Abstract
Introduction
Coronary heart disease, stroke, and peripheral vascular disease are the most common causes of mortality in patients with type 2 diabetes mellitus (T2DM). The aim of these studies was to assess the potential for pharmacokinetic interaction between dapagliflozin, a sodium glucose co-transporter-2 inhibitor being developed for the treatment of T2DM, and four medications commonly prescribed in patients with T2DM and cardiovascular disease: simvastatin, valsartan, warfarin, and digoxin.
Methods
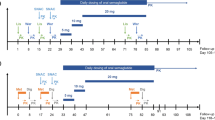
Potential pharmacokinetic interactions between 20 mg dapagliflozin, 40 mg simvastatin, or 320 mg valsartan were assessed in an openlabel, randomized, five-period, five-treatment, unbalanced crossover study in 24 healthy subjects. In a second study, the effects of steadystate dapagliflozin on the pharmacokinetics of 25 mg warfarin or 0.25 mg digoxin were assessed in an open-label, randomized, two-period, two-treatment crossover study in 30 healthy subjects divided into two cohorts. The potential pharmacodynamic interaction between dapagliflozin and warfarin was also evaluated.
Results
All treatments were well tolerated. Neither simvastatin nor valsartan had any clinically meaningful effect on the pharmacokinetics of dapagliflozin. Dapagliflozin increased the area under the curve for simvastatin, simvastatin acid, and valsartan by approximately 19%, 30%, and 6%, respectively, and decreased the maximum observed plasma concentration of valsartan by approximately 6%. These effects were not considered clinically meaningful. In addition, dapagliflozin had no effect on the pharmacokinetics of either digoxin or warfarin. The pharmacodynamics of warfarin were also unaffected by dapagliflozin.
Conclusion
In these studies the co-administration of dapagliflozin and simvastatin, valsartan, warfarin, or digoxin was well tolerated without clinically meaningful drug-drug interaction.
Article PDF
Similar content being viewed by others
References
International Diabetes Federation. The global burden. Available at: http://www.diabetesatlas.org/book/export/html/36. Accessed October 12, 2010.
World Diabetes Foundation. Diabetes facts. Available at: http://www.worlddiabetesfoundation.org/composite-35.htm. Accessed September 1, 2010.
World Health Organization. Global strategy on diet, physical activity and health. Available at: http://www.who.int/dietphysicalactivity/publications/facts/obesity/en/. Accessed April 21, 2010.
Misra A, Ganda OP. Migration and its impact on adiposity and type 2 diabetes. Nutrition. 2007;23:696–708.
Bays HE, Chapman RH, Grandy S. The relationship of body mass index to diabetes mellitus, hypertension and dyslipidaemia: comparison of data from two national surveys. Int J Clin Pract. 2007;61:737–747.
Johansen OE, Birkeland KI. Preventing macrovascular disease in patients with type 2 diabetes mellitus. Am J Cardiovasc Drugs. 2003;3:283–297.
Nathan DM, Buse JB, Davidson MB, et al. Medical management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2009;32:193–203.
Marsenic O. Glucose control by the kidney: an emerging target in diabetes. Am J Kidney Dis. 2009;53:875–883.
Meng W, Ellsworth BA, Nirschl AA, et al. Discovery of dapagliflozin: a potent, selective renal sodiumdependent glucose cotransporter 2 (SGLT2) inhibitor for the treatment of type 2 diabetes. J Med Chem. 2008;51:1145–1149.
Han S, Hagan DL, Taylor JR, et al. Dapagliflozin, a selective SGLT2 inhibitor, improves glucose homeostasis in normal and diabetic rats. Diabetes. 2008;57:1723–1729.
Komoroski B, Vachharajani N, Boulton D, et al. Dapagliflozin, a novel SGLT2 inhibitor, induces dose-dependent glucosuria in healthy subjects. Clin Pharmacol Ther. 2009;85:520–526.
Komoroski B, Vachharajani N, Feng Y, Li L, Kornhauser D, Pfister M. Dapagliflozin, a novel, selective SGLT2 inhibitor, improved glycemic control over 2 weeks in patients with type 2 diabetes mellitus. Clin Pharmacol Ther. 2009;85:513–519.
Bailey CJ, Gross JL, Pieters A, Bastien A, List JF. Effect of dapagliflozin in patients with type 2 diabetes who have inadequate glycaemic control with metformin: a randomised, double-blind, placebo-controlled trial. Lancet. 2010;375:2223–2233.
Ferrannini E, Ramos SJ, Salsali A, Tang W, List JF. Dapagliflozin monotherapy in type 2 diabetic patients with inadequate glycemic control by diet and exercise: a randomized, double-blind, placebo-controlled, phase III trial. Diabetes Care. 2010;33:2217–2224.
List JF, Woo V, Morales E, Tang W, Fiedorek FT. Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes. Diabetes Care. 2009;32:650–657.
Wilding JPH, Norwood P, T’joen C, Bastien A, List JF, Fiedorek FT. A study of dapagliflozin in patients with type 2 diabetes receiving high doses of insulin plus insulin sensitizers. Applicability of a novel insulin-independent treatment. Diabetes Care. 2009;32:1656–1662.
