Abstract
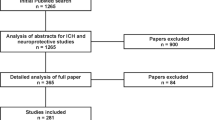
For years, strategies have been proposed to improve translational success in stroke research by improving the quality of animal studies. However, articles that report preclinical intracerebral hemorrhage (ICH) studies continue to lack adequate qualitative and quantitative descriptions of fresh brain tissue collection. They also tend to lack transparency about animal model quality. We conducted a systematic review of 82 ICH research articles to determine the level of detail reported for brain tissue collection. We found that only 24 (29 %) reported the volume, weight, or thickness of tissue collected and a specific description of the anatomical location. Thus, up to 71 % of preclinical ICH research articles did not properly define how fresh specimens were collected for biochemical measurements. Such omissions may impede reproducibility of results between laboratories. Although existing criteria have improved the quality of preclinical stroke studies, ICH researchers need to identify specific guidelines and strategies to avoid pitfalls, minimize bias, and increase reproducibility in this field.

Similar content being viewed by others

References
Manno EM. Update on intracerebral hemorrhage. Continuum. 2012;18(3):598–610. doi:10.1212/01.CON.0000415430.99394.3e.
Rutledge WC, Ko NU, Lawton MT, Kim H. Hemorrhage rates and risk factors in the natural history course of brain arteriovenous malformations. Transl Stroke Res. 2014;5(5):538–42. doi:10.1007/s12975-014-0351-0.
Chen S, Yang Q, Chen G, Zhang JH. An update on inflammation in the acute phase of intracerebral hemorrhage. Transl Stroke Res. 2015;6(1):4–8. doi:10.1007/s12975-014-0384-4.
Xiong XY, Wang J, Qian ZM, Yang QW. Iron and intracerebral hemorrhage: from mechanism to translation. Transl Stroke Res. 2013. doi:10.1007/s12975-013-0317-7.
Dezfulian C, Garrett M, Gonzalez NR. Clinical application of preconditioning and postconditioning to achieve neuroprotection. Transl Stroke Res. 2013;4(1):19–24. doi:10.1007/s12975-012-0224-3.
Pandey AS, Xi G. Intracerebral hemorrhage: a multimodality approach to improving outcome. Transl Stroke Res. 2014;5(3):313–5. doi:10.1007/s12975-014-0344-z.
Wang J. Preclinical and clinical research on inflammation after intracerebral hemorrhage. Prog Neurobiol. 2010;92(4):463–77. doi:10.1016/j.pneurobio.2010.08.001.
Hafez S, Coucha M, Bruno A, Fagan SC, Ergul A. Hyperglycemia, acute ischemic stroke, and thrombolytic therapy. Transl Stroke Res. 2014;5(4):442–53. doi:10.1007/s12975-014-0336-z.
Donnan GA, Hankey GJ, Davis SM. Intracerebral haemorrhage: a need for more data and new research directions. Lancet Neurol. 2010;9(2):133–4. doi:10.1016/S1474-4422(10)70001-6.
Selim M, Sheth KN. Perihematoma edema: a potential translational target in intracerebral hemorrhage? Transl Stroke Res. 2015;6(2):104–6. doi:10.1007/s12975-015-0389-7.
Rosenberg GA, Mun-Bryce S, Wesley M, Kornfeld M. Collagenase-induced intracerebral hemorrhage in rats. Stroke J Cereb Circ. 1990;21(5):801–7.
Yang GY, Betz AL, Chenevert TL, Brunberg JA, Hoff JT. Experimental intracerebral hemorrhage: relationship between brain edema, blood flow, and blood-brain barrier permeability in rats. J Neurosurg. 1994;81(1):93–102. doi:10.3171/jns.1994.81.1.0093.
Clark W, Gunion-Rinker L, Lessov N, Hazel K. Citicoline treatment for experimental intracerebral hemorrhage in mice. Stroke J Cereb Circ. 1998;29(10):2136–40.
Wang J, Rogove AD, Tsirka AE, Tsirka SE. Protective role of tuftsin fragment 1-3 in an animal model of intracerebral hemorrhage. Ann Neurol. 2003;54(5):655–64. doi:10.1002/ana.10750.
Nakamura T, Xi G, Hua Y, Schallert T, Hoff JT, Keep RF. Intracerebral hemorrhage in mice: model characterization and application for genetically modified mice. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab. 2004;24(5):487–94. doi:10.1097/00004647-200405000-00002.
Seifert HA, Pennypacker KR. Molecular and cellular immune responses to ischemic brain injury. Transl Stroke Res. 2014;5(5):543–53. doi:10.1007/s12975-014-0349-7.
Kirkman MA, Allan SM, Parry-Jones AR. Experimental intracerebral hemorrhage: avoiding pitfalls in translational research. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab. 2011;31(11):2135–51. doi:10.1038/jcbfm.2011.124.
Fisher M. The spectrum of translational stroke research. Neurol Res. 2013;35(5):443–7. doi:10.1179/1743132813Y.0000000214.
Landis SC, Amara SG, Asadullah K, Austin CP, Blumenstein R, Bradley EW, et al. A call for transparent reporting to optimize the predictive value of preclinical research. Nature. 2012;490(7419):187–91. doi:10.1038/nature11556.
