Abstract
Purpose
We investigated whether 131I whole-body scintigraphy could predict functional changes in salivary glands after radioiodine therapy.
Methods
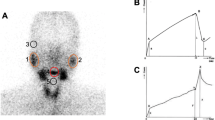
We evaluated 90 patients who received initial high-dose (≥3.7 GBq) radioiodine therapy after total thyroidectomy. All patients underwent diagnostic (DWS) and post-ablation (TWS) 131I whole-body scintigraphy. Visual assessment of salivary radioiodine retention on DWS and TWS was used to divide the patients into two types of groups: a DWS+ or DWS- group and a TWS+ or TWS- group. Salivary gland scintigraphy was also performed before DWS and at the first follow-up visit. Peak uptake and %washout were calculated in ROIs of each gland. Functional changes (Δuptake or Δwashout) of salivary glands after radioiodine therapy were compared between the two groups.
Results
Both peak uptake and the %washout of the parotid glands were significantly lower after radioiodine therapy (all p values <0.001), whereas only the %washout were significantly reduced in the submandibular glands (all p values <0.05). For the parotid glands, the TWS+ group showed larger Δuptake and Δwashout after radioiodine therapy than did the TWS- group (all p values <0.01). In contrast, the Δuptake and Δwashout of the submandibular glands did not significantly differ between the TWS+ and TWS- groups (all p values >0.05). Likewise, no differences in Δuptake or Δwashout were apparent between the DWS+ and DWS- groups in either the parotid or submandibular glands (all p values >0.05).
Conclusion
Salivary gland radioiodine retention on post-ablation 131I scintigraphy is a good predictor of functional impairment of the parotid glands after high-dose radioiodine therapy.


Similar content being viewed by others
References
Schlumberger MJ. Papillary and follicular thyroid carcinoma. N Engl J Med. 1998;338:297–306.
Mazzaferri EL, Jhiang SM. Long-term impact of initial surgical and medical therapy on papillary and follicular thyroid cancer. Am J Med. 1994;97:418–28.
Creach KM, Siegel BA, Nussenbaum B, Grigsby PW. Radioactive iodine therapy decreases recurrence in thyroid papillary microcarcinoma. ISRN Endocrinol. 2012;2012:816386.
Lind P. 131I whole body scintigraphy in thyroid cancer patients. Q J Nucl Med. 1999;43:188–94.
Alexander C, Bader JB, Schaefer A, Finke C, Kirsch CM. Intermediate and long-term side effects of high-dose radioiodine therapy for thyroid carcinoma. J Nucl Med. 1998;39:1551–4.
Walter MA, Turtschi CP, Schindler C, Minnig P, Muller-Brand J, Muller B. The dental safety profile of high-dose radioiodine therapy for thyroid cancer: long-term results of a longitudinal cohort study. J Nucl Med. 2007;48:1620–5.
Cavalieri RR. Iodine metabolism and thyroid physiology: current concepts. Thyroid. 1997;7:177–81.
Mandel SJ, Mandel L. Radioactive iodine and the salivary glands. Thyroid. 2003;13:265–71.
Allweiss P, Braunstein GD, Katz A, Waxman A. Sialadenitis following I-131 therapy for thyroid carcinoma: concise communication. J Nucl Med. 1984;25:755–8.
Hyer S, Kong A, Pratt B, Harmer C. Salivary gland toxicity after radioiodine therapy for thyroid cancer. Clin Oncol (R Coll Radiol). 2007;19:83–6.
Spitzweg C, Harrington KJ, Pinke LA, Vile RG, Morris JC. Clinical review 132: The sodium iodide symporter and its potential role in cancer therapy. J Clin Endocrinol Metab. 2001;86:3327–35.
Macioszek A, Baczyk M, Kopec T, Sowinski J. Salivary gland damage after 131I therapy in patients with differentiated thyroid cancer. Preliminary report. Endokrynol Pol. 2008;59:403–10.
Kim JW, Han GS, Lee SH, Lee DY, Kim YM. Sialoendoscopic treatment for radioiodine induced sialadenitis. Laryngoscope. 2007;117:133–6.
