Abstract
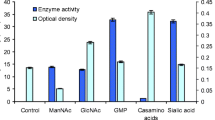
Neuraminidase is a virulence factor in many viruses and pathogenic bacteria. There are few reports of the detection of neuraminidase in the Aeromonas genus, but they do not include biochemical studies on the enzyme. In this work the neuraminidase production of Aeromonas sp. 40/02 strain and some properties of the enzyme were investigated for the first time. Neuraminidase was found in the supernatant, as well as in the cytosolic and membrane fractions of disrupted cells. It was demonstrated that the enzyme production is induced by a number of compounds structurally related to sialic acid. New data on the presence of extracellular and intracellular N-acylneuraminate lyase were obtained, suggesting a nutritional role of sialometabolism in this strain. For the first time, Aeromonas neuraminidase was isolated and partially purified by ion-exchange chromatography. Different purification profiles were registered depending on the growth media. The preparations containing neuraminidase activity were analyzed by native PAGE, and one of them was associated with an active protein band with a molecular weight of about 130 kDa. Our results suggest the presence of neuraminidase isoforms in Aeromonas sp. 40/02.




Similar content being viewed by others
References
Abrashev I, Orozova P (2004) Mucins in natural mud as inductor of some Aeromonas strains neuraminidase secretion. Probl Inf Parasit Dis 32:27–30
Abrashev I, Orozova P (2006) Erysipelothrix rhusiopathiae neuraminidase and its role in pathogenecity. Z Naturforsch 61c:434–438
Abrashev I, Velcheva P, Nikolov P, Kourteva J (1980) Substrate for colorimetric determination of enzyme activity. Bulgarian patent N 47647/IIR.
Aisaka K, Igarashi A, Uwajima T (1991) Purification, crystallization and characterization of neuraminidase from Micromonospora viridifaciens. Agric Biol Chem 55(4):997–1004
Almagro-Moreno S, Boyd F (2009) Sialic acid catabolism confers a competitive advantage to pathogenic Vibrio cholerae in the mouse intestine. Infect Immun 77(9):3807–3816. doi:10.1128/IAI.00279-09
Altwegg M, Geiss HK, Freij BJ (1989) Aeromonas as a human pathogen. Crit Rev Microbiol 16(4):253–286. doi:10.3109/10408418909105478
Beighton D, Whiley RA (1990) Sialidase activity of the “Streptococus milleri group” and other viridians group streptococci. J Clin Microbiol 28(6):1431–1433
Berg W, Gutschker-Gdaniec G, Schauer R (1985) Fluorescent staining of sialidases in polyacrylamide gel electrophoresis and ultrathin-layer isoelectric focusing. Anal Biochem 145(2):339–342. doi:10.1016/0003-2697(85)90371-9
Bouwstra JB, Deyl CM, Vliegenthart FG (1987) Purification and kinetic properties of sialidase from Clostridium perfringens. Biol Chem Hoppe Seyler 368:269–275
Byers HL, Tarelli E, Homer KA, Beighton D (2000) Isolation and characterization of sialidase from a strain of Streptococcus oralis. J Med Microbiol 49:235–244
Chiarezza M, Lyras D, Pidot SJ, Flores-Díaz M, Awad MM, Kennedy CL, Cordner LM, Phumoonna T, Poon R, Hughes ML, Emmins JJ, Alape-Giroń A, Rood JI (2009) The NanI and NanJ sialidases of Clostridium perfringens are not essential for virulence. Infect Immun 77(10):4421–4428. doi:10.1128/IAI.00548-09
Corfield T (1992) Bacterial sialidases: roles in pathogenecty and nutrition. Glycobiology 2(6):509–521. doi:10.1093/glycob/2.6.509
Corfield AP, Wagner SA, Clamp JR, Kriaris MS, Hoskins LS (1992) Mucin degradation in the human colon: production of sialidse, sialate O-acetylesterase, N-acetylneuraminate lyase, arylesterase and glycosulfatase activities by strains of fecal bacteria. Infect Immun 60(10):3971–3978
