Abstract
Purpose
The aim of the study was to compare response evaluation criteria in solid tumours 1.1 (RECIST 1.1), positron emission tomography response criteria in solid tumours (PERCIST), European organisation for research and treatment of cancer (EORTC), and MD Anderson (MDA) criteria for response assessment by Gallium 68-prostate-specific membrane antigen positron emission tomography-computed tomography (Ga68-PSMA PET-CT) in metastatic adenocarcinoma prostate cancer (mPCa) patients with biochemical progression.
Methods
Eighty-eight mPCa patients with pre and post treatment Ga68-PSMA PET-CT were included. A ≥ 25% increase and ≥ 2 ng/ml above the nadir if prostate specific antigen (PSA) drop or ≥ 2 ng/ml above the baseline if PSA does not drop was considered as biochemical progression. RECIST 1.1 and MDA criteria for morphology and PERCIST and EORTC criteria for molecular response were investigated. Percentages of progressive disease (PD), partial response (PR), and stable disease (SD) were calculated. Chi-square test was used for statistical significance.
Results
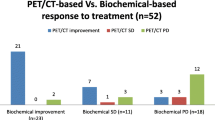
Proportion of PD, SD, and PR by RECIST 1.1 and MDA criteria were 44 (50.57%), 39 (44.83%), 4 (4.6%), and 33 (39.76%), 48 (57.83%), 2 (2.41%) respectively. Proportion of PD, SD, and PR by PERCIST and EORTC criteria were 71 (80.68%), 11 (12.50%), 6 (6.82%), and 74 (84.09%), 8 (9.09%), 6 (6.82%) respectively. Chi-square test showed statistically significant (P < 0.05) higher proportion of progression detected by both molecular criteria as compare to both morphological criteria.
Conclusion
We concluded that for Ga68-PSMA PET-CT response evaluation, molecular criteria performed better than morphological criteria in mPCa patient with PSA progression.



Similar content being viewed by others
Change history
17 August 2020
The following affiliation has been added to the first author, Manoj Gupta.
References
Curran SD, Muellner AU, Schwartz LH. Imaging response assessment in oncology. Cancer Imaging. 2006;6:S126–30.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
Carnaghi C, Sclafani F, Basilico V, Doherty M. Response assessment in oncology: limitations of anatomic response criteria in the era of tailored treatments. Q J Nucl Med Mol Imaging. 2011;55:589–602.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;50(Suppl 1):122S–50.
Eiber M, Maurer T, Souvatzoglou M, Beer AJ, Wester HJ, et al. Evaluation of hybrid 68Ga-PSMA ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56:668–74.
Perera M, Papa N, Christidis D, Hofman MS, Bolton D, et al. Sensitivity, specificity, and predictors of positive 68Ga-prostate-specific membrane antigen positron emission tomography in advanced prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016;70:926–37.
Gupta M, Choudhury PS, Hazarika D, Rawal S. A comparative study of 68Gallium-prostate specific membrane antigen positron emission tomography-computed tomography and magnetic resonance imaging for lymph node staging in high risk prostate cancer patients: an initial experience. World J Nucl Med. 2017;16:186–91.
Hruby G, Eade T, Emmett L, Ho B, Hsiao E, Schembri G, et al. 68 Ga-PSMA-PET/CT staging prior to definitive radiation treatment for prostate cancer. Asia Pac J Clin Oncol. 2018. https://doi.org/10.1111/ajco.12872.
Scher HI, Morris MJ, Stadler WM, et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the Prostate Cancer Clinical Trials Working Group 3. J Clin Oncol. 2016;34:1402–18.
Amor-Coarasa A, Schoendorf M, Meckel M, Vallabhajosula S, Babich JW. Comprehensive quality control of the ITG 68Ge/68Ga generator and synthesis of 68Ga-DOTATOC and 68Ga-PSMA-HBED-CC for clinical imaging. J Nucl Med. 2016;57:1402–5.
Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S, et al. 68Ga-PSMA PET/CT: joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017;44:1014–24.
Young H, Baum R, Cremerius U, et al. Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. European Organization for Research and Treatment of Cancer (EORTC) PET Study Group. Eur J Cancer. 1999;35:1773–82.
