Abstract
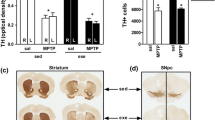
Both clinical and laboratory studies have demonstrated that different types of physical exercise may alleviate Parkinsonism yet evidence for complete restoration of motor function and biomarker integrity are difficult to identify. MPTP (1 × 30 mg/kg, s.c., 4 groups) or saline (vehicle 1 × 5 ml/kg, s.c., 1 group) were administered in a single dose regime over three consecutive weeks on Fridays. Three MPTP groups were given four 30-min periods/week (Mondays to Thursdays), of these two groups, MPTP + Exer + M(i) and MPTP + Exer + M(ii); the former were introduced to exercise and Milmed (oral injection) on the week following the 1st MPTP injection and the latter on the Monday prior to the 1st injection of MPTP onwards. One MPTP group, MPTP + Exer, was given access to exercise (running wheels) from the week following the 1st MPTP injection onwards. The fourth MPTP group, MPTP–NoEx, and the Vehicle group were only given access to exercise on a single day each week (Wednesdays, exercise test) from the week following the 1st MPTP injection onwards. The exercise/exercise + Milmed regime was maintained for a further 9 weeks. It was observed that exercise by itself ameliorated MPTP-induced deficits regarding motor function and dopamine loss only partially whereas in the groups combining exercise with twice weekly dosages of Milmed the MPTP-induced deficits were abolished by the 10th week of the intervention. The three main conclusions that were drawn from correlational analyses of individual mice were: (i) that DA integrity was observed to be a direct function of ability to express running exercise in a treadmill wheel-running arrangement, and (ii) that DA integrity was observed to be a direct function of the capacity for motor performance as measured by spontaneous motor activity and subthreshold l-Dopa (5 mg/kg) induced activity in the motor activity test chambers, and (iii) that the extent to which running exercise in a running wheel was predictive of later motor performance in the activity test chambers was highly convincing.









Similar content being viewed by others
References
Adkins DL, Boychuk J, Remple MS, Kleim JA (2006) Motor training induces experience-specific patterns of plasticity across motor cortex and spinal cord. J Appl Physiol 101:1776–1782
Archer T, Fredriksson A (2003) An antihypokinesic action of α2-adrenoceptors upon MPTP-induced behavior deficits in mice. J Neural Transm 110:183–200
Archer T, Fredriksson A (2006) Influence of noradrenaline denervation upon MPTP-induced deficits in mice. J Neural Transm 113:1119–1129
Archer T, Fredriksson A (2010) Physical exercise attenuates MPTP-induced deficits in mice. Neurotox Res 18:313–327
Archer T, Fredriksson A (2012) Delayed exercise-induced functional and neurochemical partial restoration following MPTP. Neurotox Res. doi:10.1007/s12640-011-9261-z
Archer T, Fredriksson A (2013a) Pharmacogenomics and personalized medicine in Parkinsonism. In: Barh D (ed) Pharmacogenomics. Springer, Berlin
Archer T, Fredriksson A (2013b) Physical exercise as intervention in Parkinsonism. In: Kostrzewa RM (ed) Handbook of neurotoxicity. Springer, New York
Archer T, Fredriksson A, Jonsson G, Lewander T, Mohammed AK, Ross SB, Söderberg U (1986) Central noradrenaline depletion antagonizes aspects of d-amphetamine-induced hyperactivity in the rat. Psychopharmacology 88:141–146
Archer T, Johansson B, Fredriksson A (2011) Exercise alleviates Parkinsonism: clinical and laboratory evidence. Acta Neurolog Scand. doi:10.1111/j.1600-0404.2010.01360.x
Arendash GW (2012) Transcranial electromagnetic treatment against Alzheimer’s disease: why it has the potential to trump Alzheimer’s disease drug development. J Alzheimers Dis 32:243–266. doi:10.3233/JAD-2012-120943