Davis L, Baig MM, Ayoub EM (1979) Properties of extracellular neuraminidase produced by group A Streptococcus. Infect Immun 24(3):780–786
Drzeniek R, Scharmann W, Balke E (1972) Neuraminidase and N-acetylneuraminate pyruvate-lyase of Pasteurella multocida. J Gen Microbiol 72:357–368. doi:10.1099/00221287-72-2-35
Eneva R, Engibarov S, Strateva T, Abrashev R, Abrashev I (2011) Biochemical studies on the production of neuraminidase by environmental isolates of Vibrio cholerae non-O1 from Bulgaria. Can J Microbiol 57(7):606–610. doi:10.1139/W11-042
Engibarov S, Eneva R, Abrashev R, Abrashev I (2010) Neuraminidase from environmental and clinical isolates of Aeromonas spp.-biochemical studies on the enzyme production. Probl Inf Parasit Dis 38(1):37–39
Flashner M, Wang P, Hurley JB, Tanenbaum SW (1977) Properties of an inducible extracellular neuraminidase from an Artrobacter isolate. J Bacteriol 129(3):1457–1465
Fraser AG, Collee JG (1975) The production of neuraminidase by food-poisoning strains of Clostridium welchii (C. perfringens). J Med Microbiol 8:251–263
Ghazaei C, Ahmadi M, Hosseini Jazani N (2010a) Optimization and comparative characterization of neuraminidase activities from Pseudomonas aeruginosa with Klebsiella pneumoniae, Hep-2 cell, sheep kidney and rat liver lysosome. Iran J Microbiol 2(1):30–37
Ghazaei C, Ahmadi M, Hosseini Jazani N (2010b) Detection of neuraminidase activity in Pseudomonas aeruginosa PAO1. Iran J Basic Med Sci 13(3):69–75
Gualdi L, Hayre JK, Gerlini A, Bidossi A, Colomba L, Trappetti C, Pozzi G, Docquer J-D, Andrew P, Ricci S, Oggioni MR (2012) Regulation of neuraminidase expression in Streptococcus pneumoniae. BMC Microbiol 12:200. doi:10.1186/1471-2180-12-200
Havelaar AH, Schets FM, Van Silfhout A, Jansen WH, Wieten G, Van Der Kooij D (1992) Typing of Aeromonas strains from patients with diarrhoea and from drinking water. J Appl Bacteriol 72(5):435–444. doi:10.1111/j.1365-2672.1992.tb01857.x
Hughes RC, Jeanloz RW (1964) The extracellular glycosidases of D. pneumoniae. I. Purification and properties of a neuraminidase and a beta-galactosidase. Action on alpha-1-acid glycoprotein. Biochemistry 3(10):1535–1543. doi:10.1021/bi00898a025
Jost BH, Songer JG, Bilington SJ (2002) Identification of a second Arcanobacterium pyogenes neuraminidase and involvement of neuraminidase activity in host cell adhesion. Infect Immun 70(3):1106–1112. doi:10.1128/IAI.70.3.1106-1112.2002
Kabir S, Ahmad N, Ali S (1984) Neuraminidase production by Vibrio cholerae 01 and other diarrheagenic bacteria. Infect Immun 44(3):747–749
Li J, Sayeed S, Robertson S, Chen J, McClane BA (2011) Sialidases affect the host cell adherence and epsilon toxin-induced cytotoxicity of Clostridium perfringens Type D strain CN3718. PLoS Pathog 7(12):e1002429. doi:10.1371/journal.ppat.1002429
Lichtensteiger CA, Vimr ER (2003) Purification and renaturation of membrane neuraminidase from Haemophilus parasuis. Vet Microbiol 93:79–87. doi:10.1016/S0378-1135(02)00443-1
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurment with the folin phenol reagent. J Biol Chem 193:265–275
Milligan TW, Straus DC, Mattingley SJ (1977) Extracellular neuraminidase production by group B streptococci. Infect Immun 18(1):189–195
Mizan S, Henk A, Stallings A, Maier M, Lee MD (2000) Cloning and characterization of sialidases with 2-6′ and 2-3′ sialyl lactose specificity from Pasteurella multocida. J Bacteriol 182(24):6874–6883. doi:10.1128/JB.182.24.6874-6883.2000
Muller HE (1974) Neuraminidases of bacteria and protozoa and their pathogenic role. Behring Inst Mitt 55:34–56
Murray PR, Pfaller MA, Rosenthal KS (2005) Vibrio and Aeromonas. In: Murray PR, Pfaller MA, Rosenthal KS (eds) Medical microbiology, 5th edn. Elsevier, Philadelphia, pp 339–346
