Abstract
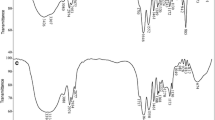
This paper describes the synthesis of a new chelating poly(acrylamide-co-dimethylaminoethyl methacrylate-co-1-acryloyl-3-phenyl thiourea) (PAPDM) hydrogel. The PAPDM hydrogels were prepared by the simple free radical polymerization of monomers acrylamide, dimethylamino ethyl methacrylate and 1-acryloyl-3-phenyl thiourea. The free radical initiator used for this study was ammonium persulphate and the cross-linker was ethylene glycol di methacrylate. The swelling study of the PAPDM hydrogels were performed in the aqueous environment to calculate the morphological parameters, such as the volume fraction in a swollen gel (v 2m ), polymer-solvent interaction parameter (χ) and molecular weight between the crosslinks (M c ). These hydrogels were characterized by Fourier transform infrared spectroscopy, differential scanning calorimetry and scanning electron microscopy. Further these chelating hydrogels were investigated for the removal of rare earth metal ions (samarium and terbium) from aqueous environments by varying the adsorption time, adsorbate concentration and monomer ratio. The adsorption time data for samarium and terbium were fitted to two simple kinetic models, pseudo-first and pseudo-second-order, and tested to examine the adsorption mechanisms. The kinetic parameters were calculated. The equilibrium data was fitted well to the Langmuir and Freundlich models.

Similar content being viewed by others
References
M. Humphries, Rare Earth Element, Global Supply Chain, Congressional Research Service, 2013. pp 1–3.
J. C. Laul, J. Radioanal. Nucl. Chem., 156, 235 (1992).
G. E. Fryxell, H. Wu, Y. Lin, W. J. Shaw, J. C. Birnbaum, J. C. Linehan, Z. Nie, K. Ken, and S. Kelly, J. Mater. Chem., 14, 3356 (2004).
A. Zhang, E. Kuraoka, and M. Kumagai, Eur. Polym. J., 43, 529 (2007).
K. K. Yadav, D. K. Singh, M. Anitha, L. Varshney, and H. Singh, Sep. Purif. Technol., 118, 350 (2013).
J. Roosen and K. Binnemans, J. Mater. Chem. A, 2, 1530 (2014).
L. Wang, X. Huang, Y. Yu, and Z. Long, Sep. Purif. Technol., 122, 490 (2014).
V. K. Jain, A. Handa, S. S. Sait, P. Shrivastav, and Y. K. Agarval, Anal. Chim. Acta, 429, 237(2001).
T.-L. Ho, Chem. Rev., 75, 1 (1975).
J. Soedarsono, A. Hagege, M. Burgard, Z. Asfari, and J. Vicens, Ber. Bunsenges. Phys. Chem., 100, 477 (1996).
H. Mukai, S. Miyazaki, S. Umetani, S. Kihara, and M. Matsui, Anal. Chim. Acta, 220, 111 (1989).
M. L. P. Reddy, J. R. B. Bharathi, S. Peter, and T. R. Ramamohan, Talanta, 50, 79 (1999).
K. S. V. Krishna Rao, B. V. K. Naidu, M. C. S. Subha, and T. M. Aminabhavi, Carbohydr. Polym., 66, 333 (2006).
K. S. V. Krishna Rao and C.-S. Ha, Polym. Bull., 62, 167 (2009).
T. J. Sudha Vani, N. Sivagangi Reddy, and K. S. V. Krishna Rao, Indian J. Adv. Chem. Sci., 2, 110 (2014).
A. Denizli, R. Say, S. Patir, and Y. Arica, Sep. Sci. Technol., 36, 2213 (2001).
B. L. Rivas, B. Quilodra, and E. Quiroz, J. Appl. Polym. Sci., 88, 2614 (2003).
J. Hendri, A. Hiroki, Y. Maekawa, M. Yoshida, and R. Katakai, Radiat. Phys. Chem., 60, 617 (2001).
H. Bessbousse, T. Rhlalou, J.-F. Verchere, and L. Lebrun, J. Membrane. Sci., 307, 249 (2008).
J. Ramkumar, B. Maiti, and T. S. Krishnamoorthy, J. Membr. Sci., 125, 269 (1997).
S. Doker, S. Malci, M. Dogan, and B. Salih, Anal. Chim. Acta, 553, 73(2005).
S. Doker, O. Çelikbicak, M. Dogan, and B. Salih, Microchem. J., 84, 80 (2006).
L. Wang, R. Xing, S. Liu, H. Yu, Y. Qin, K. Li, J. Feng, R. Li, and P. Li, J. Hazard. Mater., 180, 577 (2010).
M. Merdian, M. Z. Duz, and C. Hamamci, Talanta, 16, 639 (2001).
A. T. Mubarak and A. Z. El-Sonbati, Polym. Bull., 57, 683 (2006).
L. Zhao, J. Sun, Y. Zhao, L. Xu, and M. Zhai, Chem. Eng. J., 170, 162 (2011).
Z.-Y. Kong, J.-F. Wei, Y.-H. Lia, N.-N. Liu, H. Zhang, Y. Zhang, and L. Cuia, Chem. Eng. J., 254, 365 (2014).
M. Liu, F. Bian, and F. Sheng, Eur. Polym. J., 41, 283 (2005).
D. P. Drolet, D. M. Manuta, A. J. Lees, A. D. Katnani, and G. J. Coyle, Inorg. Chim. Acta, 146, 173 (1988).
P. J. Flory, Principles of Polymer Chemistry, Cornell University Press, Ithaca, 1953.
J. E. Mark and B. Erman, Rubberlike Elasticity: A Molecular Primer, Wiley, New York, 1988.
W. Xue, S. Champ, and M. B. Huglin, Polymer, 42, 3665 (2001).
S. Lagergren, in Zur Theorie der sogenannten Adsorption gelöster Stoffe, Kungliga Sveska Vetenskapskadomiens Handl, 1898, 24, pp 1–39.
N. S. Reddy, K. M. Rao, T. J. S. Vani, K. S. V. K. Rao, and Y. I. Lee, Desalination Water Treat., 57, 6503 (2015).
T. J. S. Vani, N. S. Reddy, K. S. V. K. Rao, and P. S. Rao, Desalination Water Treat., doi: 10.1080/19443994.2016.1151380 (2016).
G. Blanchard, M. Maunaye, and G. Martin, Water Res., 18, 1501 (1984).
E. Repo, J. K. Warchol, T. A. Kurniawan, and M. E. T. Sillanpaa, Chem. Eng. J., 161, 73 (2010).
M. S. Chiou and H. Y. Li, Chemosphere, 50, 1095 (2003).
I. Langmuir, J. Am. Chem. Soc., 38, 2221 (1916).
H. M. F. Freundlich, J. Phys. Chem., 57, 385 (1906).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Reddy, N.S.G., Rao, K.M., Rao, K.S.V.K. et al. Synthesis of 1-acryloyl-3-phenyl thiourea based pH sensitive hydrogels for removal of samarium and terbium. Macromol. Res. 24, 494–501 (2016). https://doi.org/10.1007/s13233-016-4068-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-016-4068-7