Arendash GW, Sanchez-Ramos J, Mori T, Mamcarz M, Lin X, Runfeldt M, Wang L, Zhang G, Sava V, Tan J, Cao C (2010) Electromagnetic field treatment protects against and reverses cognitive impairment in Alzheimer’s disease mice. J Alzheimers Dis 19:191–210. doi:10.3233/JAD-2010-1228
Baptista PP, de Senna PN, Paim MF, Saur L, Blank M, do Nascimento P, Ilha J, Vianna MR, Mestriner RG, Achaval M, Xavier LL (2013) Physical exercise down-regulated locomotor side effects induced by haloperidol treatment in Wistar rats. Pharmacol Biochem Behav 104:113–118. doi:10.1016/j.pbb.2012.12.020
Bilowit DS (1956) Establishing physical objectives in rehabilitation of patients with Parkinson’s disease. Phys Ther Rev 36:176–178
Björk L, Lindgren S, Hacksell U, Lewander T (1991) (S)-UH-301 antagonises ®-8-OH-DPAT-induced cardiovascular effects in the rat. Eur J Pharmacol 199:367–370
Braun RJ, Büttner S, Ring J, Kroemer G, Madeo F (2009) Nervous yeast: modeling neurotoxic cell death. Trends Biochem Sci 35:135–144
Carmona-Gutierrez D, Jungwirth H, Eisenberg T, Madeo F (2010) Cell cycle control of cell death in yeast. Cell Cycle 9:4046
Corcos DM, Robichaud JA, David FJ, Leurgans SE, Vaillancourt DE, Poon C, Rafferty MR, Kohrt WM, Comella CL (2013) A two-year randomized controlled trial of progressive resistance exercise for Parkinson’s disease. Mov Disord. doi:10.1002/mds.25380
Fisher BE, Petzinger GM, Nixon K, Hogg E, Bremmer S, Meshul CK, Jakowec MW (2004) Exercise-induced behavioral recovery and neuroplasticity in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse basal ganglia. J Neurosci Res 77:378–390
Fredriksson A, Archer T (1994) MPTP-induced behavioural and biochemical deficits: a parametric analysis. J Neural Transm Park Dis Dement Sect 7:123–132
Fredriksson A, Archer T (2003) Effect of postnatal iron administration on MPTP-induced behavioural deficits and neurotoxicity: behavioural enhancement by l-Dopa-MK-801 co-administration. Behav Brain Res 139:31–46
Fredriksson A, Plaznik A, Sundström E, Jonsson G, Archer T (1990) MPTP-induced hypoactvity in mice: reversal by l-Dopa. Pharmacol Toxicol 67:295–301
Fredriksson A, Palomo T, Chase TN, Archer T (1999) Tolerance to a suprathreshold dose of l-Dopa in MPTP mice: effects of glutamate antagonists. J Neural Transm 106:283–300
Fredriksson A, Stigsdotter IM, Hurtig A, Ewalds-Kvist B, Archer T (2011) Running wheel activity restores MPTP-induced functional deficits. J Neural Transm 18:407–420
Fusco S, Maulucci G, Pani G (2012) Sirt1: def-eating senescence? Cell Cycle 11:4135–4146. doi:10.4161/cc.22074
Gedymin LE, Golant MB, Kuznetsov AP, Mudrik DG, Kolpikova TV, Balakireva LZ (1999) The use of yeast living cells as a biological retranslation media for the therapeutic EHF effects. Millim Waves Biol Med 16:10–15 (in Russian)
Golant MB (1994) Physical laws of medicine and their use in the realization of living organisms with EHF radiation. Radiophys Quantum Electron 37:45–47
Golant MB, Mudrik DG, Kruglyakova OP, Izvol’skaya VE (1994) Effect of EHF-radiation polarization on yeast cells. Radiophys Quantum Electron 37:82–84. doi:10.1007/BF01039307
Gorton LM, Vuckovic MG, Vertelkina N, Petzinger GM, Jakowec MW, Wood RI (2010) Exercise effects on motor and affective behavior and catecholamine neurochemistry in the MPTP-lesioned mouse. Behav Brain Res 213:253–262. doi:10.1016/j.bbr.2010.05.009
Heikkila RE, Sieber B-A, Manzino L, Sonsalla PK (1989) Some features of the nigrostriatal dopaminergic neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) in the mouse. Mol Chem Neuropathol 10:171–183
Hunsberger JG, Newton SS, Bennett AH, Duman CH, Russell DS, Salton SR, Duman RS (2007) Antidepressant actions of the exercise-regulated gene VGF. Nat Med 13:1476–1482