Nakano V, Piazza RMF, Avila-Campos MJ (2006) A rapid assay of the sialidase activity in species of the Bacteroides fragilis group by using peanut lectin hemagglutination. Anaerobe 12:238–241. doi:10.1016/j.anaerobe.2006.07.003
Orozova P, Petrov P, Nenova R, Abrashev I (2007) Characterization of some virulence determinants of Aeromonas spp. isolated from meat, drinking water and patients in Bulgaria. Probl Inf Parasit Dis 35(1):13–17
Pemberton JM, Kidd SP, Scmidt R (1997) Secreted enzymes of Aeromonas. FEMS Microbiol Lett 152(1):1–10
Pettigrew MM, Fennie KP, York MP, Daniels J, Ghaffar F (2006) Variation in the presence of neuraminidase genes among Streptococcus pneumoniae isolates with identical sequence types. Infect Immun 74(6):3360–3365. doi:10.1128/IAI.01442-05
Reith ME, Singh RK, Curtis B, Boyd JM, Bouevitch A, Kimball J, Munholland J, Murphy C, Sarty D, Williams J, Nash JH, Johnson SC, Brown LL (2008) The genome of Aeromonas salmonicida subsp. salmonicida A449: insights into the evolution of a fish pathogen. BMC Genomics 9:427
Straus DC, Portnoy-Duran C (1983) Neuraminidase production by a Streptococcus sanguis strain associated with subacute bacterial endocarditis. Infect Immun 41(2):507–515
Tekedar HC, Waldbieser GC, Karsi A, Liles MR, Griffin MJ, Vamenta S, Sonstegard T, Hossain M, Schroeder SG, Khoo L, Lawrence ML (2013) Complete genome sequence of a channel catfish epidemic isolate, Aeromonas hydrophila strain ML09-119. Genome Announc 1(5):e00755–13. doi:10.1128/genomeA.00755-13
Thompson H, Homer K, Rao S, Booth V, Hosie AHF (2009) An orthologue of Bacteroides fragilis NanH is the principal sialidase in Tannerella forsythia. J Bacteriol 191(11):3623–3628. doi:10.1128/JB.01618-08
Traving C, Schauer R (1998) Structure, function and metabolism of sialic acids. Cell Mol Life Sci 54:1330–1349. doi:10.1007/s000180050258
Uchida Y, Tsukada Y, Sugimori T (1977) Distribution of neuraminidase in Arthrobacter and its purification by affinity chromatography. J Biochem 82(5):1425–1433
Vertiev YV, Ezepchuk YV, Abrashev IR, Khorlin AY, Krasnova IN (1975) Purification and characterization of neuraminidase produced by NAG-vibrio. Russ J Bioorg Chem 1(11):1639–1645
Vertiev YV, Krasnova IN, Ezepchuk YV (1978) Neuraminidase activity of NAG vibrios and of some representatives of the intestinal group of bacteria. Folia Microbiol 23(6):433–437. doi:10.1007/BF02885571
von Nicolai H, Hamman R, Salehnia S, Zilliken F (1984) A newly discovered sialidase from Gardnerella vaginalis. Zentralbl Bacteriol Microbiol Hyg A 258(1):20–26. doi:10.1016/S0176-6724(84)80003-6
Wang P, Schafer D, Miller CA, Tanenbaum SW, Flashner M (1978) Induction and regulation of neuraminidase synthesis in Arthrobacter sialophilus. J Bacteriol 136(3):874–879
Wang Q, Chang BJ, Mee BJ, Riley TV (2005) Neuraminidase production by Erysipelothrix rhusiopathiae. Vet Microbiol 107:265–272. doi:10.1016/j.vetmic.2005.01.022
Xu G, Li X, Andrew PW, Taylor GL (2008) Structure of the catalytic domain of Streptococcus pneumoniae sialidase NanA. Acta Crystallogr Sect F: Struct Biol Cryst Commun 64(Pt 9):772–775. doi:10.1107/S1744309108024044
Acknowledgments
This work was supported by grant G-2-02/05 from the Ministry of Education and Science, Science Research Fund, Bulgaria. We thank Mr. Dejan Draganov for providing the 4MU-Neu5Ac substrate.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Engibarov, S., Еneva, R. & Abrashev, I. Neuraminidase (sialidase) from Aeromonas sp. strain 40/02 – isolation and partial purification. Ann Microbiol 65, 1515–1523 (2015). https://doi.org/10.1007/s13213-014-0990-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-014-0990-0