S. K. Sahoo, Nanomedicine, 7, 859 (2011).
G. Pasut, F. Canal, L. Dalla Via, S. Arpicco Veronese, and M. F. O. Schiavon, J. Control. Release, 127, 239 (2008).
G. Cavallaro, L. Mariano, S. Salmaso, P. Caliceti, and G. Gaetano, Int. J. Pharm., 307, 258 (2006).
S. M. Ali, M. U. Ahmad Chen, P., S. Sheikh, and I. Ahmad, Bioorganic Med. Chem. Lett., 15, 2571 (2005).
G. C. Chiou and C. Y. Chuang, J. Pharm. Sci., 78, 815 (1989).
S. Antoyo, A. Arellano, P. Ygartua, and C. Martin, Int. J. Pharm., 117, 219 (1995).
S. S. Davis and L. Illum, Clin. Pharmacokinet., 42, 1107 (2003).
S. Senel and A. A. Hincal, J. Control. Release, 72, 133 (2001).
J. L. Richardson, L. Illum, and N. W. Thomas, Pharm. Res., 9, 878 (1992).
A. Yamamoto, E. Hayakawa, Y. Kato, A. Nishiura, and V. H. Lee, J. Pharmacol. Exp. Ther., 263, 25 (1992).
I. B. Pathan and C. M. Setty, Trop. J. Pharm. Res., 8, 173 (2009).
H. Trommer and R. H. H. Neubert, Skin Pharmacol. Physiol., 19, 106 (2006).
S. Santoyo, A. Arellano, P. Ygartua, and C. Martin, Int. J. Pharm., 117, 218 (1995).
G. M. Goldenm, J. E. McKie, and R. O. Potts, J. Pharm. Sci., 76, 25 (1987).
M. L. Francoeur, G. M. Golden, and R. O. Potts, J. Pharm. Sci, 76, 25 (1978).
M. Goodman and B. W. Barry, J. Pharm. Pharmacol., 38, 71 (1986).
C. L. Gay, T. M. Murphy, J. Hadgraft, I. W. Kellaway, J. C. Evans, and C. C. Rowlands, Int. J. Pharm., 49, 39 (1989).
G. Mani, M. D. Feldman, D. Patel, and C. M. Agrawal, Biomaterials, 28, 1689 (2007).
K. Kandimalla, N. Kanikkannan, S. Andega, and M. Singh, J. Pharm. Pharmacol., 51, 783 (1999).
R. J. Babu, L. Chen, and N. Kanikkannan, in Percutaneous Penetration Enhancers Chemical Methods in Penetration Enhancement, Springer, Berlin Heidelberg, 2015, p 133.
R. M. Phillips, P. Loadman, and B. Cronin, Br. J. Cancer, 77, 2112 (1998).
E. R. Cooper, E. W. Merritt, and R. L. Smith, J. Pharm. Sci., 74, 688 (1985).
A. Naik, L. A. Pechtold, R. O. Potts, and R. H. Guy, J. Control. Release, 37, 299 (1995).
A. C. Williams and B. W. Barry, Adv. Drug Deliv. Rev., 64, 128 (2012).
M. A. Lampe, A. L. Burlingame, L. A. Whitney, J., Williams, M. L., Brown, E. B., E. Roitman, and P. M. Elias, J. Lipid Res., 24, 120 (1983).
E. Boelsma, H. Tanojo, H. E. Boddé, and M. Ponec, Toxicol. In Vitro, 10, 728 (1996).
J. Jiang, R. K. Wang, Phys. Med. Biol., 49, 5283 (2004)
A. H. Kyle, L. A. Huxham, A. S. Chiam, D. H. Sim, and A. I. Minchinton, Cancer Res., 64, 6304 (2004).
S. Moon, S.-G. Yang, and K. Na, Biomaterials, 32, 3603 (2011).
D. Min, D. Jeong, M. G. Choi, and K. Na, Biomaterials, 52, 484 (2015).
M. J. Chung, H. Kim, K. S. Kim, S. K., S. Park, J. B. Chung, and S. W. Park, J. Gastroenterol. Hepatol., 27, 261 (2012).
G. Acharya and K. Park, Adv. Drug Deliv. Rev., 58, 387 (2006).
M. M. Alves, J. M. Vieira, R. A. Pereira, M. A. Pereira, and M. Mota, Water Res., 35, 264 (2001).
H. J. Kuh, H. S. Jang, M. G. Wientjes, G. M., J. R. Weaver, and J. L. S. Au, J. Pharmacol. Exp. Ther., 290, 871 (1999).
Z. Xiong and F. A. Kapral, J. Med. Microbiol., 37, 192 (2015).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seo, J., Lee, J. & Na, K. Oleic acid incorporated gemcitabine-eluting stents for treatment of cholangiocarcinoma. Macromol. Res. 25, 63–69 (2017). https://doi.org/10.1007/s13233-017-5010-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-017-5010-3