Abstract
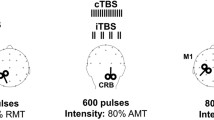
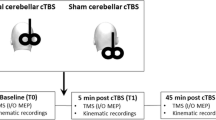
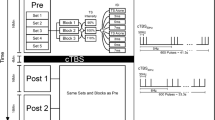
The use of cerebellar repetitive transcranial magnetic stimulation has been attempted for perturbing reflexive and voluntary eye movements, but discrepancies are seen between the results of distinct studies possibly due to the different stimulation sites, intensities, and paradigms. We describe the after effects of 20 and 40 s continuous Theta Burst Stimulation (cTBS) as compared to sham stimulation, applied over the lateral cerebellar vermis and paravermis on Reflexive Saccades (RS) and Smooth Pursuit (SP) eye movements, recorded in the 30 min following stimulation. The experiments were carried out in eight healthy volunteers, and eye movements were recorded monocularly with video-oculography. The 40 s cTBS significantly increased the amplitude of ipsilateral RS and the acceleration of the ipsilateral SP, and this effect was detectable all over the 30-min recording period; 40 s cTBS did not modify the other parameters, namely the peak velocity, the duration and the latency of RS, and the latency and the velocity of SP. The 20 s cTBS was ineffective on all RS and SP parameters. Finally, we detected a significant quite-linear reduction of RS peak velocity over time, but this was independent from cTBS and was probably caused by fatigue. The effects of 40 s cTBS in our experiments mimic the disorder of ocular motility in Wallenberg’s syndrome and could result from functional impairment of cerebellopontine pathways. This effect lasts 30 min at least, and can provide a useful framework for adaptive ocular motor studies.



Similar content being viewed by others
References
Huang YZ, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC. Theta burst stimulation of the human motor cortex. Neuron. 2005;45(2):201–6.
Huang YZ, Rothwell JC, Chen RS, Lu CS, Chuang WL. The theoretical model of theta burst form of repetitive transcranial magnetic stimulation. Clin Neurophysiol. 2011;122(5):1011–8.
Koch G. Repetitive transcranial magnetic stimulation: a tool for human cerebellar plasticity. Funct Neurol. 2010;25(3):159–63.
Koch G, Mori F, Marconi B, Codeca C, Pecchioli C, Salerno S, et al. Changes in intracortical circuits of the human motor cortex following theta burst stimulation of the lateral cerebellum. Clin Neurophysiol. 2008;119(11):2559–69.
Popa T, Russo M, Meunier S. Long-lasting inhibition of cerebellar output. Brain Stimul. 2010;3(3):161–9.
Brusa L, Ponzo V, Mastropasqua C, Picazio S, Bonni S, Di Lorenzo F, et al. Theta burst stimulation modulates cerebellar-cortical connectivity in patients with progressive supranuclear palsy. Brain Stimul. 2014;7(1):29–35.
Colnaghi S, Honeine JL, Sozzi S, Schieppati M. Body Sway Increases After Functional Inactivation of the Cerebellar Vermis by cTBS. Cerebellum. 2016.
Di Lorenzo F, Martorana A, Ponzo V, Bonni S, D'Angelo E, Caltagirone C, et al. Cerebellar theta burst stimulation modulates short latency afferent inhibition in Alzheimer’s disease patients. Front Aging Neurosci. 2013;5:2.
Monaco J, Casellato C, Koch G, D'Angelo E. Cerebellar theta burst stimulation dissociates memory components in eyeblink classical conditioning. Eur J Neurosci. 2014;40(9):3363–70.
Colnaghi S, Ramat S, D'Angelo E, Cortese A, Beltrami G, Moglia A, et al. Theta-burst stimulation of the cerebellum interferes with internal representations of sensory-motor information related to eye movements in humans. Cerebellum. 2011;10(4):711–9.
Jenkinson N, Miall RC. Disruption of saccadic adaptation with repetitive transcranial magnetic stimulation of the posterior cerebellum in humans. Cerebellum. 2010;9(4):548–55.