Caglar M, Tuncel M, Alpar R. Scintigraphic evaluation of salivary gland dysfunction in patients with thyroid cancer after radioiodine treatment. Clin Nucl Med. 2002;27:767–71.
Malpani BL, Samuel AM, Ray S. Quantification of salivary gland function in thyroid cancer patients treated with radioiodine. Int J Radiat Oncol Biol Phys. 1996;35:535–40.
An YS, Yoon JK, Lee SJ, Song HS, Yoon SH, Jo KS. Symptomatic late-onset sialadenitis after radioiodine therapy in thyroid cancer. Ann Nucl Med. 2013;27:386–91.
Raza H, Khan AU, Hameed A, Khan A. Quantitative evaluation of salivary gland dysfunction after radioiodine therapy using salivary gland scintigraphy. Nucl Med Commun. 2006;27:495–9.
Kang JY, Jang SJ, Lee WW, Jang SJ, Lee YJ, Kim SE. Evaluation of salivary gland dysfunction using salivary gland scintigraphy in Sjögren’s syndrome patients and in thyroid cancer patients after radioactive iodine therapy. Nucl Med Mol Imaging. 2011;45:161–8.
American Thyroid Association Guidelines Taskforce on Thyroid N, Differentiated Thyroid C, Cooper DS, Doherty GM, Haugen BR, Kloos RT, et al. Revised american thyroid association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19:1167–214.
Chong A, Song H, Min J, Jeong SY, Ha J, Kim J, et al. Improved detection of lung or bone metastases with an I-131 whole body scan on the 7th Day after high-dose I-131 therapy in patients with thyroid cancer. Nucl Med Mol Imaging. 2010;44:273–81.
Prendes MA, Harris A, Wirostko BM, Gerber AL, Siesky B. The role of transforming growth factor beta in glaucoma and the therapeutic implications. Br J Ophthalmol. 2013;97:680–6.
Rigler RG, Scanlon PW. Radiation parotitis from radioactive iodine therapy. Proc Staff Meet Mayo Clin. 1955;30:149–53.
Esfahani AF, Fallahi B, Olamaie R, Eftekhari M, Beiki D, Saghari M. Semi-quantitative assessment of salivary gland function in patients with differentiated thyroid carcinoma after radioiodine-131 treatment. Hell J Nucl Med. 2004;7:206–9.
Lee SM, Lee JW, Kim SY, Han SW, Bae WK. Prediction of risk for symptomatic sialadenitis by post-therapeutic dual (131)I scintigraphy in patients with differentiated thyroid cancer. Ann Nucl Med. 2013;27:700–9.
Woolfenden JM. Thyroid stunning revisited. J Nucl Med. 2006;47:1403–5.
Kolla IS, Alazraki NP, Watts NB. Sialadenitis mimicking metastatic thyroid carcinoma. Clin Nucl Med. 1989;14:564–6.
Carlisle MR, McDougall IR. Dramatic parotid uptake of I-131 on a diagnostic whole-body scan. Clin Nucl Med. 2000;25:895–7.
Acknowledgement
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2012R1A1A2043103).
Conflict of Interest
Kyung Sook Jo, Young-Sil An, Su Jin Lee, Euy-Young Soh, Jeonghun Lee, Yoon-Sok Chung, Dae Jung Kim, Seok-Ho Yoon, Dong Hyun Lee, and Joon-Kee Yoon declare that they have no conflict of interest.
Ethical Statement
All procedures followed were in accordance with the ethical standards of our Institutional Review Board on human experimentation (approval no.: AJIRB-MED-MDB-12-310) and with the Helsinki Declaration of 1975, as revised in 2008. As this is a retrospective study with minimal risk, waiver of informed consent from patients was applied by our Institutional Review Board.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jo, K.S., An, YS., Lee, S.J. et al. Significance of Salivary Gland Radioiodine Retention on Post-ablation 131I Scintigraphy as a Predictor of Salivary Gland Dysfunction in Patients with Differentiated Thyroid Carcinoma. Nucl Med Mol Imaging 48, 203–211 (2014). https://doi.org/10.1007/s13139-014-0274-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13139-014-0274-4