Wilding JPH, Woo V, Soler NG, Pahor A, Sugg J, Parikh S. Sustained effectiveness of dapagliflozin over 48 weeks in patients with type 2 diabetes poorly controlled with insulin [abstract]. Diabetes. 2010;59(Suppl. 1). Abstract 0021-LB.
Stratton IM, Adler AI, Neil HA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321:405–412.
Laakso M. Hyperglycemia and cardiovascular disease in type 2 diabetes. Diabetes. 1999;48:937–942.
Bayturan O, Tuzcu EM, Uno K, et al. Comparison of rates of progression of coronary atherosclerosis in patients with diabetes mellitus versus those with the metabolic syndrome. Am J Cardiol. 2010;105:1735–1739.
Nichols GA, Koro CE, Gullion CM, Ephross SA, Brown JB. The incidence of congestive heart failure associated with antidiabetic therapies. Diabetes Metab Res Rev. 2005;21:51–57.
Obermeier M, Yao M, Khanna A, et al. In vitro characterization and pharmacokinetics of dapagliflozin (BMS-512148), a potent sodiumglucose cotransporter type II inhibitor, in animals and humans. Drug Metab Dispos. 2010;38:405–414.
Nakashima A, Kawashita H, Masuda N, et al. Identification of cytochrome P450 forms involved in the 4-hydroxylation of valsartan, a potent and specific angiotensin II receptor antagonist, in human liver microsomes. Xenobiotica. 2005;35:589–602.
Prueksaritanont T, Gorham LM, Ma B, et al. In vitro metabolism of simvastatin in humans [SBT] identification of metabolizing enzymes and effect of the drug on hepatic P450s. Drug Metab Dispos. 1997;25:1191–1199.
Garnett WR. Interactions with hydroxymethylglutaryl-coenzyme A reductase inhibitors. Am J Health Syst Pharm. 1995;52:1639–1645.
Kaminsky LS, Zhang ZY. Human P450 metabolism of warfarin. Pharmacol Ther. 1997;73:67–74.
Hinderling PH, Hartmann D. Pharmacokinetics of digoxin and main metabolites/derivatives in healthy humans. Ther Drug Monit. 1991;13:381–401.
Gatmaitan ZC, Arias IM. Structure and function of P-glycoprotein in normal liver and small intestine. Adv Pharmacol. 1993;24:77–97.
Simonson SG, Martin PD, Mitchell PD, Lasseter K, Gibson G, Schneck DW. Effect of rosuvastatin on warfarin pharmacodynamics and pharmacokinetics. J Clin Pharmacol. 2005;45:927–934.
O’Reilly RA. Studies on the optical enantiomorphs of warfarin in man. Clin Pharmacol Ther. 1974;16:348–354.
Stern R, Karlis V, Kinney L, Glickman R. Using the international normalized ratio to standardize prothrombin time. J Am Dent Assoc. 1997;128:1121–1122.
Gold JA. Managing diabetic dyslipidemia: testing is step one. WMJ. 2004;103:51, 54.
American Diabetes Association. Treatment of hypertension in adults with diabetes. Diabetes Care. 2002;25:199–201.
Dublin S, Glazer NL, Smith NL, et al. Diabetes mellitus, glycemic control, and risk of atrial fibrillation. J Gen Intern Med. 2010;25:853–858.
Bruno A, Liebeskind D, Hao Q, Raychev R. Diabetes mellitus, acute hyperglycemia, and ischemic stroke. Curr Treat Options Neurol. 2010;12:492–503.
Strojek K, Hruba V, Elze M, Langkilde A, Parikh S. Efficacy and safety of dapagliflozin in patients with type 2 diabetes mellitus and inadequate glycaemic control on glimepiride monotherapy [abstract]. Diabetologia. 2010;53(Suppl. 1):S347.
US Food and Drug Administration. Guidance for industry drug interaction studies-study design, data analysis, and implications for dosing and labeling. Available at: http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/ucm064982.htm. Accessed August 14, 2011.
Soler NG, Wilding JPH, Woo V, et al. Dapagliflozin lowered rate of insulin uptitration/study discontinuation from lack of glycaemic control in 48-week study of type 2 diabetes patients poorly controlled on insulin therapy [abstract]. Diabetologia. 2010;53(Suppl. 1):S348–S349. Abstract 872.
Kasichayanula S, Liu X, Shyu WC, et al. Lack of pharmacokinetic interaction between dapagliflozin, a novel SGLT2 inhibitor, and metformin, pioglitazone, glimepiride, or sitagliptin in healthy subjects. Diabetes Obes Metab. 2011;13:47–54.
Author information
Authors and Affiliations
Corresponding author
Additional information
To view enhanced content go to www.advancesintherapy.com
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Kasichayanula, S., Chang, M., Liu, X. et al. Lack of Pharmacokinetic Interactions Between Dapagliflozin and Simvastatin, Valsartan, Warfarin, or Digoxin. Adv Therapy 29, 163–177 (2012). https://doi.org/10.1007/s12325-011-0098-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-011-0098-x