Schulz KF, Altman DG, Moher D. CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. J Pharmacol Pharmacother. 2010;1(2):100–7. doi:10.4103/0976-500X.72352.
Macleod MR, Fisher M, O’Collins V, Sena ES, Dirnagl U, Bath PM, et al. Good laboratory practice: preventing introduction of bias at the bench. Stroke J Cereb Circ. 2009;40(3):e50–2. doi:10.1161/STROKEAHA.108.525386.
Lapchak PA, Zhang JH, Noble-Haeusslein LJ. RIGOR guidelines: escalating STAIR and STEPS for effective translational research. Transl Stroke Res. 2013;4(3):279–85. doi:10.1007/s12975-012-0209-2.
Wang MM, Xi G, Keep RF. Should the STAIR criteria be modified for preconditioning studies? Transl Stroke Res. 2013;4(1):3–14. doi:10.1007/s12975-012-0219-0.
Bahjat FR, Gesuete R, Stenzel-Poore MP. Steps to translate preconditioning from basic research to the clinic. Transl Stroke Res. 2013;4(1):89–103. doi:10.1007/s12975-012-0223-4.
Turner RJ, Jickling GC, Sharp FR. Are underlying assumptions of current animal models of human stroke correct: from stairs to high hurdles? Transl Stroke Res. 2011;2(2):138–43. doi:10.1007/s12975-011-0067-3.
Harrold JM, Ramanathan M, Mager DE. Network-based approaches in drug discovery and early development. Clin Pharmacol Ther. 2013;94(6):651–8. doi:10.1038/clpt.2013.176.
Qureshi AI, Tuhrim S, Broderick JP, Batjer HH, Hondo H, Hanley DF. Spontaneous intracerebral hemorrhage. N Engl J Med. 2001;344(19):1450–60. doi:10.1056/NEJM200105103441907.
Nagarajan R, Kalinka AT, Hogan WR. Evidence of community structure in biomedical research grant collaborations. J Biomed Inform. 2013;46(1):40–6. doi:10.1016/j.jbi.2012.08.002.
Lo EH. 2013 Thomas Willis Award Lecture: causation and collaboration for stroke research. Stroke J Cereb Circ. 2014;45(1):305–8. doi:10.1161/STROKEAHA.113.001269.
Sukumari-Ramesh S, Alleyne Jr CH, Dhandapani KM. Astrocyte-specific expression of survivin after intracerebral hemorrhage in mice: a possible role in reactive gliosis? J Neurotrauma. 2012;29(18):2798–804. doi:10.1089/neu.2011.2243.
Xu X, Zhang J, Chen X, Liu J, Lu H, Yang P, et al. The increased expression of metabotropic glutamate receptor 5 in subventricular zone neural progenitor cells and enhanced neurogenesis in a rat model of intracerebral hemorrhage. Neuroscience. 2012;202:474–83. doi:10.1016/j.neuroscience.2011.12.008.
Chang CF, Cho S, Wang J. (-)-Epicatechin protects hemorrhagic brain via synergistic Nrf2 pathways. Ann Clin Transl Neurol. 2014;1(4):258–71. doi:10.1002/acn3.54.
Sena E, van der Worp HB, Howells D, Macleod M. How can we improve the pre-clinical development of drugs for stroke? Trends Neurosci. 2007;30(9):433–9. doi:10.1016/j.tins.2007.06.009.
Wu J, Hua Y, Keep RF, Nakamura T, Hoff JT, Xi G. Iron and iron-handling proteins in the brain after intracerebral hemorrhage. Stroke J Cereb Circ. 2003;34(12):2964–9. doi:10.1161/01.STR.0000103140.52838.45.
MacLellan CL, Paquette R, Colbourne F. A critical appraisal of experimental intracerebral hemorrhage research. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab. 2012;32(4):612–27. doi:10.1038/jcbfm.2012.8.
Dirnagl U. Bench to bedside: the quest for quality in experimental stroke research. J Cereb Blood Flow Metab Off J Int Soc Cereb Blood Flow Metab. 2006;26(12):1465–78. doi:10.1038/sj.jcbfm.9600298.
Acknowledgments
This work was supported by grants from the American Heart Association (13GRNT15730001) and the National Institutes of Health (K01AG031926, R01AT007317, and R01NS078026). We thank Claire Levine, MS, ELS, for assistance with manuscript preparation.
Authors’ Contributions
C-F.C. and J.W. conceived and wrote the paper; C-F.C. and L.C. designed the search strategy and selection criteria; C-F.C., L.C., and J.W. performed article identification and evaluation; and C-F.C. performed hemorrhage models for the representative images. All authors revised the manuscript and read and approved the final version before submission.
Compliance with Ethics Requirements
All institutional and national guidelines for the care and use of laboratory animals were followed.
Conflict of Interest
C-F.C. declares that he has no conflict of interest; C.C. declares that she has no conflict of interest; J. W. declares that he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chang, CF., Cai, L. & Wang, J. Translational Intracerebral Hemorrhage: a Need for Transparent Descriptions of Fresh Tissue Sampling and Preclinical Model Quality. Transl. Stroke Res. 6, 384–389 (2015). https://doi.org/10.1007/s12975-015-0399-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-015-0399-5