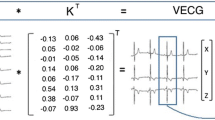
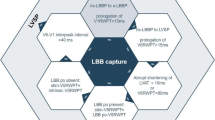
Abstract
Left ventricular (LV) dyssynchrony is a key pathophysiology in the era of cardiac resynchronization therapy (CRT). Left bundle branch block (LBBB) is the main substrate for CRT, and understanding the electrical pathophysiology is important in assessing the effects of CRT. Three-dimensional voltage mapping systems clearly demonstrate the typical propagation pattern characterized as propagation from the mid or apical septum to the lateral or posterior wall through the apex, which appears as a U shape. The electrical characteristics in LBBB closely associate with mechanical dyssynchrony, which is visualized as a septal flash motion. This rapid motion can be detected well by M-mode, tissue Doppler, and speckle tracking imaging. However, intraventricular discoordination between the septum and free wall is also a key to the response to CRT. We classified M-mode septum images into 10 patterns and septal strain pattern into two patterns. Through detailed analysis, we found that septal contraction contributes to intraventricular coordination. Therefore, in addition to septal flash, subsequent analysis of wall motion patterns also provides additional information about myocardial contractibility and the severity of electrical dyssynchrony. Recently, 3-dimensional speckle tracking imaging was introduced and used as a novel method to image electromechanical coupling. Because activation imaging by 3-dimensional speckle tracking can visualize similar U-shaped propagation images to those by 3-dimensional voltage mapping systems, it is hoped that this method will contribute to further research. Until now, it has not been fully understood how electrical dyssynchrony is expressed as mechanical abnormalities; therefore, continuous study will be required in the future.






Similar content being viewed by others
References
Epstein AE, DiMarco JP, Ellenbogen KA, et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2013;61:e6–75.
Tawara S. Das Reizleitungssystem des Säugetierherzens. Eine anatomisch–histologische Studie über das Atrioventricularbundel und die Purkinjeschen Faden. Jena, Gustav Fischer, 1906. (Tawara S. The conduction system of the mammalian heart. Suma K, Shimada M, trans. London, UK: Imperial College Press, 2000).
Ansari A, Ho SY, Anderson RH. Distribution of the Purkinje fibres in the sheep heart. Anat Rec. 1999;254:92–7.
Boyett MR. ‘And the beat goes on.’ The cardiac conduction system: the wiring system of the heart. Exp Physiol. 2009;94:1035–49.
Myerburg RJ, Nilsson K, Gelband H. Physiology of canine intraventricular conduction and endocardial excitation. Circ Res. 1972;30:217–43.
Vassallo JA, Cassidy DM, Marchlinski FE, et al. Endocardial activation of left bundle branch block. Circulation. 1984;69:914–23.
Strauss DG, Selvester RH, Wagner GS. Defining left bundle branch block in the era of cardiac resynchronization therapy. Am J Cardiol. 2011;107:927–34.
Auricchio A, Fantoni C, Regoli F, et al. Characterization of left ventricular activation in patients with heart failure and left bundle-branch block. Circulation. 2004;109:1133–9.
Duckett SG, Camara O, Ginks MR, et al. Relationship between endocardial activation sequences defined by high-density mapping to early septal contraction (septal flash) in patients with left bundle branch block undergoing cardiac resynchronization therapy. Europace. 2012;14:99–106.
Little WC, Reeves RC, Arciniegas J, et al. Mechanism of abnormal interventricular septal motion during delayed left ventricular activation. Circulation. 1982;65:1486–91.
Leenders GE, Lumens J, Cramer MJ, et al. Septal deformation patterns delineate mechanical dyssynchrony and regional differences in contractility: analysis of patient data using a computer model. Circ Heart Fail. 2012;5:87–96.
Sakamaki F, Seo Y, Atsumi A, et al. Novel dyssynchrony evaluation by M-mode imaging in left bundle branch block and the application to predict responses for cardiac resynchronization therapy. J Cardiol. 2014;64:199–206.
