Abstract
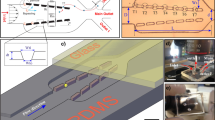
In this study, a microfluidics is integrated with a laser to develop a microfluidic system for single-cell capture and cellular DNA denaturation. The results show that the cells encountered low hydrodynamic stress at low velocities and, thus, could be captured by a cell capture structure. Laser heating of the solution at low velocity produced bubbles that pushed the cells away from the capturing areas. The released cells were subsequently lysed, and cellular DNA was extended and immobilized on the surface of pillars before undergoing laser heating to induce partial denaturation.
Similar content being viewed by others
References
Lin, T.-H. & Yao, D.-J. Applications of EWOD systems for DNA reaction and analysis. J. Adhes. Sci. Tech. 26, 1789–1804 (2012).
Hung, M.-S., Lin, C.-C. & Chen, Y.-W. Design and analysis of single cell capture microfluidic device. J. Chin. Inst. Eng. in press (2013).
Carlo, D.D., Wu, L.Y. & Lee, L.P. Dynamic single cell culture array. Lab Chip 6, 1445–1449 (2006).
Kimura, Y. et al. Dielectrophoresis-assisted massively parallel cell pairing and fusion based on field constriction created by a micro-orifice array sheet. Electrophoresis 32, 2496–2501 (2011).
Tan, W.-H. & Takeuchi, S. Dynamic microarray system with gentle retrieval mechanism for cell-encapsulating hydrogel beads. Lab Chip 8, 259–266 (2008).
Ho, C.-T., Lin, R.-Z., Chang, H.-Y. & Liu, C.-H. Micromachined electrochemical T-switches for cell sorting applications. Lab Chip 5, 1248–1258 (2005).
Maxwell, R.B., Gerhardt, A.L., Toner, M., Gray, M.L. & Schmidt, M.A. A microbubble-powered bioparticle actuator. IEEE J. Microelectromech. Syst. 12, 630–640 (2003).
Liu, Y.K. et al. Evidence for locallized cell heating induced by infrared optical tweezers. Biophys. J. 68, 2137–2144 (1995).
Baaske, P., Duhr, S. & Braun, D. Melting curve analysis in a snapshot. Appl. Phys. Lett. 91, 133901 (2007).
Thamdrup, L.H., Larsen, N.B. & Kristensen, A. Lightinduced local heating for thermophoretic manipulation of dna in polymer micro- and nanochannels. Nano Lett. 10, 826–832 (2010).
Zhao, R. et al. Laser-assisted single-molecule refolding (LASR). Biophys. J. 99, 1925–1931 (2010).
Hung, M.-S., Kurosawa, O. & Washizu, M. Single DNA molecule denaturation using laser-induced heating. Mol. Cell. Probe. 26, 107–112 (2012).
Duhr, S., Arduini, S. & Braun, D. Thermophoresis of DNA determined by microfluidic fluorescence. Eur. Phys. J. E 15, 277–286 (2004).
Boyd, D.A., Adleman, J.R., Goodwin, D.G. & Psaltis, D. Chemical separations by bubble-assisted interphase mass-transfer. Anal. Chem. 80, 2452–2456 (2008).
Hung, M.-S. & Chang, Y.-T. Single cell lysis and DNA extending using electroporation microfluidic device. BioChip J. 6, 84–90 (2012).
Choi, J.S., Piao, Y. & Seo, T.S. Fabrication of various cross-sectional shaped polymer microchannels by a simple PDMS mold based stamping method. BioChip J. 6, 240–246 (2012).
Hung, M.-S. & Chang, J.-H. Developing a microfluidics for rapid protoplasts collection and lysis from plant leaf. Proc. Inst. Mech. Eng. Part N J. Nanoeng. Nanosys. 226, 15–22 (2012).
Terao, K., Kabata, H. & Washizu, M. Extending chromosomal DNA in microstructures using electroosmotic flow. J. Phys. Condens. Matter 18, 653–663 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hung, MS., Huang, YT. Laser-induced heating for cell release and cellular DNA denaturation in a microfluidics. BioChip J 7, 319–324 (2013). https://doi.org/10.1007/s13206-013-7402-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-013-7402-6