Rigaud J, Tiguert R, Le Normand L, Karam G, Glemain P, Buzelin JM, et al. Prognostic value of bone scan in patients with metastatic prostate cancer treated initially with androgen deprivation therapy. J Urol. 2002;168:1423–6.
Soloway MS, Hardeman SW, Hickey D, aymond J, Todd B, Soloway S, et al. Stratification of patients with metastatic prostate cancer based on extent of disease on initial bone scan. Cancer. 1988;61:195–202.
Costelloe CM, Chuang HH, Madewell JE, Ueno NT. Cancer response criteria and bone metastases: RECIST 1.1, MDA and PERCIST. J Cancer. 2010;1:80–92.
Torre LA, Siegel RL, Ward EM, Jemal A. Global cancer incidence and mortality rates and trends-an update. Cancer Epidemiol Biomark Prev. 2016;25:16–27.
Sridhar SS, Freedland SJ, Gleave ME, Higano C, Mulders P, Parker C, et al. Castration-resistant prostate cancer: from new pathophysiology to new treatment. Eur Urol. 2014;65:289–99.
Emmenegger U, Ko Y-J. PSA-based treatment response criteria in castration-resistant prostate cancer: promises and limitations. Can Urol Assoc J. 2009;3:375–6.
Bronsert P, Reichel K, Ruf J. Loss of PSMA expression in non-neuroendocrine dedifferentiated acinar prostate cancer. Clin Nucl Med. 2018;43:526–8.
Seitz AK, Rauscher I, Haller B, Krönke M, Luther S, Heck MM, et al. Preliminary results on response assessment using (68)Ga-HBED-CC-PSMA PET/CT in patients with metastatic prostate cancer undergoing docetaxel chemotherapy. Eur J Nucl Med Mol Imaging. 2017;45:602–12.
Zschaeck S, Wust P, Beck M, Wlodarczyk W, Kaul D, Rogasch J, et al. Intermediate-term outcome after PSMA-PET guided high-dose radiotherapy of recurrent high-risk prostate cancer patients. Radiat Oncol. 2017;12:140.
Baumann R, Koncz M, Luetzen U, Krause F, Dunst J. Oligometastases in prostate cancer: metabolic response in follow-up PSMA-PET-CTs after hypofractionated IGRT. Strahlenther Onkol. 2018;194:318–24.
De Giorgi U, Caroli P, Burgio SL, Menna C, Conteduca V, Bianchi E, et al. Early outcome prediction on 18F-fluorocholine PET/CT in metastatic castration-resistant prostate cancer patients treated with abiraterone. Oncotarget. 2014;5:12448–58.
De Giorgi U, Caroli P, Scarpi E, Conteduca V, Burgio SL, Menna C, et al. (18)F-Fluorocholine PET/CT for early response assessment in patients with metastatic castration-resistant prostate cancer treated with enzalutamide. Eur J Nucl Med Mol Imaging. 2015;42:1276–83.
Schwarzenböck SM, Eiber M, Kundt G, Retz M, Sakretz M, Kurth J, et al. Prospective evaluation of [11C]choline PET/CT in therapy response assessment of standardized docetaxel first-line chemotherapy in patients with advanced castration refractory prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43:2105–13.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Manoj Gupta, Partha Sarathi Choudhury, Harish Chandra Goel, S Avinash Rao, and Sudhir Rawal declare that they have no conflict of interest.
Ethical Statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
The institutional review board waived the need to obtain informed consent for this retrospective study.
Electronic Supplementary Material
ESM 1
(DOCX 33 kb)
Rights and permissions
About this article
Cite this article
Gupta, M., Choudhury, P.S., Rawal, S. et al. Evaluation of RECIST, PERCIST, EORTC, and MDA Criteria for Assessing Treatment Response with Ga68-PSMA PET-CT in Metastatic Prostate Cancer Patient with Biochemical Progression: a Comparative Study. Nucl Med Mol Imaging 52, 420–429 (2018). https://doi.org/10.1007/s13139-018-0548-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-018-0548-3