Søgaard P, Egeblad H, Kim WY, et al. Tissue Doppler imaging predicts improved systolic performance and reversed left ventricular remodeling during long-term cardiac resynchronization therapy. J Am Coll Cardiol. 2002;40:723–30.
Gorcsan J 3rd, Kanzaki H, Bazaz R, et al. Usefulness of echocardiographic tissue synchronization imaging to predict acute response to cardiac resynchronization therapy. Am J Cardiol. 2004;93:1178–81.
Yu CM, Zhang Q, Fung JW, et al. A novel tool to assess systolic asynchrony and identify responders of cardiac resynchronization therapy by tissue synchronization imaging. J Am Coll Cardiol. 2005;45:677–84.
Bax JJ, Bleeker GB, Marwick TH et al. Left ventricular dyssynchrony predicts response and prognosis after cardiac resynchronization therapy. J Am Coll Cardiol. 2004;44:1834–40.
Chung ES, Leon AR, Tavazzi L, et al. Results of the predictors of response to CRT (PROSPECT) trial. Circulation. 2008;117:2608–16.
Seo Y, Ito H, Nakatani S, et al. The role of echocardiography in predicting responders to cardiac resynchronization therapy: results from the Japan cardiac resynchronization therapy registry trial (J-CRT). Circ J. 2011;75:1156–63.
Seo Y, Ishizu T, Sakamaki F, et al. Analysis of the origin of cardiac wall motion that constitutes myocardial velocity–time curves in patients with left bundle branch block. J Am Soc Echocardiogr. 2009;22:331–6.
Miyazaki C, Lin G, Powell BD, et al. Strain dyssynchrony index correlates with improvement in left ventricular volume after cardiac resynchronization therapy better than tissue velocity dyssynchrony indexes. Circ Cardiovasc Imaging. 2008;1:14–22.
Sakamaki F, Seo Y, Ishizu T, et al. Tissue Doppler imaging dyssynchrony parameter derived from the myocardial active wall motion improves prediction of responders for cardiac resynchronization therapy. Circ J. 2012;76:689–97.
Amundsen BH, Helle-Valle T, Edvardsen T, et al. Noninvasive myocardial strain measurement by speckle tracking echocardiography: validation against sonomicrometry and tagged magnetic resonance imaging. J Am Coll Cardiol. 2006;47:789–93.
Korinek J, Kjaergaard J, Sengupta PP, et al. High spatial resolution speckle tracking improves accuracy of 2-dimensional strain measurements: an update on a new method in functional echocardiography. J Am Soc Echocardiogr. 2007;20:165–70.
Kwan J. Three-dimensional echocardiography: a new paradigm shift. J Echocardiogr. 2014;12:1–11.
Seo Y, Yamasaki H, Kawamura R, et al. Left ventricular activation imaging by 3-dimensional speckle-tracking echocardiography. Comparison with electrical activation mapping. Circ J. 2013;77:2481–9.
Fung JW, Chan JY, Yip GW, et al. Effect of left ventricular endocardial activation pattern on echocardiographic and clinical response to cardiac resynchronization therapy. Heart. 2007;93:432–7.
Sipahi I, Chou JC, Hyden M, et al. Effect of QRS morphology on clinical event reduction with cardiac resynchronization therapy: meta-analysis of randomized controlled trials. Am Heart J. 2012;163:260–7.
Seo Y, Ishizu T, Sakamaki F, et al. Mechanical dyssynchrony assessed by speckle tracking imaging as a reliable predictor of acute and chronic response to cardiac resynchronization therapy. J Am Soc Echocardiogr. 2009;22:839–46.
Conflict of interest
Yoshihiro Seo, Tomoko Ishizu, Fumiko Sakamaki, Masayoshi Yamamoto, and Kazutaka Aonuma declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seo, Y., Ishizu, T., Sakamaki, F. et al. Left bundle branch block and echocardiography in the era of CRT. J Echocardiogr 13, 6–14 (2015). https://doi.org/10.1007/s12574-014-0233-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12574-014-0233-1