Abstract
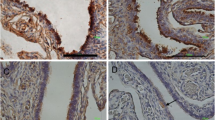
Five samples of human midterm fetal uterus and fallopian tube (four donor bodies) were used to assess whether or not processes of angiogenesis are guided by endothelial tip cells (ETCs), and if cytokine-receptors, such as CD117/c-kit and PDGFR-α, are expressed in the microenvironment of the endothelial tubes. CD34 labeled microvessels in the uterine wall (myometrium and endometrium) and in the wall of the uterine (fallopian) tube, and accurately identified ETCs in both organs. We conclude that sprouting angiogenesis in the developing human female tract is guided by ETCs. Moreover, CD117/c-kit antibodies labeled mural networks of pericytes, α-SMA-positive and desmin-negative, related to the endometrial (but not myometrial) microvessels, and similar labeling was identified in the wall of the uterine tube. PDGFR-α positive labeling, stromal and pericytary, was also found. Thus, sprouting angiogenesis in human fetal genital organs appears to be guided by tip cells and is influenced by tyrosine kinase receptor signaling.



Similar content being viewed by others
References
Adams RH, Eichmann A (2010) Axon guidance molecules in vascular patterning. Cold Spring Harb Perspect Biol 2:a001875
Cai X, Lin Y, Friedrich CC et al (2009) Bone marrow derived pluripotent cells are pericytes which contribute to vascularization. Stem Cell Rev 5:437–445
Cao R, Brakenhielm E, Li X et al (2002) Angiogenesis stimulated by PDGF-CC, a novel member in the PDGF family, involves activation of PDGFR-alphaalpha and -alphabeta receptors. FASEB J 16:1575–1583
Crisan M, Corselli M, Chen WC, Peault B (2012) Perivascular cells for regenerative medicine. J Cell Mol Med. doi: 10.1111/j.1582-4934.2012.01617.x
Dentelli P, Rosso A, Balsamo A et al (2007) C-KIT, by interacting with the membrane-bound ligand, recruits endothelial progenitor cells to inflamed endothelium. Blood 109:4264–4271
Gerhardt H, Betsholtz C (2003) Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res 314:15–23
Gerhardt H, Betsholtz C (2005) How do endothelial cells orientate? EXS 94:3–15
Greenberg JI, Shields DJ, Barillas SG et al (2008) A role for VEGF as a negative regulator of pericyte function and vessel maturation. Nature 456:809–813
Hall AP (2006) Review of the pericyte during angiogenesis and its role in cancer and diabetic retinopathy. Toxicol Pathol 34:763–775
Hellstrom M, Kalen M, Lindahl P, Abramsson A, Betsholtz C (1999) Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development 126:3047–3055
Jindatip D, Fujiwara K, Kouki T, Yashiro T (2012) Transmission and scanning electron microscopy study of the characteristics and morphology of pericytes and novel desmin-immunopositive perivascular cells before and after castration in rat anterior pituitary gland. Anat Sci Int 87:165–173
Melo SA, Kalluri R (2012) Angiogenesis is controlled by miR-27b associated with endothelial tip cells. Blood 119:2439–2440
Morikawa S, Ezaki T (2011) Phenotypic changes and possible angiogenic roles of pericytes during wound healing in the mouse skin. Histol Histopathol 26:979–995
Morikawa S, Baluk P, Kaidoh T, Haskell A, Jain RK, McDonald DM (2002) Abnormalities in pericytes on blood vessels and endothelial sprouts in tumors. Am J Pathol 160:985–1000
Nehls V, Drenckhahn D (1991) Heterogeneity of microvascular pericytes for smooth muscle type alpha-actin. J Cell Biol 113:147–154
Roskoski R Jr (2007) Sunitinib: a VEGF and PDGF receptor protein kinase and angiogenesis inhibitor. Biochem Biophys Res Commun 356:323–328
Siemerink MJ, Klaassen I, Vogels IM, Griffioen AW, van Noorden CJ, Schlingemann RO (2012) CD34 marks angiogenic tip cells in human vascular endothelial cell cultures. Angiogenesis 15:151–163
Song S, Ewald AJ, Stallcup W, Werb Z, Bergers G (2005) PDGFRbeta+ perivascular progenitor cells in tumours regulate pericyte differentiation and vascular survival. Nat Cell Biol 7:870–879
Stanescu R, Didilescu AC, Jianu AM, Rusu MC (2012) Angiogenesis in the reparatory mucosa of the mandibular edentulous ridge is driven by endothelial tip cells. Rom J Morphol Embryo 53:375–378
Stefansson IM, Salvesen HB, Akslen LA (2006) Vascular proliferation is important for clinical progress of endometrial cancer. Cancer Res 66:3303–3309
Suciu LC, Popescu BO, Kostin S, Popescu LM (2012) Platelet-derived growth factor receptor-beta-positive telocytes in skeletal muscle interstitium. J Cell Mol Med 16:701–707
Acknowledgment
M.C.R. was supported in this study by the Sectoral Operational Programme Human Resources Development (SOP HRD), financed from the European Social Fund and by the Romanian Government under the contract number POSDRU/89/1.5/S/64153.
Conflict of interest
The authors confirm that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rusu, M.C., Motoc, A.G.M., Pop, F. et al. Sprouting angiogenesis in human midterm uterus and fallopian tube is guided by endothelial tip cells. Anat Sci Int 88, 25–30 (2013). https://doi.org/10.1007/s12565-012-0154-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-012-0154-x