Hurwitz A (1989) The benefit of a home exercise regime for ambulatory Parkinson’s disease patients. J Neurosci Nurs 21:180–184
Jackson-Lewis V, Przedborski S (2007) Protocol for the MPTP mouse model of Parkinson’s disease. Nat Protoc 2:141–151
Jackson-Lewis V, Jakowec M, Burke RE, Przedborski S (1995) Time course and morphology of dopaminergic neuronal death caused by the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. Neurodegeneration 4:257–269
Jones-Humble SA, Morgan PF, Cooper BR (1994) The novel anticonvulsant lamotrigine prevents dopamine depletion in C57 black mice in the MPTP animal model of Parkinson’s disease. Life Sci 54:245–252
Jonsson G, Sundström E, Mefford I, Olson L, Johnson S, Freedman R, Hoffer B (1985) Electrophysiological and neurochemical correlates of the neurotoxic effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) on central catecholamine neurons in the mouse. Naunyn Schmiedebergs Arch Pharmacol 331:1–6
Kirk R (1995) Experimental design: procedures for the behavioral sciences. Brooks/Cole, Belmont
Kolosova LI, Akoev GN, Ryabchikova OV, Avelev VD (1998) Effect of low-intensity millimeter-range electromagnetic irradiation on the recovery of function in lesioned sciatic nerves in rats. Neurosci Behav Physiol 28:26–30
Langston JW (1985) MPTP neurotoxicity: an overview and characterization of phases of toxicity. Life Sci 36:201–206
Lau YS, Patki G, Das-Panja K, Le WD, Ahmad SO (2011) Neuroprotective effects and mechanisms of exercise in a chronic mouse model of Parkinson’s disease with moderate neurodegeneration. Eur J Neurosci 33:1264–1274. doi:10.1111/j.1460-9568.2011.07626.x
Liu Y, Yu H, Mohell N, Nordvall G, Lewander T, Hacksell U (1995) Derivatives of cis-2-amino-8-hydroxy-1-methyltetralin: mixed 5-HT1A-receptor agonists and dopamine D2-receptor antagonists. J Med Chem 38:150–160
Mahajan SS, Leko V, Simon JA, Bedalov A (2011) Sirtuin modulators. Handb Exp Pharmacol 206:241–255. doi:10.1007/978-3-642-21631-2_11
Morishima M, Harada N, Hara S, Sano A, Seno H, Takahashi A, Marita Y, Nakaya Y (2006) Monoamine oxidase A activity and norepinephrine level in hippocampus determines hyperwheel running in SPORTS rats. Neuropsychopharmacology 31:2627–2638
Nielsen J, Jewett MC (2008) Impact systems biology on metabolic engineering of Saccharomyces cerevisiae. FEMS Yeast Res 8:122–131
O’Dell SJ, Gross NB, Fricks AN, Casiano BD, Nguyen TB, Marshall JF (2007) Running wheel exercise enhances recovery from nigrostriatal dopamine injury without inducing neuroprotection. Neuroscience 144:1141–1151
Ödman T, Ödman N, Rabotchi E, Archer T (2013) Design experiments of a wireless performance measuring system for different sizes of mammals. Int J Commun Antenn Propag (IRECAP) (submitted)
Ogawa N, Mizukawa K, Hirose Y, Kajita S, Ohara S, Watanabe Y (1987) MPTP-induced parkinsonian model in mice: biochemistry, pharmacology and behavior. Eur Neurol 26(Suppl 1):16–23
Oscarson S, Archer T, Fredriksson A (2009) Physical exercise and Milmed: synergism and neuroreparation in antiparkinsonian effects. In: Abstracts of staging neuropsychiatric disorders: implications for etiopathogenesis and treatment. Fundacion Cerebro y Mente, Mojacar, Spain, p 88
Outeiro TF, Lindquist S (2003) Yeast cells provide insight into alpha-synuclein biology and pathobiology. Science 302:1772–1775
Palmer SS, Mortimer JA, Webster DD, Bistevins R, Dickinson GL (1986) Exercise therapy for Parkinson’s disease. Arch Phys Med Rehab 67:741–745
Patki G, Lau YS (2011) Melatonin protects against neurobehavioral and mitochondrial deficits in a chronic mouse model of Parkinson’s disease. Pharmacol Biochem Behav 99:704–711. doi:10.1016/j.pbb.2011.06.026
Petranovic D, Tyo K, Vemuri GN, Nielsen J (2010) Prospects of yeast systems biology for human health: integrating lipid, protein and energy metabolism. FEMS Yeast Res 10:1046–1059