Panouilleres M, Neggers SF, Gutteling TP, Salemme R, van der Stigchel S, van der Geest JN, et al. Transcranial magnetic stimulation and motor plasticity in human lateral cerebellum: dual effect on saccadic adaptation. Hum Brain Mapp. 2012;33(7):1512–25.
Colnaghi S, Ramat S, D'Angelo E, Versino M. Transcranial magnetic stimulation over the cerebellum and eye movements: state of the art. Funct Neurol. 2010;25(3):165–71.
Hashimoto M, Ohtsuka K. Transcranial magnetic stimulation over the posterior cerebellum during visually guided saccades in man. Brain. 1995;118(Pt 5):1185–93.
Ohtsuka K, Enoki T. Transcranial magnetic stimulation over the posterior cerebellum during smooth pursuit eye movements in man. Brain. 1998;121(Pt 3):429–35.
Nagel M, Behrmann H, Zangemeister WH. Disturbance of predictive response initiation of eye and head movements in cerebellar patients. Eur Neurol. 2008;60(4):179–85.
Rossini PM, Barker AT, Berardelli A, Caramia MD, Caruso G, Cracco RQ, et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord and roots: basic principles and procedures for routine clinical application. Report of an IFCN committee. Electroencephalogr Clin Neurophysiol. 1994;91(2):79–92.
Rothwell JC. Techniques and mechanisms of action of transcranial stimulation of the human motor cortex. J Neurosci Methods. 1997;74(2):113–22.
Tofts PS. The distribution of induced currents in magnetic stimulation of the nervous system. Phys Med Biol. 1990;35(8):1119–28.
Leigh RJ, Zee DS. The neurology of eye movements. Fifth ed. New York: Oxford University Press; 2015.
Ohtsuka K, Noda H. Saccadic burst neurons in the oculomotor region of the fastigial nucleus of macaque monkeys. J Neurophysiol. 1991;65(6):1422–34.
Robinson FR, Straube A, Fuchs AF. Role of the caudal fastigial nucleus in saccade generation. II. Effects of muscimol inactivation. J Neurophysiol. 1993;70(5):1741–58.
Helmchen C, Straube A, Buttner U. Saccadic lateropulsion in Wallenberg’s syndrome may be caused by a functional lesion of the fastigial nucleus. J Neurol. 1994;241(7):421–6.
Kojima Y, Robinson FR, Soetedjo R. Cerebellar fastigial nucleus influence on ipsilateral abducens activity during saccades. J Neurophysiol. 2014;111(8):1553–63.
Eggert T, Robinson FR, Straube A. Modeling inter-trial variability of saccade trajectories: effects of lesions of the oculomotor part of the fastigial nucleus. PLoS Comput Biol. 2016;12(6):e1004866.
Fuchs AF, Robinson FR, Straube A. Participation of the caudal fastigial nucleus in smooth-pursuit eye movements. I Neuronal activity J Neurophysiol. 1994;72(6):2714–28.
Vahedi K, Rivaud S, Amarenco P, Pierrot-Deseilligny C. Horizontal eye movement disorders after posterior vermis infarctions. J Neurol Neurosurg Psychiatry. 1995;58(1):91–4.
Waespe W, Wichmann W. Oculomotor disturbances during visual-vestibular interaction in Wallenberg's lateral medullary syndrome. Brain. 1990;113(Pt 3):821–46.
Gamboa OL, Antal A, Moliadze V, Paulus W. Simply longer is not better: reversal of theta burst aftereffect with prolonged stimulation. Exp Brain Res. 2010;204(2):181–7.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Colnaghi, S., Colagiorgio, P., Ramat, S. et al. After Effects of Cerebellar Continuous Theta Burst Stimulation on Reflexive Saccades and Smooth Pursuit in Humans. Cerebellum 16, 764–771 (2017). https://doi.org/10.1007/s12311-017-0852-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-017-0852-y