Abstract
Background
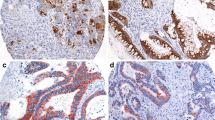
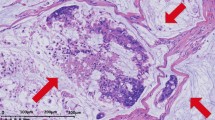
A hallmark of colorectal carcinomas is their ability to secrete mucus. Aberrant expression of mucins and alterations in their glycosylation are associated with the development and progression of malignant diseases. Therefore, mucins can be used as markers of malignancy. Tumor-associated mucins are also used as immunotargets in the treatment of cancer. The present work aimed to study the patterns of expression of MUC1, MUC2 and MUC5AC in colorectal carcinoma using immunohistochemistry and their relationship with site, histological differentiation and stage.
Methods
Fifty cases of colorectal carcinoma were chosen for the study. The histopathology slides were reviewed and blocks were retrieved. Using manual method, tissue microarray blocks were prepared. Immunostaining for MUC1, MUC2 and MUC5AC was performed on slides cut from the tissue microarray block.
Results
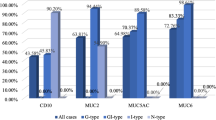
We found that MUC1 expression was upregulated to 39 %, MUC2 expression was downregulated to 43 % and MUC5AC was aberrantly expressed in 24 % of colorectal cancer (CRC). There was a significant correlation between MUC1 positivity and tumor differentiation. As the grade increased from well to moderately differentiated, MUC1 expression increased from 11 % to 55 % (p-value 0.01). There was a statistically significant difference between MUC5AC positivity and grade of tumor (p-value 0.006). The percentage of cases showing MUC5AC expression increased as the stage of disease progressed from 1 to 4. However, there was no significant difference in MUC5AC positivity and stage of CRC (p-value 0.77).
Conclusion
We do not find any correlation between tumor stage or site and MUC1, MUC2 or MUC5AC expression. MUC1and MUC5AC expression showed significant correlation with tumor grade.




Similar content being viewed by others
References
Ferlay J, Bray F, Forman D, et al. Estimates of worldwide burden of cancer in 2008: GLOBOCON 2008. Int J Cancer. 2010;127:2893–917.
Rachagani S, Torres MP, Moniaux N, Batra SK. Current status of mucins in the diagnosis and therapy of cancer. Biofactors. 2009;35:509–27.
Pathak GS, Deshmukh SD, Ashturkar AV. Construction of tissue arrays without prefabricated recipient paraffin block: experience of a novel technique in resource poor settings. Indian J Pathol Microbiol. 2011;54:3–7.
Sule AZ, Mandong BM, Iya D. Malignant colorectal tumors: a ten year review in Jos, Nigeria. West Afr J Med. 2001;20:251–5.
Saidi H, Nyaim EO, Githaiga JW, Karuri D. CRC surgery trends in Kenya, 1993-2005. World J Surg. 2008;32:217–23.
El-Bolkainy TN, Sakr MA, Nouh AA, El-Din NH. A comparative study of rectal and colonic carcinoma: demographic, pathologic and TNM staging analysis. J Egypt Natl Cancer Int. 2006;18:258–63.
Fazeli MS, Adel MG, Lebaschi AH. Colorectal carcinoma: a retrospective descriptive study of age, gender, subsite, stage and differentiation in Iran from 1995 to 2001 as observed in Tehran University. Dis Colon Rectum. 2007;50:990–5.
Mohandas KM. Colorectal cancer in India: controversies, enigmas and primary prevention. Indian J Gastroenterol. 2011;30:3–6.
Hamilton W, Round A, Sharp D, Peters TJ. Clinical features of colorectal cancer before diagnosis: a population based case-control study. Br J Cancer. 2005;93:399–405.
You JF, Hsieh LL, Changchien CR, et al. Inverse effects of mucin on survival of matched hereditary nonpolyposis colorectal cancer and sporadic colorectal cancer patients. Clin Cancer Res. 2006;12:4244–50.
Gomez D, Dalal Z, Raw E, Roberts C, Lyndon PJ. Anatomical distribution of colorectal cancer over a ten year period in a district general hospital: is there a true “rightward shift”? Postgrad Med J. 2004;80:667–9.
Matsuda K, Masaki T, Watanabe T, et al. Clinical significance of MUC1 and MUC2 mucin and p53 protein expression in colorectal carcinoma. Jpn J Clin Oncol. 2000;30:89–94.
Ji BT, Devesa SS, Chow WH, Jin F, Gao YT. Colorectal cancer incidence trends by subsite in urban Shanghai, 1972-1994. Cancer Epidemiol Biomarkers Prev. 1998;7:661–6.
Manne U, Weiss HL, Grizzle WE. Racial differences in the prognostic usefulness of MUC1 and MUC2 in colorectal adenocarcinomas. Clin Cancer Res. 2000;6:4017–25.
Duncan TJ, Watson N, Attar A, Scholefield JH, Durrant LG. The role of MUC1 and MUC3 in the biology and prognosis of colorectal cancer. World J Surg Oncol. 2007;5:31–42.
Baldus SE, Hanisch FG, Kotlarek GM, Zirbes TK, Thiele J, Isenberg J. Co-expression of MUC1 core peptide and the Thomsen Friedenreich antigen in colorectal neoplasms. Cancer. 1998;82:1019–27.
Hiraga Y, Tanaka S, Haruma K, et al. Immunoreactive MUC1 expression at the deepest invasive portion correlates with prognosis of colorectal cancer. Oncology. 1998;55:307–9.
Kimura T, Tanaka S, Haruma K, et al. Clinical significance of MUC1 and E-cadherin expression, cellular proliferation and angiogenesis at the deepest invasive portion of colorectal cancer. Int J Oncol. 2000;16:55–64.
Lugli A, Zlobec I, Baker K, et al. Prognostic significance of mucins in colorectal cancer with different DNA mismatch-repair status. J Clin Pathol. 2006;60:534–39.
Yu XW, Rong W, Xu FL, Xu GY, Sun YR, Feng MY. Expression and clinical significance of mucin and E-cadherin in colorectal tumors. Ai Zheng. 2007;26:1204–10.
Biemer-Huttmann AE, Walsh MD, McGuckin MA, Simms LA, Young J, Leggett BA. Mucin core protein expression in colorectal cancers with high levels of microsatellite instability indicates a novel pathway of morphogenesis. Clin Cancer Res. 2000;6:1909–16.
Li L, Huang P-l, Xiao-jin Y, Xiao-dong B. Clinicopathological significance of mucin 2 immuno-histochemical expression in colorectal cancer: a meta-analysis. Chin J Cancer Res. 2012;24:190–95.
Kocer B, Soran A, Erdoqan S, et al. Expression of MUC5AC in colorectal carcinoma and relationship with prognosis. Pathol Int. 2002;52:470–7.
Sources of support
None
Conflict of interest
MVK, VLG, ARJ, SVB, BPG, and AVK confirm that they have no conflicts of interest to declare.
Ethics statement
The study was performed in a manner to conform to the Helsinki Declaration of 1975, as revised in 2000 and 2008 concerning Human and Animal Rights, and the authors followed the policy concerning Informed Consent as shown on Springer.com.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kesari, M.V., Gaopande, V.L., Joshi, A.R. et al. Immunohistochemical study of MUC1, MUC2 and MUC5AC in colorectal carcinoma and review of literature. Indian J Gastroenterol 34, 63–67 (2015). https://doi.org/10.1007/s12664-015-0534-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-015-0534-y