Petzinger GM, Walsh JP, Akopian G, Hogg E, Abernathy A, Arevalo P, Turnquist P, Vucković M, Fisher BE, Togasaki DM, Jakowec MW (2007) Effects of treadmill exercise on dopaminergic transmission in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. J Neurosci 27:5291–5300
Petzinger GM, Fisher BE, Van Leeuwen JE, Vukovic M, Akopian G, Meshul CK, Holschneider DP, Nacca A, Walsh JP, Jakowec MW (2010) Enhancing neuroplasticity in the basal ganglia: the role of exercise in Parkinson’s disease. Mov Disord 25(Suppl 1):S141–S145. doi:10.1002/mds.22782
Ragimov CR, Ter-Asaturov GP, Golant MB, Rogov KA, Balakireva LZ (1991) Stimulation of reparative osteogenesis by millimeter band electromagnetic radiation in experimental traumatic defects of the mandible. Bull Exp Biol Med 111:562–565
Sebastián JL, Muñoz S, Sancho M, Martínez G, Alvarez G (2011) Electromechanical effects on multilayered cells in nonuniform rotating fields. Phys Rev E: Stat Nonlin Soft Matter Phys 84(1 Pt 1):011926
Shulman LM, Katzel LI, Ivey FM, Sorkin JD, Favors K, Anderson KE, Smith BA, Reich SG, Weiner WJ, Macko RF (2013) Randomized clinical trial of 3 types of physical exercise for patients with Parkinson disease. JAMA Neurol 70:183–190. doi:10.1001/jamaneurol.2013.646
Silva LA, Bom KF, Tromm CB, Rosa GL, Mariano I, Pozzi BG, Tuon T, Stresck EL, Souza CT, Pinho RA (2013) Effect of eccentric training on mitochondrial function and oxidative stress in the skeletal muscle of rats. Braz J Med Biol Res 11:1–7
Sonsalla PK, Heikkila RE (1986) Neurotoxic effects of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and methamphetamine in several strains of mice. Prog Neuropsychopharmacol Biol Psychiatry 12:345–354
Sundström E, Fredriksson A, Archer T (1990) Chronic neurochemical and behavioural changes in MPTP-lesioned C57 BL/6 mice: a model for Parkinson’s disease. Brain Res 528:181–188
Suvarna BS (2013) Sirtuins: the future insight. Kathmandu Univ Med J (KUMJ) 10:77–82
Tenreiro S, Outeiro TF (2010) Simple is good: yeast models of neurodegeneration. FEMS Yeast Res 10:970–979
Tuon T, Valvassori SS, Lopes-Borges J, Luciano T, Trom CB, Silva LA, Quevedo J, Souza CT, Lira FS, Pinho RA (2012) Physical training exerts neuroprotective effects in the regulation of neurochemical factors in an animal model of Parkinson’s disease. Neuroscience 227:305–312. doi:10.1016/j.neuroscience.2012.09.063
Tenreiro S, Munder MC, Alberti S, Outeiro TF (2013) Harnessing the power of yeast to unravel the molecular basis of neurodegeneration. J Neurochem. doi:10.1111/jnc.12271
VanLeeuwen JE, Petzinger GM, Walsh JP, Akopian GK, Vuckovic M, Jakowec MW (2010) Altered AMPA receptor expression with treadmill exercise in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. J Neurosci Res 88:650–668. doi:10.1002/jnr.22216
Vucković MG, Wood RI, Holschneider DP, Abernathy A, Togasaki DM, Smith A, Petzinger GM, Jakowec MW (2010) Memory, mood, dopamine, and serotonin in the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned mouse model of basal ganglia injury. Neurobiol Dis 32(2):319–327. doi:10.1016/j.nbd.2008.07.015
Waters RP, Renner KJ, Pringle RB, Summers CH, Britton SL, Koch LG, Swallow JG (2008) Selection for aerobic capacity affects corticosterone, monoamines and wheel-running activity. Physiol Behav 18:1044–1054
Zhang J, Vemuri G, Nielsen J (2010) Systems biology of energy homeostasis in yeast. Curr Opin Microbiol 13:382–388
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Archer, T., Fredriksson, A. The Yeast Product Milmed Enhances the Effect of Physical Exercise on Motor Performance and Dopamine Neurochemistry Recovery in MPTP-Lesioned Mice. Neurotox Res 24, 393–406 (2013). https://doi.org/10.1007/s12640-013-9405-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-013-9405-4